* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download GLAUCOMA MEDICATIONS - Nevada Optometric Association
Compounding wikipedia , lookup
Discovery and development of beta-blockers wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Pharmacognosy wikipedia , lookup
Polysubstance dependence wikipedia , lookup
Drug discovery wikipedia , lookup
Theralizumab wikipedia , lookup
Drug design wikipedia , lookup
Drug interaction wikipedia , lookup
Pharmaceutical industry wikipedia , lookup
Psychopharmacology wikipedia , lookup
Pharmacokinetics wikipedia , lookup
Prescription costs wikipedia , lookup
Pharmacogenomics wikipedia , lookup
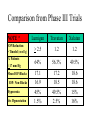
GLAUCOMA MEDICATIONS WHAT WE HAVE, WHERE WE’RE GOING… Jill Autry, OD, RPh Eye Center of Texas, Houston [email protected] THE HISTORY OF GLAUCOMA • The terms “glaucosis” and “hypochyma” were used synonymously • Vague terminology meaning “greenish/bluish” discoloration • Used indiscriminately in discussions of blindness until 1800s • “Hypochyma” linked later to cataract and considered treatable • “Glaucosis” was the incurable association THE HISTORY OF GLUCOMA • In 1622, linked to firmness of globe – “humour settled in hollow nerves…the eye grown more solid and hard than natural…” – Thought to be disorder of vitreous or choroid • In 1820, glaucoma and cataract were differentiated • In 1840, the term glaucoma was linked to increased IOP but only in regards to acute or absolute glaucoma • In 1857, the ophthalmoscope was invented allowing for view of optic nerve damage THE HISTORY OF GLAUCOMA TREATMENT • In 1857, iridectomy was introduced for acute glaucoma • In 1875, the use of a miotic for acute glaucoma • In 1935, used medications for the treatment of a less acute form of increased IOP which can lead to same end result • In 1957, oral CAI for use in glaucoma treatment DIAGNOSIS • • • • • • Gonioscopy Optic nerve examination Intraocular pressure Pachymetry Visual field Nerve fiber layer analysis AQUEOUS AND ANATOMY • Aqueous is continuously produced by the ciliary body – 2-3 µl/minute produced on a diurnal curve – Turnover every 1.5-3 hours • Aqueous flows from the posterior chamber through the pupil into the anterior chamber • Aqueous filters largely through the trabecular meshwork (90%) • Aqueous also exits to a smaller extent through the ocular venous system (10%) – Uveoscleral outflow (ciliary body, choroid, scleral vessels) AUTONOMIC NERVOUS SYSTEM • Sympathetic regulation – Fight and flight – 2 main classes of receptors • Beta receptors (ß1 and ß2) • Alpha receptors (α1 and α2) • Parasympathetic regulation – Lay down on the couch and go to sleep AUTONOMIC NERVOUS SYSTEM • Pupil is controlled by both – Sympathetic system dilates the pupil by stimulating the contraction of dilator muscle – Parasympathetic system constricts the pupil by causing contraction of the sphincter muscle. • Ciliary body is controlled by both – Sympathetic system for aqueous production – Parasympathetic system causing ciliary body muscle movement PILOCARPINE • Cholinergic, parasympathomimetic agent • Mechanisms of action are completely mechanical • Causes miosis of pupil by contraction of iris sphincter muscle – Constricts the pupil pulling the peripheral iris away from the trabecular meshwork • Pulls scleral spur posteriorly and internally – Produces alterations in ciliary body mediated configuration of the outflow apparatus PILOCARPINE • • • • • Max of 10-20% IOP reduction Available in 0.5%, 1%, 2%, 3%, 4%, 6% Pilopine HS 4% gel 1% or 2% is most widely used Chronic use limited by efficacy, compliance factors, and side effects PILOCARPINE • Adverse effects and limitations – Pupil constriction • Permanent after long term use – Induced myopia – Headache • Accommodative spasm – Blurred vision • Accommodative spasm – Retinal detachment – Frequency of use WHEN TO USE PILO • Acute angle closure – Verify with gonioscopy • Other eye should be narrow, too – Usually hyperopic, older patients with increasing lens size – Can be precipitated by certain medications – ??? Wait until IOP is below 40mmHG???? • Prophylaxis against angle closure WHY NOT USE PILO MORE? • Miosis • Young patients – Increased headache – Blurred vision secondary to fluctuating myopia • Inflammatory conditions – Increases flare in the anterior chamber • Myopic patients – More at risk for retinal detachment • Patients with cataracts – Pupil constriction limits vision – Small pupil can complicate cataract extraction Prostaglandins • Prostaglandin F2α analogues – Xalatan (latanoprost) – Lumigan (bimatoprost) – Travatan (travoprost); Travatan Z (BAK free) • Increase fluid outflow through ocular venous system (uveoscleral outflow) • Max IOP reduction of 33-40% • Once daily (qhs) meds – Twice daily yields less IOP reduction Prostaglandins • Systemic side effects are extremely rare • Allergy is extremely rare • Most side effects are local and cosmetic – – – – Conjunctival hyperemia Iris pigmentation Periorbital darkening Eyelash growth/thickening/darkening WHEN PROSTAGLANDINS ARE YOUR FIRST CHOICE • • • • • Primary Open Angle Glaucoma Ocular Hypertension Pigmentary Glaucoma Pseudoexfoliative glaucoma Angle recession glaucoma – Not during acute episode if possible WHEN PROSTAGLANDINS ARE YOUR LAST CHOICE • Elevated IOP secondary to trauma • Inflammatory glaucoma – Glacomatocyclitic iritis (aka Possner-Schlossman) – Fuch’s Heterochromic iridocyclitis – IOP increases due to herpetic disease • History of/concern of inducing macular edema – Diabetic with macular edema, epiretinal membrane • • • • Steroid induced glaucoma Post-surgical IOP spike Neovascular glaucoma Unilateral treatment Comparison of Prostaglandins • Similar in ability to lower pressure – Lumigan 0.03% is lower across more time points – Xalatan has highest non-responder rate • All are associated with hyperemia – Structure mediated, not preservative mediated – Lumigan 0.03%=Travatan Z>Xalatan – Less severe hyperemia (66%) with new Lumigan 0.01% compared with Lumigan 0.03% • All may cause iris color and eyelash growth – Iris color changes permanent • Least with Lumigan; most with Xalatan – Eyelash changes impermanent; most with Lumigan Comparison from Phase III Trials NOTE * Lumigan Travatan Xalatan IOP Reduction > Timolol (mmHg) > 2.5 1.2 1.2 % Patients < 17 mm Hg 64% 56.3% 49.5% 17.1 16.9 45% 1.5% 17.2 18.5 49.5% 2.5% 18.6 18.6 15% 16% Mean IOP-Blacks IOP- Non-Blacks Hyperemia Iris Pigmentation WHEN ADDING AGENTS • Think mechanism of action • Best chance of additivity by combining medications with different mechanisms • PGAs lower IOP by increasing aqueous outflow (uveoscleral/trabecular) • Complement a PGA by adding a drug that inhibits aqueous production – Brimonidine (also has uveoscleral MOA) – CAI – Beta-blocker CILIARY BODY AND AUTONOMIC NERVOUS SYSTEM • Sympathetic system increases aqueous production – Through stimulation of ß receptors – ß blockade decreases aqueous production • Sympathetic system decrease aqueous production – Through activation of α2 receptors – α2 agonists decrease aqueous production ALPHA-2 AGONISTS • 2-adrenergic agonist • Apraclonidine (Iopidine) • Brimonidine (Alphagan) – Enhanced α2 selectivity due to double ring structure BRIMONIDINE • Primary mechanism of action is decreased aqueous production – Great additive agent to PGA • Secondary mechanism of action is enhanced uveoscleral outflow – Great combination agent with timolol • Max IOP reduction of 20-30% • Bid to tid dosing ALPHAGAN P 0.1% • Purite preservative • Higher pH – Neutral, nonionized form is better absorbed • Decreased drug concentration – 50% decrease • Unaltered efficacy • Less chance for local allergy • Less chance for systemic side effects – Dry mouth, fatigue, hypotension OCULAR ALLERGY • Ocular allergies in up to 30% of patients – Original Alphagan 0.2% and generic brimonidine 0.2% • 30% allergy rate – Alphagan P 0.15% • 20% allergy – Alphagan P 0.1% • 10% allergy WHEN TO USE BRIMONIDINE • Additive agent to a PGA – First or second line addition • • • • Monotherapy with PGA is contraindicated Post-op IOP spikes Concerns for preservative toxicity Only category B glaucoma drop for pregnancy WHEN NOT TO USE BRIMONIDINE • History of allergy to brimonidine in any concentration • Eyelid swelling, tenderness, itching, follicular reaction – Can develop within weeks/months of initiation or even years later • Patients prone to hypotension • Patients with complaints of somnolence CILIARY BODY AND AUTONOMIC NERVOUS SYSTEM • Sympathetic system increases aqueous production – Through stimulation of ß receptors – Beta blockade decreases aqueous production • Sympathetic system decrease aqueous production – Through activation of α2 receptors – α2 agonists decrease aqueous production BETA BLOCKERS • • • • Decrease aqueous production No effect on outflow Max IOP reduction of 20-30% Once to twice daily (qd to bid) dosing – qd dosing equivalent to bid dosing • May be less effective if on oral beta blocker Beta Blockers Timoptic (timolol) Timoptic XE (timolol gel) Betimol (timolol) Betagan (levobunolol) OptiPranolol (metipranolol) Ocupress (carteolol) Betoptic S (betaxolol) CONCERNS WITH TREATMENT • Elderly • Lung Disease – Contraindicated in asthma, COPD, etc. • Heart disease – Contraindicated in CHF (heart failure) • Diabetes • Impotence Selective Beta Blocker • May cause less side effects • Still use cautiously • Betoptic S (betaxolol suspension) – Fewer side effects on the lung – Decreased efficacy vs. other beta blockers LONG TERM EFFICACY • Effect diminishes with time • First few weeks is “short-term” escape – Up-regulation of beta receptor numbers • Long-term drift – A receptor or intracellular tolerance develops WHAT ABOUT COMBIGAN? • • • • Alphagan 0.2% with timolol 0.5% Complementary mechanism of actions Dosed BID Less allergy than any of the other Alphagan products (5% vs 20% allergy rate) – 50% less than 0.2 brimonidine • Advantages of combination therapy COMBIGAN™ in Adjunctive Therapy With a PGA: Mean IOP Mean IOP (mm Hg) 24 21.9 Added to a PGA baseline -6.9 mm Hg (29%) 20 16 15.2 * 15.3 * 12 COMBIGAN™(brimonidine tartrate/timolol maleate 8 ophthalmic solution) 0.2%/0.5% + PGA (n = 37) 4 *P < .0001 vs baseline 0 0 1 2 3 Month 1Nixon and Hollander. 2AAO, 2007. Data on file, Allergan, Inc. CARBONIC ANHYDRASE • Carbonic anhydrase is an enzyme present in the biochemical production of aqueous – Causes bicarbonate and hydrogen movement • Inhibition of carbonic anhydrase – Blocks active transport needed for aqueous production • End result is reduction of aqueous humor formation • Subsequent decrease in intraocular pressure TOPICAL CAI • • • • • • Reduce aqueous humor production Max IOP reduction of 15-20% bid to tid dosing Dorzolamide (Trusopt®) Brinzolamide (Azopt®) Dorzolamide + Timolol – Cosopt® ADVERSE EFFECTS/CONCERNS • • • • • • Bitter taste Stinging Conjunctival hyperemia Tachyphylaxis Concerns with history of sulfa allergies Corneal concerns SULFA ALLERGY • Sulfa allergy not sulfur allergy • Rash is common sign; usually seen in the antibiotic class of sulfonamides (like Septra or sulfacetamide ointment) • Less likely to see in non-antibiotic meds • Diamox, Neptazane, Azopt, Trusopt, Cosopt • Even less likely to see with topical medications • Sulfites and sulfates are chemically different-no cross reactivity with sulfa allergies CORNEA AND CAI • Invest Ophthalmol Vis Sci, 2008 Mar;49(3):104855. – Role of carbonic anhydrase in corneal endothelial HCO3-transport. • Arch Ophthalmol. 2007 Oct;125(10):1345-50. – Effect of dorzolamide on central corneal thickness in humans with cornea guttata. • Arch Ophthalmol. 204 Jul;122(7):1089. – Short-term effect of dorzolamide on central corneal thickness in humans with cornea guttata. INDIGENT PROGRAMS • Allergan (Lumigan, Alphagan P, Combigan) – 1-800-553-6783 • Alcon (Azopt, Travatan Z, Betoptic S) – 1-800-222-8103 DIAMOX (Acetazolamide) • Nonbacteriostatic sulfonamide • Decreases carbonic anhydrase – Decreases hydrogen and bicarbonate formation • Results in decreased aqueous production in the ciliary body by producing a systemic acidosis • Results in alkaline diuresis in the kidney but tolerance develops quickly • Contraindicated in renal, hepatic, or respiratory disease DIAMOX (Acetazolamide) • Decreases carbonic anhydrase in the ciliary body which decreases aqueous humor formation • Decreases IOP by 40-60% • Starts to work in 1 hour, peak effect at 4 hours • Comes in 125mg, 250mg, 500mg sequels • Angle closure dose: (2) 250mg tablets initially—needs PI ADVERSE EFFECTS • Metallic taste • Paresthesias (“pins and needles”) • Used mostly in emergencies because of side effects with chronic use – – – – – Kidney stones Acute respiratory failure Acid-base imbalances Blood dyscrasias (aplastic anemia) Induced myopia USE IN LOWERING IOP • Treatment of acute angle closure glaucoma • Treatment of less acute increased IOP • Treatment of post-surgical IOP spikes ORAL CAI USES • Treatment of pseudotumor cerebri • Treatment of other causes of increased intracranial pressure • Controversial treatment of serous retinal detachments • Treatment/prevention of “altitude sickness” OTHER USES FOR GLAUCOMA DROPS • May be helpful with Fuch’s patients – Avoid CAIs • Stabilizing visual acuity/Rx in RK patients – Especially prostaglandins given duration of action • Brimonidine can – Decrease pupil size to eliminate glare – Decrease hyperemia – Lacks side effects of pilocarpine OTHER USES FOR GLAUCOMA DROPS • Iopidine® to diagnose Horner’s? – Has been demonstrated to have same sensitivity as cocaine test for diagnosis of Horner’s – Alphagan less likely to give reliable results secondary to increased alpha-2 selectivity • Pilocarpine for diagnosis of Adie’s pupil – 1% dilute 1:10 to make 0.1% solution – 2% dilute 1:20 to make 0.1% solution ADIE’S TONIC PUPIL • • • • • • Usually young female Poor reaction to light Slow constriction to near Slow redilation following near constriction Vermiform movement Constricts to 0.125% pilocarpine – May not constrict in initial stage • Long standing can result in small pupil Oral Pilocarpine • Salagen® (oral pilocarpine) – 5 mg qid for dry mouth • Approved for dry mouth with Sjogren’s patients • Approved for dry mouth associated with head/neck radiation • Also used Evoxac® – 30 mg tid Novel Pharmaceuticals • • • • Adenosine A1 receptor antagonist TGF-beta2 growth factor inhibition Wnt antagonist sFRP1 Decreasing neuronal toxicity – Glutamate inhibition – Nitrous oxide inhibition – Capsase enzyme antagonists • Decrease matrix metalloproteinases TOPICAL APPLICATION AND GLAUCOMA • Corneal permeability – Less than 5% of drop is absorbed through cornea • • • • • • Conjunctival vasculature loss of drug Tear wash out Drug solution drainage through nasolacrimal system Spillover out of the eye High concentrations inducing local reactions Preservative toxicity SYSTEMIC TREATMENT AND GLAUCOMA • Blood-Aqueous Barrier – prevents systemically administered substances from entering into the aqueous humor • Blood-Retinal Barrier – severely limits drug entry into the extravascular space of the retina and into the vitreous • Need high systemic doses to overcome • Results in higher side effects/toxicity Current Ocular Drug Delivery • Drops – Less than 5% of drop is absorbed into cornea or anterior chamber – Ocular toxicity of preservatives • Subconjunctival injections • Periocular injections • Intravitreal injections – Risk of infection/IOP spikes/RD • Sustained release implants • Systemic-oral and intravenous – Exposes whole body to potential toxicity Advances in Ocular Drug Delivery • Formulation factors – Surfactants-dispersion of solutes – Viscolyzers-retention and bioavailability • Instilled volume – Reduced with thicker agents • Administration technique • Non-preserved buffering techniques – Minimizes toxicity – More natural pH Advances in Ocular Delivery • Increased ocular contact time – – – – Ointments Gels Liposome formulations Nanoemulsions • Colloidal systems – Disperse and encapsulate meds GLAUCOMA AND DRUG DELIVERY • Controlled release – Sustained pharmaceutical levels – Sustained IOP control • Decreased frequency of dosing – Compliance • Reduced drug concentration – Decreased local side effects – Avoid systemic side effects DRUG RELEASING CONTACT LENS • Concerns – – – – – – – Ability to load total drug mass onto contact lens Ability to release drug at therapeutic doses Avoid creating ocular toxicity Maintaining optical clarity Maintaining refractive properties Amenability to storage Biocompatibility DRUG RELEASING CONTACT LENS • Advantages – – – – Increase compliance Increase contact time Decrease medication concentration Up to 50% absorption through cornea DRUG RELEASING CONTACT LENS • Hydrogel contact lens trial – Timolol and Brimonidine – Maximum uptake of drug and release was limited – Silicon-hydrogels were similar • Molecularly imprinted hydrogel CL – Better retention of drug and slower release – Smaller molecules load better DRUG RELEASING CONTACT LENS • Liposomal coated hydrogel contact lenses – Lidocaine and levofloxacin trials • Drug entrapped contact lenses – Polymer technology – Drug containing surfactants – Drug polymer film coated in a hydrogel lens • Proven linear release kinetics PLUG BASED DELIVERY SYSTEM • Drug core inside punctal plug • Sustained drug release over time • Phase II clinical trial – Latanoprost was used – L-PPDS • Latanoprost punctal plug delivery system Subconjunctival Route • Safer and less invasive than the intravitreal route • Systemic absorption is low • Lower systemic side effects while providing a localized drug effect • Substantial evidence indicating that drugs administered subconjunctivally can reach the vitreous effectively • Injectable microspheres have already been tested – Timolol with continual release up to 107 days INTRAVITREAL DELIVERY • Vitrasert – Ganciclovir implant for AIDS associated CMV • Retisert – Fluocinolone implant for posterior uveitis • Ozurdex – Dexamethasone injectable, biodegradable pellets for RVO • Medidur – Fluocinolone injectable implant for diabetic macular edema IN CONCLUSION… Jill Autry, OD, RPH [email protected]