* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter 7 - Midway ISD
Rigid rotor wikipedia , lookup
Drug discovery wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Stoichiometry wikipedia , lookup
Chemistry: A Volatile History wikipedia , lookup
Mass spectrometry wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Magnetorotational instability wikipedia , lookup
Nanofluidic circuitry wikipedia , lookup
Homoaromaticity wikipedia , lookup
Atomic theory wikipedia , lookup
Debye–Hückel equation wikipedia , lookup
Gas chromatography–mass spectrometry wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
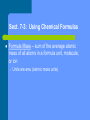
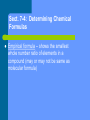
Chapter 7 Chemical Formulas and Chemical Compounds Sect. 7-1: Chemical Names and Formulas Chemical formulas indicate what elements/ions are in the compound and in what ratio Naming monatomic ions – – Cations get the element name Anions get the root of the element name with –ide ending Polyatomic ions get ion name ( pg. 210) Binary Ionic Compounds – Steps for formula writing 1. Write down the symbols for the ions, cation listed first 2. Identify charges and “criss-cross” as needed 3. Re-write formula without showing charges Naming binary ionic compounds 1. 2. 3. Name cation Name anion (remember –ide ending) If cation is multi-valent (can have more than one charge), add roman numeral in parenthesis following cation name to indicate charge Stock system of nomenclature Compounds containing polyatomic Ions Oxyanions – polyatomic ions containing oxygen – – Many are made of same 2 elements, so more common ion gets –ate ending and the one with less oxygen gets –ite ending If more than 2 oxyanions, hypo- is added for less then the –ite and per- is added for more than –ate Naming of compounds is same as binary When more than one polyatomic ion is needed to balance the charges in a formula, parenthesis are required around the polyatomic ion Naming Binary Molecular Compounds “Old” system of naming – – – – Prefixes are used to tell how many of each element are present in the molecular formula Less electronegative element is listed first and is only given a prefix if there is more than one 2nd element is given a prefix, then the root of the element name, and then –ide ending “o” or “a” at end of prefix is dropped when element name starts with a vowel Numerical Prefixes 1 mono2 di3 tri4 tetra5 penta- 6 hexa7 hepta8 octa9 nona10 deca- Rules for naming acids Naming Acids Binary Acids (contains hydrogen and one other element) – Use prefix hydro- to name the hydrogen part of the compound – The rest of the name consists of a form of the root of the element plus the suffix –ic and then the word acid Naming Oxyacids Oxyacid- acid that contains hydrogen and an oxyanion – First determine anion present – If anion ends with –ate, replace with suffix –ic Ex: HNO3; NO3-1 is nitrate – Name is Nitric Acid anion ends with –ite, replace with suffix –ous – If Ex: HNO2; NO2 is nitrite – Name is Nitrous Acid Sect. 7-3: Using Chemical Formulas Formula Mass – sum of the average atomic mass of all atoms in a formula unit, molecule, or ion – Units are amu (atomic mass units) Molar mass for a compound is calculated by summing the masses of all elements present in one mole of the compound Molar mass is numerically equal to formula mass, but units are g/mol Ex: Calculate the molar mass of water Molar mass can be used as a conversion factor to convert between moles and grams (use t-chart method) – 1mol = molar mass in grams Percent by mass of a compound = Mass of element in sample of compound x 100 Mass of sample of compound Can also be calculated by Mass of element in 1 mol of compound x 100 Molar mass of compound Percent composition – the percent by mass of each element in a compound Sect. 7-4: Determining Chemical Formulas Empirical formula – shows the smallest whole number ratio of elements in a compound (may or may not be same as molecular formula) Steps for determining Empirical Formula 1. 2. 3. 4. Determine percent composition (if not already given) Assume 100 g sample and change % to units of grams Convert grams of each element to moles Divide all answers from step #3 by the smallest answer to obtain a whole number ratio If step #4 does not give very close to whole numbers, multiply all answers by the same number to obtain whole numbers – Example 2.5 would be multiplied by 2 to get 5 or 2.3 would be multiplied by 3 to obtain 7 Calculation of a Molecular Formula X (empirical formula) = molecular formula X = molar mass of molecular formula molar mass of empirical formula X must be a whole number