* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Thermodynamics
Marcus theory wikipedia , lookup
Solar air conditioning wikipedia , lookup
Heat transfer wikipedia , lookup
Thermodynamics wikipedia , lookup
Transition state theory wikipedia , lookup
Internal energy wikipedia , lookup
Chemical thermodynamics wikipedia , lookup
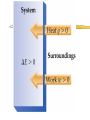
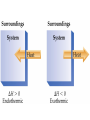
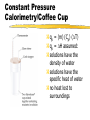
Thermodynamics Thermodynamics responds to four questions: Can the reaction occur spontaneously or must it be driven? How much useful work is produced or consumed when the reaction takes place? How far will the reaction go? How can the reaction be made to advance further? Chemical reactions involve changes in energy. Thermodynamics is the study of energy and its transformations. Force is a push or pull on an object. Work is the product of force applied to an object over a distance. W = F x d. Energy is the work done to move an object against a force. Heat is the transfer of energy between two objects. Energy is the capacity to do work or transfer heat. Heat vs. Temperature Heat is energy that is transferred from one object to another because of a difference in temperature. Temperature is a measure of the average kinetic energy of the molecules of a substance. Kinetic Energy Energy of motion 1/2 mv2 depends on mass and velocity of object Potential Energy Energy by virtue of position relative to other objects (mgh) Stored energy Arises when there is a force acting on an object Potential energy can be converted into kinetic energy. Ex: a bicyclist at the top of a hill. Kinetic and Potential Energy Electrostatic potential energy, Ed, is the attraction between two oppositely charged particles, Q1 and Q2, a distance d apart: Ed = κQ1Q2 d The constant κ = 8.99 x 109 J-m/C2 If the two particles are of similar charge, then Ed is the electrostatic repulsion between them. Units of Energy SI unit = Joule (J) 1 J = 1 kg-m2/s2 2 kg object moving at a speed of 1 m/s has a kinetic energy of 1 J. 1 cal = 4.184 J A nutritional Calorie: 1 Cal = 1000 cal = 1kcal. System and Surroundings System = part of the universe we are interested in; what is under study. Surroundings = everything else in the universe. Energy lost by system = Energy gained by surroundings. Energy out of system = - E Energy into system = + E First Law of Thermodynamics Internal energy: total energy of a system. Cannot measure absolute internal energy. Change in internal energy: E = Efinal - Einitial E - system loses E + system gains First Law of Thermodynamics Energy can neither be created nor destroyed. Systems tend to lose energy to be as stable as possible. Energy of system + surroundings is constant. Any energy lost by a system must be transferred to the surroundings. Energy is exchanged by changes in heat or work. E = q + w The internal energy is the sum of the kinetic and potential energies of all the particles in a system. Internal energy can be changed by work or heat flow or both. q + heat in q - heat out w + work done on w - work done by Calculate the change in energy of the system for a process where the system absorbs 140 J of heat from the surroundings and does 85 J of work on the surroundings. State v. Path Functions State: •property of a system not determined by its history •depends only on its present condition •E is a state function Path: •depends on manner in which process is carried out •q and w are path functions State functions depend only on the initial and final states of the system, not on how the internal energy is used. Enthalpy Chemical reactions have the ability to do work. In chemistry most mechanical work is PV work. When a gas is produced in a reaction, the gas can be used to push a piston, thus doing work. For a constant pressure process, the changes in internal energy and enthalpy are related by the equation: H=E +PV Enthalpy When the pressure is constant, w = -PΔV Like internal energy, enthalpy cannot be determined exactly, but changes in enthalpy (H) can be determined. Consider work done under constant pressure: E = qp - P V or qp = E + P V H = E + P V so qp = H H = qp H, enthalpy, like internal energy is a state function. AKA heat of reaction, the enthalpy of reaction. Enthalpy Enthalpy is the heat exchanged between the system and surroundings at constant pressure, as the result of a chemical reaction. H = H(products) - H(reactants) The enthalpy change of a reaction is equal in magnitude but opposite in sign of the enthalpy change of the reverse reaction: Hforward = - Hreverse The enthalpy change of a reaction depends on the states of matter of the products and reactants. Exothermic Reactions Heat flows out of system Heat is a product System has less energy H is negative to indicate direction of heat flow Energy released making bonds > energy absorbed to break bonds Product bond energy > reactant bond energy Endothermic Reactions Heat flows into system Heat is a reactant System has more energy H is positive to indicate direction of heat flow Energy absorbed to break bonds> energy released making bonds Reactant bond energy > product bond energy Hess’s Law If a reaction is carried out in a series of steps, H for the reaction will be equal to the sum of the enthalpy changes for the individual steps. When solving Hess’s Law problems keep the following in mind: 1. If the reaction is reversed, the sign of H must also be reversed. 2. If the coefficients in a balanced equation are multiplied by an integer, the value of H must also be multiplied by the same integer. Likewise, if the coefficients are divided, then H must also be divided. Hess’s Law For example: CH4(g) + 2O2(g) CO2(g) +2H2O(g) ΔH = -802 kJ 2H2O(g) 2H2O(l) ΔH = -88 kJ CH4(g) + 2O2(g) CO2(g) + 2H2O(l) ΔH = -890 kJ Given: 2C(s) + 2H2(g) C2H4(g) H = ? Use these: C(s) + O2(g) CO2(g) C2H4(g) + 3O2(g) 2CO2(g) + 2H2O H2(g) + ½ O2(g) H2O(l) H = -393.5 kJ -1410.9 -285.6 Diborane, B2H6, is a highly reactive boron hydride, which was once considered a possible rocket fuel. Calculate the H for the synthesis of diborane from its elements using the following information: H 2B(s) + 3/2O2(g) B2O3(g) -1273 kJ B2H6(g) + 3O2(g) B2O3(g) + 3H2O(g) -2035 kJ H2(g) + 1/2O2(g) H2O(l) -286 kJ H2O(l) H2O(g) 44 kJ Standard enthalpy of formation (Hof) …the energy associated with the formation of a compound from its elements in their standard state. Standard state is 25oC and 1 atm. Hof for the most stable form of an element = 0 Horxn = nprod Hof (prod) - nreact Hof (reac) Calculate the change in enthalpy for: 4NH3(g) + 7O2(g) 4NO2(g) + 6H2O(l) Calorimetry Method of measuring heat flow between systems and surroundings. Heat capacity = the amount of heat necessary to raise the temperature of an object by one degree Celsius or Kelvin The heat capacity of one mole of a substance is called its molar heat capacity, joules per mole per degree Celsius or Kelvin Specific heat also known as specific heat capacity is defined as the amount of heat necessary to raise the temperature of 1 g of a substance by one degree. Note that heat capacity applies to objects, while molar and specific heat capacities apply to substances. The device used to measure heat flow between systems and surroundings is called a calorimeter. Constant Pressure Calorimetry/Coffee Cup qp = (m) (Cp) (T) qp = H assumed: solutions have the density of water solutions have the specific heat of water no heat lost to surroundings Constant Volume Calorimetry/Bomb Calorimeter qv = E If given heat capacity of calorimeter and water as one quantity qv = C x T If the heat capacity of the bomb and the amount of water is given qv =(mCT)water + (C x T)bomb A 1.5 gram sample of methane gas was burned with an excess of oxygen in a bomb calorimeter with a heat capacity of 11.3 kJ/oC, the temperature increase was 14.3oC. Calculate the energy of combustion per gram of methane. Combustion of a 0.1584 gram sample of vanillin, C8H8O3, in a calorimeter containing 987 grams of water with a heat capacity of 6.66 J/oC, increased the temperature of the bomb calorimeter 3.25oC. What is the enthalpy of combustion per mole of vanillin? The complete combustion of octane to produce gaseous carbon dioxide and liquid water at standard conditions gives off 47.9 kJ of heat per gram of octane. Write a chemical equation to represent this information. 1. What transfer of energy takes place when: a. a saucepan of water is heated to boiling b. a tennis ball bounces to rest on the floor c. two automobiles collide head on d. a flashlight is left on until the battery runs down e. a fire burns f. a wind turbine generates electricity g. a chemical reaction liberates heat 2. If the temperature of a 50.0-g block of aluminum increases by 10.9 K when heated by 500 J, calculate the a. heat capacity of the aluminum block b. molar heat capacity of aluminum c. specific heat of aluminum 3. If the enthalpy change for the combustion of propane is -2220 kJ/mol propane, what quantity of heat is released when 1 kg of propane is burned? If you have 10 moles each of copper and water– one is very hot and the other is very cold and you simultaneously toss them into the same insulated container, a. Which will gain or lose more heat? b. What will be true of the magnitudes of the ΔT’s? c. If the copper was originally the hotter object, what will be the sign of ΔT for each substance? d. Is this a chemical or physical change? Consider the process: Z2 (g) 2Z (g) which takes place inside a rigid, sealed container at constant temperature. Answer the Following using +, -, or 0. q= w= ΔE = ΔEuniv=