* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Document
Survey
Document related concepts
Transcript
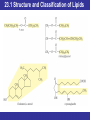
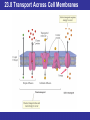
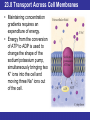
Outline 23.1 Structure and Classification of Lipids 23.2 Fatty Acids and Their Esters 23.3 Properties of Fats and Oils 23.4 Chemical Reactions of Triacylglycerols 23.5 Phospholipids and Glycolipids 23.6 Sterols 23.7 Structure of Cell Membranes 23.8 Transport Across Cell Membranes 23.9 Eicosanoids: Prostaglandins and Leukotrienes © 2013 Pearson Education, Inc. Goals 1. What are the major classes of fatty acids and lipids? Be able to describe the chemical structures and general properties of fatty acids, waxes, fats, sterols, and oils. 2. What reactions do triacylglycerols undergo? Be able to describe the results of hydrogenation and hydrolysis of triacylglycerols, and, given the reactants, predict the products. 3. What are sterols? Be able to identify sterols and their derivatives, describe their structures and roles. 4. What are the membrane lipids? Be able to identify the membrane lipids, describe their structures, and roles. 5. What is the nature of a cell membrane? Be able to describe the general structure of a cell membrane and its chemical composition. 6. How do substances cross cell membranes? Be able to distinguish between passive transport and active transport and between simple diffusion and facilitated diffusion. 7. What are eicosanoids? Be able to describe the general structure of prostaglandins and leukotrienes, and some of their functions. © 2013 Pearson Education, Inc. 23.1 Structure and Classification of Lipids • Lipids are defined by solubility in nonpolar solvents (a physical property) rather than by chemical structure. • There are a great many different kinds and they serve a variety of functions in the body. • Many lipids have hydrocarbon or modified hydrocarbon structure, properties, and behavior. © 2013 Pearson Education, Inc. 23.1 Structure and Classification of Lipids © 2013 Pearson Education, Inc. 23.1 Structure and Classification of Lipids © 2013 Pearson Education, Inc. 23.1 Structure and Classification of Lipids Lipids that are esters or amides of fatty acids • Waxes are carboxylic acid esters with long, straight hydrocarbon chains in both R groups; they are secreted by sebaceous glands in the skin of animals and perform mostly external protective functions. • Triacylglycerols are carboxylic acid triesters of glycerol, a three-carbon trialcohol. Triacylglycerols are found in most dietary fats and oils, and are also the fat storage molecules in our bodies. They are a major source of biochemical energy. • Glycerophospholipids are triesters of glycerol that contain charged phosphate diester groups and are abundant in cell membranes. Together with other lipids, they help to control the flow of molecules into and out of cells. • Sphingomyelins are amides derived from an amino alcohol (sphingosine), also contain charged phosphate-diester groups; essential to the structure of cell membranes and especially abundant in nerve cell membranes. • Glycolipids are different amides derived from sphingosine, contain polar carbohydrate groups; on cell surfaces the carbohydrate portion is recognized and interacts with intercellular messengers. © 2013 Pearson Education, Inc. 23.2 Fatty Acids and Their Esters • Naturally occurring fats and oils are triesters formed between glycerol and fatty acids. • Fatty acids are long, unbranched hydrocarbon chains with a carboxylic acid group at one end. Most have even numbers of carbon atoms. • Those without double bonds are saturated. • Those with double bonds are unsaturated. • In naturally occurring fats and oils, the double bonds are usually cis rather than trans. © 2013 Pearson Education, Inc. 23.2 Fatty Acids and Their Esters • Chemists use a shorthand nomenclature for fatty acids that avoids using the common names. • The notation uses C for carbon followed by the number of carbon atoms present, a colon and the number of unsaturated bonds present. • Palmitic acid (C16:0) and stearic acid (C18:0) are the most common saturated acids; • Oleic (C18:1) and linoleic acids (C18:2) are the most common unsaturated ones. • Polyunsaturated fatty acids have more than one carbon–carbon double bond. © 2013 Pearson Education, Inc. 23.2 Fatty Acids and Their Esters © 2013 Pearson Education, Inc. 23.2 Fatty Acids and Their Esters Waxes • A wax is a mixture of fatty acid–long-chain alcohol esters. • The acids usually have an even number of carbon atoms, generally from 16 to 36 carbons; • The alcohols have an even number of carbon atoms ranging from 24 to 36. © 2013 Pearson Education, Inc. 23.2 Fatty Acids and Their Esters Triacylglycerols • Animal fats and vegetable oils are the most plentiful lipids in nature. • Animal fats are solid, whereas vegetable oils are liquid, but their structures are closely related. • All fats and oils are composed of triesters of glycerol (1,2,3-propanetriol, also known as glycerine) with three fatty acids. • They are named chemically as triacylglycerols, but are often called triglycerides. © 2013 Pearson Education, Inc. 23.2 Fatty Acids and Their Esters Triacylglycerols • The fat or oil from a given natural source is a complex mixture of many different triacylglycerols. © 2013 Pearson Education, Inc. 23.2 Fatty Acids and Their Esters Triacylglycerols • Vegetable oils consist of mainly unsaturated fatty acids. • Animal fats contain a large percentage of saturated fatty acids. • This difference in composition is the primary reason for the different melting points of fats and oils. © 2013 Pearson Education, Inc. 23.2 Fatty Acids and Their Esters • • • • Lipids in the Diet Fats and oils are a popular component of our diet; excess energy from dietary fats and oils is mostly stored as fat in adipose tissue. Concern for the relationships among saturated fats, cholesterol levels, and various diseases caused a decrease of the average calories from fats and oils in the U.S. diet. Several organizations recommend a diet with not more than 30% of its calories from fats and oils. The FDA recommends that not more than 10% of daily calories come from saturated fat and not more than 300 mg of cholesterol be included in the daily diet. © 2013 Pearson Education, Inc. 23.3 Properties of Fats and Oils • The more double bonds a fatty acid has, the lower its melting point. • The difference in melting points between fats and oils is a consequence of this difference. • Vegetable oils are lower melting because oils generally have a higher proportion of unsaturated fatty acids than animal fats. © 2013 Pearson Education, Inc. 23.3 Properties of Fats and Oils • The hydrocarbon chains in saturated acids are uniform in shape with identical angles at each carbon atom, and flexible chains, allowing them to nestle together. • Unsaturated acids have rigid kinks wherever they contain cis double bonds. The kinks make it difficult for such chains to fit next to each. The more double bonds there are in a triacylglycerol, the harder it is for it to solidify. © 2013 Pearson Education, Inc. 23.3 Properties of Fats and Oils © 2013 Pearson Education, Inc. 23.3 Properties of Fats and Oils • Triacylglycerols are uncharged, nonpolar, hydrophobic molecules that coalesce when stored in fatty tissue. • The primary function of triacylglycerols is long-term storage of energy. • Adipose tissue serves to provide thermal insulation and protective padding. • Color and flavor are contributed by natural materials. • Oxidation causes rancidity, decomposition to products with unpleasant odors or flavors. © 2013 Pearson Education, Inc. 23.3 Properties of Fats and Oils Properties of the Triacylglycerols in Natural Fats and Oils • Nonpolar and hydrophobic • No ionic charges • Solid triacylglycerols (fats)—high proportion of saturated fatty acid chains • Liquid triacylglycerols (oils)—high proportion of unsaturated fatty acid chains © 2013 Pearson Education, Inc. 23.4 Chemical Reactions of Triacylglycerols Hydrogenation • The carbon–carbon double bonds in vegetable oils can be hydrogenated to yield saturated fats. • Margarine and solid cooking fats are produced commercially by hydrogenation of vegetable oils. • By controlling the extent of hydrogenation and monitoring the composition of the product, consistency can be controlled. © 2013 Pearson Education, Inc. 23.4 Chemical Reactions of Triacylglycerols © 2013 Pearson Education, Inc. 23.4 Chemical Reactions of Triacylglycerols Hydrolysis of Triacylglycerols • Triacylglycerols can be hydrolyzed. In the body, this hydrolysis is catalyzed by enzymes (hydrolases) and is the first reaction in the digestion of dietary fats and oils. • Soap-making involves the base-catalyzed hydrolysis of triacylglycerols. Any mixture of triacylglycerols can be used; the second ingredient needed is lye or potash. © 2013 Pearson Education, Inc. 23.4 Chemical Reactions of Triacylglycerols • • • • • • • Detergents Detergent is a term usually applied to synthetic materials made from petroleum chemicals. Like soaps, synthetic detergent molecules have hydrophobic hydrocarbon tails and hydrophilic heads, and cleanse by forming micelles around greasy dirt. All substances that function in this manner are described as surfactants. The hydrophilic heads may be anionic, cationic, or non-ionic. Soaps work as cleaning agents because the two ends of a soap molecule are so different. The ionic end is hydrophilic (water-loving); it tends to dissolve in water. The long hydrocarbon chain portion of the molecule, however, is nonpolar and therefore hydrophobic (water-fearing). Because of these opposing tendencies, soap molecules are attracted to both grease and water. When soap is dispersed in water, the big organic anions cluster together so that their long, hydrophobic hydrocarbon tails are in contact. By doing so, they avoid disrupting the strong hydrogen bonds of water and create a nonpolar microenvironment. Hydrophilic ionic heads on the surface of the cluster stick out into the water. The resulting spherical clusters are called micelles. Grease and dirt become coated by the nonpolar tails of the soap molecules and trapped in the center of the micelles as they form. Once suspended within micelles, the grease and dirt can be rinsed away. © 2013 Pearson Education, Inc. 23.5 Phospholipids and Glycolipids • Cell membranes separate the aqueous interior of cells from the aqueous environment surrounding the cells. • The three major kinds of cell membrane lipids in animals are phospholipids, glycolipids, and cholesterol. © 2013 Pearson Education, Inc. 23.5 Phospholipids and Glycolipids • Phospholipids are built up from glycerol (to give glycerophospholipids) or sphingosine (to give sphingomyelins). • Glycolipids are also derived from sphingosine, but have an attached carbohydrate that is a monosaccharide or a short chain of monosaccharides. • Cholesterol is a sterol, a class of biomolecules that are characterized by a system of four fused rings. © 2013 Pearson Education, Inc. 23.5 Phospholipids and Glycolipids © 2013 Pearson Education, Inc. 23.5 Phospholipids and Glycolipids Phospholipids • Similar to soap and detergent molecules in having ionic, hydrophilic heads and hydrophobic tails. They differ, however, in having two tails instead of one. Glycerophospholipids or phosphoglycerides • Triesters of glycerol 3-phosphate, the most abundant membrane lipids. • Two of the ester bonds are with fatty acids. • The third position is a phosphate ester linked to ethanolamine, choline, or serine. © 2013 Pearson Education, Inc. 23.5 Phospholipids and Glycolipids © 2013 Pearson Education, Inc. 23.5 Phospholipids and Glycolipids • Glycerophospholipids are named as derivatives of phosphatidic acids. • Lipids with a phosphate ester to choline are known as phosphatidylcholines, or lecithins. • Hydrophobic tails and hydrophilic head groups make glycerophospholipids emulsifying agents. © 2013 Pearson Education, Inc. 23.5 Phospholipids and Glycolipids • In sphingolipids, the amino alcohol sphingosine provides one of the hydrophobic tails. • The second tail is from a fatty acid acyl group connected by an amide link to the —NH2 group in sphingosine. • Sphingomyelins are sphingosine derivatives with a phosphate ester group at C1 of sphingosine. The sphingomyelins are major components of the coating around nerve fibers. © 2013 Pearson Education, Inc. 23.5 Phospholipids and Glycolipids Glycolipids • Glycolipids are also derived from sphingosine. They have a carbohydrate group at C1 instead of a phosphate bonded to an amino alcohol. • Glycolipids reside in cell membranes with their carbohydrate segments extending into the fluid surrounding the cells. © 2013 Pearson Education, Inc. 23.5 Phospholipids and Glycolipids • Cerebrosides contain a single monosaccharide. • They are abundant in nerve cell membranes in the brain with the monosaccharide D-galactose. • In other membranes, the sugar unit is D-glucose. • Gangliosides are glycolipids in which the carbohydrate is a small polysaccharide rather than a monosaccharide. • Over 60 different gangliosides are known. © 2013 Pearson Education, Inc. 23.6 Sterols • Sterols are lipids whose structure is based on a tetracyclic (four-ring) carbon skeleton. © 2013 Pearson Education, Inc. 23.6 Sterols Cholesterol • Cholesterol is the most abundant animal sterol. • It is a component of cell membranes and the starting material for all other sterols. • Within a cell membrane, nearly-flat cholesterol molecules are distributed among the tails of phospholipids. • Because they are more rigid than the hydrophobic tails, the cholesterol molecules help to maintain the structural rigidity of the membrane. • Approximately 25% of liver cell membrane is cholesterol. © 2013 Pearson Education, Inc. 23.6 Sterols • Bile acids are essential for the emulsification of fats during digestion. • Synthesized in liver cells from cholesterol and stored in the gall bladder, these molecules have a polar end and a nonpolar end. • Solubility of bile acids is increased by conjugation with either taurine, a cysteine derivative, or glycine, which increases solubility and enhances the formation of micelles. • Micelles are essential for the digestion of dietary fat. • The two most common bile acids are cholic acid and chenodeoxycholic acid. In the intestinal tract these acids are ionized to anions and referred to as bile salts. © 2013 Pearson Education, Inc. 23.6 Sterols • Steroid Hormones are divided into three types. • Mineralocorticoids, such as aldosterone, regulate the cellular fluid balance between Na+ and K+ ions. • Glucocorticoids help to regulate glucose metabolism and inflammation. Anti-inflammatory ointments contain hydrocortisone to reduce swelling and itching. • Sex hormones—The two most important androgens, are testosterone and androsterone. Estrone and estradiol, the estrogens, are synthesized from testosterone. © 2013 Pearson Education, Inc. 23.6 Sterols Butter and Its Substitutes • It has become medically accepted that butter can contribute to elevated blood cholesterol, which is to be avoided because of cholesterol’s role in heart disease. • Margarine, which is made from vegetable oils, contains no cholesterol and much less saturated fat than butter. • Margarine contains what might be an even more unhealthful ingredient—trans fatty acids. Oils are catalytically hydrogenated to give them a firmer consistency. During the partial hydrogenation, some of the cis double bonds are converted to trans double bonds. • Numerous studies have linked the quantity of trans fatty acids in a person’s diet to a greater risk for heart disease and cancer. • Meat and dairy products contain a very small amount of trans fatty acids (about 0.2% in butter), but the quantities in foods containing hydrogenated oils are much higher—up to 40% in the stiffer margarines. © 2013 Pearson Education, Inc. 23.7 Structure of Cell Membranes • Phospholipids provide the basic structure of cell membranes, where they aggregate in a closed, sheet-like, double leaflet structure—the lipid bilayer. © 2013 Pearson Education, Inc. 23.7 Structure of Cell Membranes • The bilayer is highly ordered and stable, but still flexible. • When phospholipids are shaken vigorously with water, they spontaneously form liposomes—small spherical vesicles with a lipid bilayer surrounding an aqueous center. • Water-soluble substances can be trapped in the center of liposomes, and lipid-soluble substances can be incorporated into the bilayer. • Liposomes are potentially useful as carriers for drug delivery because they can fuse with cell membranes and empty their contents into the cell. © 2013 Pearson Education, Inc. 23.7 Structure of Cell Membranes © 2013 Pearson Education, Inc. 23.7 Structure of Cell Membranes • The overall structure of cell membranes is represented by the fluid-mosaic model. The membrane is fluid because molecules can move around within it, and as a mosaic because it contains many kinds of molecules. • 20% or more of a membrane consists of protein molecules. • Peripheral proteins are associated with just one face of the bilayer and are held by noncovalent interactions with the hydrophobic lipid tails or the hydrophilic head groups. • Integral proteins extend completely through the membrane and are anchored by hydrophobic regions that extend through the bilayer. • The carbohydrate parts of glycoproteins and glycolipids mediate the interactions of the cell with outside agents. © 2013 Pearson Education, Inc. 23.7 Structure of Cell Membranes • Because the bilayer membrane is fluid rather than rigid, it is not easily ruptured. • Proteins move sideways in the membrane layers continuously, not unlike floating on a pond; this is an energetically neutral motion. • Membrane components do not flip from the inside of the membrane to the outside. • Small nonpolar molecules can easily enter the cell through the membrane and individual lipid or protein molecules can diffuse rapidly from place to place. © 2013 Pearson Education, Inc. 23.8 Transport Across Cell Membranes • There are two modes of passage across the membrane. • Passive transport, in which substances move across the membrane freely by diffusion from regions of higher concentration to regions of lower concentration. • Active transport, in which substances cross the membrane only when energy is supplied because they go from lower to higher concentration regions. © 2013 Pearson Education, Inc. 23.8 Transport Across Cell Membranes © 2013 Pearson Education, Inc. 23.8 Transport Across Cell Membranes • Some solutes enter and leave cells by simple diffusion. Small, nonpolar molecules and lipidsoluble substances, including steroid hormones, move through the hydrophobic lipid bilayer in this way. • In facilitated diffusion, solutes are helped across the membrane by proteins. The molecule binds to a membrane protein, which changes shape so that the transported molecule is released on the other side of the membrane. © 2013 Pearson Education, Inc. 23.8 Transport Across Cell Membranes • Maintaining concentration gradients requires an expenditure of energy. • Energy from the conversion of ATP to ADP is used to change the shape of the sodium/potassium pump, simultaneously bringing two K+ ions into the cell and moving three Na+ ions out of the cell. © 2013 Pearson Education, Inc. 23.8 Transport Across Cell Membranes Properties of cell membranes • Cell membranes are composed of a fluid-like phospholipid bilayer. • The bilayer incorporates cholesterol, proteins (including glycoproteins), and glycolipids. • Small nonpolar molecules cross by simple diffusion through the lipid bilayer. • Small ions and polar molecules diffuse across the membrane via protein pores (simple diffusion). • Glucose and certain other substances (including amino acids) cross with the aid of proteins and without energy input (facilitated diffusion). • Na+, K+, and other substances that maintain concentration gradients across the cell membrane cross with expenditure of energy and the aid of proteins (active transport). © 2013 Pearson Education, Inc. 23.9 Eicosanoids: Prostaglandins and Leukotrienes • Eicosanoids are a group of compounds derived from 20-carbon unsaturated fatty acids (eicosanoic acids) and synthesized throughout the body. They function as short-lived chemical messengers that act near their points of synthesis (“local hormones”). • Prostaglandins and leukotrienes are differ somewhat in their structure; prostaglandins contain a five-membered ring, which the leukotrienes lack. © 2013 Pearson Education, Inc. 23.9 Eicosanoids: Prostaglandins and Leukotrienes • Prostaglandins and leukotrienes are synthesized from arachidonic acid, which is itself synthesized from linolenic acid. © 2013 Pearson Education, Inc. 23.9 Eicosanoids: Prostaglandins and Leukotrienes • Prostaglandins can lower blood pressure, influence platelet aggregation during blood clotting, stimulate uterine contractions, and lower the extent of gastric secretions. In addition, they are responsible for some of the pain and swelling that accompany inflammation. • The anti-inflammatory and fever-reducing (antipyretic) action of aspirin results in part from its inhibition of prostaglandin synthesis. • Leukotriene release has been found to trigger the asthmatic response, severe allergic reactions, and inflammation. © 2013 Pearson Education, Inc. Chapter Summary 1. What are the major classes of fatty acids and lipids? • Fatty acids are carboxylic acids with long, straight (unbranched) hydrocarbon chains; they may be saturated or unsaturated. • Waxes are esters of fatty acids and alcohols with long, straight hydrocarbon chains. • Fats and oils are triacylglycerols—triesters of glycerol with fatty acids. In fats, the fatty acid chains are mostly saturated; in oils, the proportions of unsaturated fatty acid chains vary. Fats are solid because the saturated hydrocarbon chains pack together neatly; oils are liquids because the kinks at the cis double bonds prevent such packing. © 2013 Pearson Education, Inc. Chapter Summary, Continued 2. What reactions do triacylglycerols undergo? • The principal reactions of triacylglycerols are catalytic hydrogenation and hydrolysis. • Hydrogen adds to the double bonds of unsaturated hydrocarbon chains in oils, thereby thickening the consistency of the oils and raising their melting points. • Treatment of a fat or oil with a strong base such as NaOH hydrolyzes the triacylglycerols to give glycerol and salts of fatty acids. Such saponification reactions produce soap, a mixture of fatty acid salts. © 2013 Pearson Education, Inc. Chapter Summary, Continued 3. What are sterols? • The unifying feature of sterols is a fused four-ring system. • Sterols include cholesterol, an important participant in membrane structure. • Bile acids and salts, necessary for the emulsification of fats during digestion, are synthesized from cholesterol. • The third major group of sterols includes steroid hormones, including the sex hormones, which function as signaling molecules. © 2013 Pearson Education, Inc. Chapter Summary, Continued 4. What are the membrane lipids? • The membrane lipids include phospholipids and glycolipids (which have hydrophilic, polar head groups and a pair of hydrophobic tails) and cholesterol (a steroid). • Phospholipids, which are either glycerophospholipids (derived from glycerol) or sphingomyelins (derived from the amino alcohol sphingosine), have charged phosphate diester groups in their hydrophilic heads. • Sphingolipids, which are either sphingomyelins or glycolipids, are sphingosine derivatives. The glycolipids have carbohydrate head groups. © 2013 Pearson Education, Inc. Chapter Summary, Continued 5. What is the nature of a cell membrane? • The basic structure of cell membranes is a bilayer of lipids, with their hydrophilic heads in the aqueous environment outside and inside the cells, and their hydrophobic tails clustered together in the center of the bilayer. • Cholesterol molecules fit between the hydrophobic tails and help maintain membrane structure and rigidity. • The membrane also contains glycoproteins and glycolipids (with their carbohydrate segments at the cell surface, where they serve as receptors), as well as proteins. • Some of the proteins extend through the membrane (integral proteins), and others are only partially embedded at one surface. © 2013 Pearson Education, Inc. Chapter Summary, Continued 6. How do substances cross cell membranes? • Small molecules and lipid-soluble substances can cross the lipid bilayer by simply diffusing through it. Ions and hydrophilic substances can move through aqueous fluidfilled channels in membrane proteins. Some substances cross the membrane by binding to an integral protein, which then releases them inside the cell. These modes of crossing are all passive transport—they do not require energy because the substances move from regions of higher concentration to regions of lower concentration. Passive transport takes the form of simple diffusion, crossing the membrane by passing through it unimpeded, or facilitated diffusion, crossing the membrane with the aid of a protein embedded in the membrane. Active transport, which requires energy and is carried out by certain integral membrane proteins, moves substances against their concentration gradients. • • • • • © 2013 Pearson Education, Inc. Chapter Summary, Continued 7. What are eicosanoids? • The eicosanoids are a group of compounds derived from 20-carbon unsaturated fatty acids. • They are local hormones—that is, they act near their point of origin and are short-lived. • Prostaglandins, which contain a five-membered ring, have a wide range of actions (such as stimulating uterine contractions and causing inflammation). • Leukotrienes, which do not contain a fivemembered ring, trigger the asthmatic response, severe allergic reactions, and inflammation. © 2013 Pearson Education, Inc.