* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Document
Pharmacogenomics wikipedia , lookup
Discovery and development of neuraminidase inhibitors wikipedia , lookup
Drug discovery wikipedia , lookup
CCR5 receptor antagonist wikipedia , lookup
Prescription costs wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Neuropharmacology wikipedia , lookup
Pharmacokinetics wikipedia , lookup
Drug interaction wikipedia , lookup
Theralizumab wikipedia , lookup
Discovery and development of non-nucleoside reverse-transcriptase inhibitors wikipedia , lookup
Discovery and development of HIV-protease inhibitors wikipedia , lookup
HIV vaccine wikipedia , lookup
Discovery and development of integrase inhibitors wikipedia , lookup
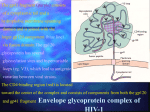
Update on HIV Therapy Hail M. Al-Abdely, MD Consultant, Infectious Diseases Clinical, Virological and Immunological Course of HIV Infection Symptoms Virus in Plasma Infection Death Detectable Time 0 VIRUS IN PLASMA Detectable 12 Years Clinical, Virological and Immunological Course of HIV Infection CD4 Cell Count Symptoms Virus in Plasma Infection Time 0 Death Detectable VIRUS IN PLASMA Detectable > 500 cells CD4 COUNTS < 200 cells 12 Years Clinical, Virological and Immunological Course of HIV Infection CD4 Cell Count Symptoms Virus in Plasma Infection Death Detectable VIRUS IN PLASMA Detectable > 500 cells CD4 COUNTS < 200 cells Time 0 12 Years Seroconversion Asymptomatic AIDS Clinical, Virological and Immunological Course of HIV Infection 1000 CD4 Cell Count 800 600 RNA in Plasma 400 Symptoms Virus in Plasma 200 Infection0 Death Detectable VIRUS IN PLASMA Detectable > 500 cells CD4 COUNTS < 200 cells Time 0 12 Years Seroconversion Asymptomatic AIDS Development of AIDS is like an impending train wreck Viral Load = Speed of the train CD4 count = Distance from cliff HIV infection J. Coffin, XI International Conf. on AIDS, Vancouver, 1996 Viral Dynamics of HIV-1 Infection Latently infected CD4 lymphocytes Productively infected CD4 lymphocytes <1% T 1/2 ~1.6d 99% Uninfected CD4 lymphocytes 2.6 days per generation T1/2 ~5.7 hrs HIV <1% Uninfected activated CD4 lymphocytes Long-lived cell populations Perelson et.al. Science 271:1582 (1996) CD4 lymphocytes infected with defective virus Viral dynamics It takes 2.6 days to produce a new generation of viral particles Estimated total HIV production is 10.3 x 109 virions per day 99% of the virus pool is produced by recently infected cells Retroviral therapy should be able to reduce viral load within a few days GOALS OF THERAPY • Clinical goals: Prolongation of life and improved quality of life • Virologic goals: Reduction in viral load as much as possible for as long as possible to: 1) halt disease progression, and 2) prevent/reduce resistant variants • Immunologic goals: Achieve immune reconstitution that is quantitative (CD4 to normal range) and qualitative (pathogen-specific immune response) • Therapeutic goals: Rational sequencing of drugs in a fashion that achieves virologic goals, but also: 1) maintains therapeutic options; 2) is relatively free of side effects; and 3) is realistic in terms of probability of adherence • Epidemiologic goals: Reduce HIV transmission 19 1 Antiretroviral Drugs Approved by FDA for HIV Generic Name Class FDA Approval Date Zidovudine, AZT NRTI March 87 Didanosine, ddI NRTI October 91 Zalcitabine, ddC NRTI June 92 Stavudine, d4T NRTI June 94 Lamivudine, 3TC NRTI November 95 Saquinavir, SQV, hgc PI December 95 Ritonavir, RTV PI March 96 Indinavir, IDV PI March 96 Nevirapine, NVP NNRTI June 96 Nelfinavir, NFV PI March 97 Delavirdine, DLV NNRTI April 97 Combivir (AZT+3TC) NRTI September 97 Saquinavir, SQV, sgc PI November 97 Efavirenz, EFV NNRTI September 98 Abacavir, ABC NRTI February 99 Amprenavir (AMP) PI April 99 Lopinavir (LPV) PI September 00 EC Didanosine(EC DDI) NRTI September 00 Trizivir (AZT+3TC+ABC) NRTI September 00 Current antiretroviral targets Viral protease RNA Reverse transcriptase RNA Proteins RT RNA ZDV, ddI, ddC, d4T, 3TC, ABC, RNA DNA RT DNA DLV, NVP, EFV DNA Provirus SQV RTV IDV NFV APV LPV 90 100 80 80 80 70 70 60 60 60 40 50 20 50 Monot herapy Monotherapy Monot herapy 90 Dual therapy Monot herapy Viral Suppression with Monotherapy versus Multiple Drugs Triple therapy Deaths per 100,000 Population Trends in Age-Adjusted* Rates of Death due to HIV Infection, USA, 1982-1998 18 16 14 12 10 8 6 4 2 0 82 84 86 88 *Using the age distribution of the projected year 2000 US population as the standard. 90 Year 92 94 96 **Preliminary 1998 data 98** Good News Highly active antiretroviral therapy has Changed our view toward HIV from inevitably fatal to a manageable disease over several decades Bad News 1. 2. 3. 4. 5. Incomplete response Complexity of treatment Short and long term side effects Resistance Drug-drug interactions Bad News 1. Incomplete response • • 2. 3. 4. 5. Complete RNA suppression and sustained CD4 increase happens only in 60-80%. Effectiveness is even lower in patients with high replication rates and extensive antiretroviral experience. Complexity of treatment Short and long term side effects Resistance Drug-drug interactions Viral Suppression with Monotherapy versus Multiple Drugs 90 90 80 80 70 70 60 60 Monot herapy Monotherapy 50 Monot herapy 50 Dual therapy Monot herapy Triple therapy Virologic nadir predicts duration of response Bad News 1. Incomplete response 2. Complexity of treatment • • • Too many tablets. Difficult schedule. Food factor 3. Short and long term side effects 4. Resistance 5. Drug-drug interactions Bad News 1. Incomplete response 2. Complexity of treatment 3. Short and long term side effects 4. Drug-drug interactions 5. Resistance Side Effects of NRTIs Drug Common Side effects Zidovudine (azt, zdv) Initial nausea, headache, fatigue, anemia, neutropenia, neuropathy, myopathy. Lamivudine (3TC) GI side effects. Didanosine (ddl) GI side effects. Peripheral neuropathy in 15%, pancreatitis. Zalcitabine (ddC) Peripheral neuropathy in 17-31% of trial participants; oral ulcers. Stavudine (d4T) Peripheral neuropathy (1-4% in early studies; 24% in expanded access patients with CD4+ counts < 50) Abacavir (ABC) About 3%-5% hypersensitivity reaction: malaise, fever, possible rash, GI. Resolves within 2 days after discontinuation. Side Effects of NNRTIs Drug Common Side effects Delavirdine Transient rash. P450 3A4 inhibitor Nevirapine Transient rash, hepatitis. P450 3A4 inducer. Efavirenz Initial dizziness, insomnia, transient rash, P450 3A4 inducer. Side Effects of PIs Drug Common Side effects Amprenavir Rash (20%), diarrhea, nausea Indinavir Kidney stones in 6 to 8%: good hydration essential. Occasional nausea and GI upset. Nelfinavir Diarrhea common; occasional nausea Ritonavir Nausea, diarrhea, numb lips for up to 5 weeks; occasional hepatitis. Saquinavir Nausea, diarrhea. Metabolic Complications of PIs • Hyperbilirubinemia • Hyperlipidemia – Coronary artery disease • Insulin resistance • Abnormal fat distribution. • Lipodystrophy Bad News 1. Incomplete response 2. Complexity of treatment 3. Short and long term side effects 4. Drug-drug interactions 5. Resistance Drugs That Should Not Be Used With Antiretrovirals Drug Category Indinavir Ritonavir* Saquinavir Nelfinavir Amprenavir Nevirapine Delavirdine Efavirenz Ca++ channel blocker (none) bepridil (none) (none) bepridil (none) (none) (none) Cardiac (none) amioderone flecainide propafenone quinidine (none) (none) (none) (none) (none) (none) Lipid Lowering Agents simvastatin lovastatin simvastatin lovastatin simvastatin lovastatin simvastatin lovastatin simvastatin lovastatin (none) simvastatin lovastatin (none) AntiMycobacterial rifampin none rifampin rifabutin rifampin rifampin (none) rifampin rifabutin (none) Antihistamine astemizole terfenadine astemizole terfenadine astemizole terfenadine astemizole terfenadine astemizole terfenadine (none) astemizole terfenadine astemizole terfenadine cisapride cisapride cisapride cisapride cisapride (none) cisapride H-2 blockers Proton pump inhibitors cisapride Neuroleptic (none) clozapine pimozide (none) (none) (none) (none) (none) (none) Psychotropic midazolam triazolam midazolam triazolam midazolam triazolam midazolam triazolam midazolam triazolam (none) midazolam triazolam midazolam triazolam Gastrointestinal Drugs Bad News 1. Incomplete response 2. Complexity of treatment 3. Short and long term side effects 4. Drug-drug interactions 5. Resistance Resistance Genotypic Mutations Associated With Resistance to NRTI & NNRTIs Agent ZDV 41 67 69* 3TC 69* ddI 65 ddC 65 d4T 70 Resistance mutations 151 69 50 ABC 151 184 69* 74 151 184 69* 74 151 184 69* 65 210 75 69* 74 Agent 151 115 215 219 333 333 178 151 184 Resistance mutations DLV 103 EFV 100 103 NV 100 103 181 108 106 108 179 236 181 188 190 181 188 190 225 Resistance Genotypic Mutations Associated With Resistance to PIs Agent APV 10 IDV 10 NFV 10 RTV 10 20 SQV 10 20 LPV 10 20 24 32 30 32 24 30 32 33 36 Resistance mutations 46 47 48 50 54 63 71 36 46 48 63 71 36 46 48 36 46 36 46 46 54 48 47 50 82 84 82 84 71 82 84 82 84 90 82 84 90 54 63 71 54 63 71 73 73 84 90 88 90 Overcoming Drug Resistance Increase exposure to drug RESISTANCE Change to a drug to which virus shows greater susceptibility Drug Overcoming Drug Resistance Change to a drug to which virus shows greater susceptibility Guided by Genotypic resistance testing Switching within a drug class Example - Switching within PI class of drugs: • Primary mutation associated with reduced susceptibility to nelfinavir is D30N • Timely switching of patients on a failing NFV regimen harboring D30N has resulted in good clinical response • New PI regimen has increased susceptibility due to non-cross resistance to D30N Switching to a different drug class • Often switching within a drug class not effective due to class cross-resistance – NRTI: Q151M, 69 insertion (other multiple MU) – NNRTI: K103N (others) – PI: G48V + V82A (other multiple primary) • Switching to new class of drugs not previously used most effective Percentage of patients with plasma HIV-RNA below 200 copies/ml in the VIRADAPT study % <200 copies/ml Randomized Study 35 30 25 Control Genotypic 20 15 10 5 0 0 3 6 9 Months (Adapted from Clevenbergh et al. Antiviral Therapy 2000; 5:65–70) 12 Percentage of patients with plasma HIV-RNA below 200 copies/ml in the VIRADAPT study % <200 copies/ml Open Study Randomized Study 35 30 25 Control Genotypic 20 15 10 5 0 0 3 6 9 Months (Adapted from Clevenbergh et al. Antiviral Therapy 2000; 5:65–70) 12 Overcoming Drug Resistance Increase exposure to drug RESISTANCE Change to a drug to which virus shows greater susceptibility Drug Saquinavir boosted by ritonavir SQV conc (ng/mL) 9000 8000 7000 Fortovase 1600 mg + ritonavir 100 mg qd 6000 5000 4000 Fortovase 1200 mg tid 3000 2000 1000 0 0 5 10 15 20 25 Time (hours) Kilby et al. Antimicrob Agents Chemother Vol 44 2000 Increase - above efficacy, below toxicity Drug conc (ng/mL) 9000 8000 7000 6000 5000 4000 3000 2000 Drug A 1000 Drug A level required to overcome WT virus 0 0 5 10 Time (hours) 15 20 25 Increase - above efficacy, below toxicity Drug conc (ng/mL) 9000 8000 7000 Boosted Drug A 6000 5000 Drug A level required to overcome “resistant” virus 4000 3000 2000 Drug A 1000 Drug A level required to overcome WT virus 0 0 5 10 Time (hours) 15 20 25 Increase - above efficacy, below toxicity Drug conc (ng/mL) Drug A Toxicity threshold 9000 8000 7000 Boosted Drug A 6000 5000 Drug A level required to overcome “resistant” virus 4000 3000 2000 Drug A 1000 Drug A level required to overcome WT virus 0 0 5 10 Time (hours) 15 20 25 The benefits of therapeutic drug monitoring HIV RNA* 0.05 -0.15 Control sub-optimal concentration -0.35 -0.55 -0.75 -0.95 -1.15 -1.35 -1.55 0 * viral load from baseline, log10 copies/ml 3 6 Months (Adapted from Garaffo et al. Antiviral Therapy 1999; 4 (Suppl 1):75– 76) The benefits of therapeutic drug monitoring HIV RNA* 0.05 -0.15 Control sub-optimal concentration -0.35 -0.55 Genotypic sub-optimal concentration -0.75 -0.95 -1.15 -1.35 -1.55 0 * viral load from baseline, log10 copies/ml 3 6 Months (Adapted from Garaffo et al. Antiviral Therapy 1999; 4 (Suppl 1):75– 76) The benefits of therapeutic drug monitoring HIV RNA* 0.05 -0.15 Control sub-optimal concentration -0.35 -0.55 Genotypic sub-optimal concentration -0.75 Control optimal concentration -0.95 -1.15 -1.35 -1.55 0 * viral load from baseline, log10 copies/ml 3 6 Months (Adapted from Garaffo et al. Antiviral Therapy 1999; 4 (Suppl 1):75– 76) The benefits of therapeutic drug monitoring HIV RNA* 0.05 -0.15 Control sub-optimal concentration -0.35 -0.55 Genotypic sub-optimal concentration -0.75 Control optimal concentration -0.95 -1.15 -1.35 Genotypic optimal concentration -1.55 0 * viral load from baseline, log10 copies/ml 3 6 Months (Adapted from Garaffo et al. Antiviral Therapy 1999; 4 (Suppl 1):75– 76) Indications for the Initiation of Antiretroviral Therapy in the Chronically HIV-Infected Patient Clinical Category CD4+ T-Cell Count and HIV RNA Recommendation Symptomatic (AIDS, thrush, unexplained fever) Any value Treat Asymptomatic CD4+ T Cells < 350 cells/mm3 or HIV RNA > 10,000 (bDNA) or > 20,000 (RT-PCR) copies/mL Treatment should be offered. Strength of recommendation is based on prognosis for disease-free survival and willingness of the patient to accept therapy. Asymptomatic CD4+ T Cells > 350 cells/mm3 and HIV RNA < 10,000 (bDNA) or < 20,000 (RT-PCR) copies/mL Many experts would delay therapy and observe; however, some experts would treat. Recommended Antiretroviral Agents for Treatment of Established HIV Infection Column A Column B Efavirenz Indinavir Nelfinavir Ritonavir + Saquinavir (SGC or HGC*) Stavudine + Lamivudine Stavudine + Didanosine Zidovudine + Lamivudine Zidovudine + Didanosine Recommended as an alternative Abacavir Amprenavir Delavirdine Nelfinavir + Saquinavir-SGC Nevirapine Ritonavir Saquinavir-SGC Didanosine + Lamivudine Zidovudine + Zalcitabine No recommendation; insufficient data Hydroxyurea in combination with other antiretroviral drugs Ritonavir + Indinavir Ritonavir + Nelfinavir Strongly recommended Not recommended; should not be offered (All monotherapies, whether from column A or B§) Saquinavir-HGC Stavudine + Zidovudine Zalcitabine + Lamivudine Zalcitabine + Stavudine Zalcitabine + Didanosine New agents in the pipeline New agents should: 1. 2. 3. 4. Exhibit high potency. Adequate drug levels. Activity against resistant isolates. Penetration into all cellular and bodily compartments (eg, central nervous system, genital tract). 5. Favorable drug interaction profile. 6. Minimal side effects. 7. Convenient to take, with no food restrictions and minimal dosing requirements; preferably once daily. Potential new targets Binding, fusion and entry Viral zinc-finger nucleocapsid proteins Viral protease RNA RNA Proteins Reverse transcriptase RT RNA RNA DNA RT Viral regulatory proteins DNA DNA Viral integrase Provirus HIV viral membrane fusion Stein et al. (1987) Cell 49: 664 HIV interaction with CD4 cell CXCR4 CCR5 CD4 Cell HIV interaction with CD4 cell gp41 gp120 CD4 Attachment CXCR4 CCR5 CD4 Cell HIV interaction with CD4 cell Co-receptor Interaction gp41 gp120 CD4 Attachment CXCR4 CCR5 CD4 Cell HIV HIV interaction with CD4 cell Co-receptor Interaction HIV gp41 Anchorage gp120 HIV CD4 Attachment CXCR4 CCR5 CD4 gp41 Cell HIV interaction with CD4 cell Co-receptor Interaction HIV gp41 Anchorage gp120 HIV CD4 Attachment CXCR4 CCR5 CD4 gp41 Cell HIV HR1-HR2 Interaction HIV interaction with CD4 cell Co-receptor Interaction HIV gp41 Anchorage gp120 HIV CD4 Attachment CXCR4 CCR5 CD4 gp41 Cell Fusion Complete HIV HR1-HR2 Interaction Entry inhibitors under development Class Attachment Inhibitors Co-receptor Inhibitors Fusion Inhibitors Target gp120, CD4 CXCR-4 Example Compounds specific Mab, soluble CD4 and CD4-Ig AMD-3100 CCR-5 SCH-C, specific Mab, gp41 T-20, T-1249, D-peptides HIV attachment inhibitors PRO 542 - Novel protein – Human IgG-2 Fv replaced with HIV binding domains of CD4 molecule – Neutralized broad range of HIV variants in vitro – Active in SCID-Hu model with primary isolates – Phase II clinical testing HIV attachment inhibitors • PRO 542 (rCD4-IgG2) – Single injection dose-ranging trial ~ 4 doses, 3-6 subject/dose, HIV RNA > 3,000 CD4 > 50 ~ Well tolerated, single dose non-immunogenic, linear pharmacokinetics ~ 6/6 high dose subjects had decrease in HIV RNA, infectious titers of virus declined Chemokine receptor inhibitors • CCR-5 Inhibitors – SCH-C (Schering-Plough) – PRO 140 (anti-CCR-5 monoclonal antibody) • CXCR-4 Inhibitors – AMD-3100 CCR-5 inhibitors: SCH-C • Small molecule antagonist of CCR-5 • PK profile in animals supports oral administration • Active in SCID-hu Thy/Liv model against primary HIV • Risk of switch to SI (CXCR-4) virus? CXCR-4 inhibitors: AMD 3100 • Targets CXCR-4 and dual tropic virus • Resistance develops in vitro • Active SCID-hu mouse (CXCR-4, dual tropic HIV) • IV and SC administration well tolerated • CXCR-4: importance in embryogenesis and immune function? T-20 (Fusion inhibitor) 41 patients, monitored for 48 weeks after adding T-20 to failing therapy, and a mean HIV RNA decline of -1.4 log10 copies/mL has been reported New agents - NRTI New agents: 1. Emtricitabine (FTC, Coviracil) 2. DAPD/DXG 3. Emivirine (MKC-442, Coactinon). New formulations: 1. Enteric-coated didanosine (Videx EC). 400 mg once daily. 2. Extended-release formulation of stavudine. 3. Zidovudine +lamivudine + abacavir single tablet (Trizivir) New agents - NRTI Emtricitabine: • • • Fluorinated cytosine analogue with a similar resistance profile to lamivudine, but 4- to 10-fold more active in vitro. Administered once daily. Phase II study: given with didanosine and efavirenz once daily • • 93% of patients had HIV RNA below 50 copies/mL at week 24, and 48-week lone virologic failure in this study had rebounded from below 50 copies/mL to below 400 copies/mL. New agents - NRTI DAPD: • • • • Guanosine analogue, which is metabolized to the active form, DXG. A 15-day monotherapy dose-ranging study in antiretroviral-naive patients demonstrated HIV RNA declines of 0.5-1.6 log10 copies/mL, and 0.5-1.1 log10 copies/mL in antiretroviral-experienced patients. No adverse events were reported during these studies. DAPD is likely to be active against HIV carrying the Q151M mutation, which confers cross-class resistance. New agents– Protease Inhibitors 1. 2. 3. 4. 5. Lopinavir/ritonavir (ABT-378/r, Kaletra) – approved by FDA. BMS-232632. Tipranavir. DMP-450. PD 178390 New agents– Protease Inhibitors Lopinavir/ritonavir (ABT-378/r, Kaletra): • uses a low dose of ritonavir to achieve very high plasma levels of lopinavir, enabling it to retain activity against virus with low-to-moderate levels of resistance to PIs (including to lopinavir itself). • 96% of patients with 0 to 5 PI mutations achieved HIV RNA less than 400 copies/mL at week 24 compared with 76% of those with 6 or 7 mutations and 33% of those with 8 to 10 mutations. New agents– Protease Inhibitors BMS-232632: • Active against 89% of virus isolates resistant to fewer than 4 PIs in vitro. • Loss of sensitivity is correlated with high-level resistance to at least 4 PIs. • High incidence of unconjugated hyperbilirubinemia Tipranavir • Active against multi-PI resistant isolates. • 87% of isolates > 10-fold resistance to 4 PIs remained completely susceptible to tipranavir in vitro New agents– NNRTI 1. 2. 3. 4. Capravirine. Emivirine DMP-961 DMP-083 All show activity against viruses with 1 or more of the common NNRTI mutations. Barriers to the Development of an Effective AIDS Vaccine • Sequence variation • Protective immunity in natural infection not clearly established • Lack of adequate animal model to study vaccine protection with HIV • Latency and integration of HIV into host genome • Transmission by cell-associated virus • Limited knowledge about mucosal transmission and immune responses • Financial disincentives • Ethical issues Conclusion • Better understanding of the HIV has allowed better treatment modalities. • Cure is beyond reach at this stage, but patients can survive years to decades longer. • More drugs and drug problems are on the horizon. • Control of HIV replication by the host immune system may be the best outlook for future research. • Intense vaccine research is ongoing and ultimately will be the major preventive measure against HIV infection Immunotherapy Immunotherapy • Directions – Augmentation of specific immune response to control viral replication. – Preventive Vaccines. Clues to immune control of HIV? Subject JP: Sx: Dx: F/U: Subject 161J: Fever, Rash, Headache Sx: HIV ELISA Neg. Dx: HIV RNA >700,000 Extensive Rx F/U: AIDS at 11 mo. Rapid CD4 cell decline Viral Load >750,000 Fever, Rash, Headache HIV ELISA Neg. HIV ELISA Pos. No Rx Well at 19 yrs. CD4 1000 Viral Load < 500 Acute HIV-1 infection Stimulation of HIV-1-specific immune CD4 cells (Helper cells) Generation of HIV-1-specific killer cells (CTL) Loss of CTL function due to inadequate HIV-1-specific helper cells Infection of activated helper cells Loss of HIV-1-specific helper cells Progression T helper cells are the central orchestrator of the immune system CTL Function T helper cell B Cell Function Antibody Production NK Cell Function APC Function Cytokine production Acute HIV-1 infection Stimulation of HIV-1-specific immune CD4 cells (Helper cells) Antiviral Rx Generation of HIV-1-specific killer cells (CTL) Maintenance of CTL function due to adequate HIV-1-specific helper cells Nonprogression Protection of activated helper cells Maintenance of HIV-1specific helper cells HIV-1-specific T helper cells in individuals treated during acute infection (n=7) 100 0 Months 2 Months 10 1 MB JC KM ND SJ DK KS