* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download (+) Treatment - yeditepetip4
National Institute for Health and Care Excellence wikipedia , lookup
Discovery and development of direct Xa inhibitors wikipedia , lookup
Adherence (medicine) wikipedia , lookup
Intravenous therapy wikipedia , lookup
Theralizumab wikipedia , lookup
Pharmacogenomics wikipedia , lookup
Discovery and development of direct thrombin inhibitors wikipedia , lookup
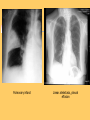
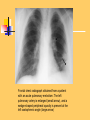
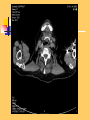
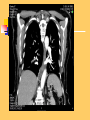
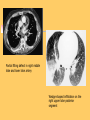
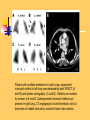
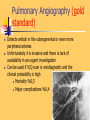
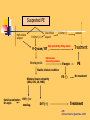
Pulmonary Thromboembolism Prof. Sevda Özdoğan MD Chest Diseases Pathophysiology of Pulmonary Embolism Virchow Triade 1. 2. 3. Venous stasis Vascular endothelial (wall) damage Hypercoagulation Risk factors The risk factors for VTE can be both genetic or acquired for a certain patient Risk increase if age>40 hypercoagulability) (Comorbidity, stasis, Acquired Risk Factors Risk Factor Previous DVT, PE Major surgery Malignancy Obesity Trauma Fracture (Hip, leg) Pregnancy MI Congestive hearth failure Stroke Estrogen treatment Immobilization Burns Hypercoagulability Stasis Trauma Genetic Risk factors Genetic Activated protein C (APC) resistance Factor V Leiden mutation Positive in 21% of VTE patients Antithrombin III deficiency Protein C and S (cofactor) deficiency Prothrombin G20210A Hyperhomocystinemia Autosomal dominant Risk of VTE x5 Autosomal dominant Risk od VTE x6 A single nucleotide change in prothrombin gene results in elevated prothrombin levels Risk of DVT x5 Defects in enzymes of homocystein disposal Risk of VTE x2 Genetic Increased Factor VIII Blood group other than O RR of DVT x2 Combination of the genetic risk factors RR for VTE x4.8 Clinical features and Diagnosis Clinical suspicion*** Medical history: To identify the patient at risk Family history Medical or acquired risk factors Symptomatology: Unexplained acute dyspnea Tachypnea Substernal chest discomfort Pleuretic chest pain Hemopthysis Cyanosis Shock / sencope Asymptomatic Physical findings: (nonspecific) 97% Diagnostic approach Semptomatology and signs Chest radiology Arterial blood gas analysis (ABG) Electrocardiography Standard laboratory tests Echocardiography (Cardiac and venous doppler of the lower extremity) D-Dimer Spiral CT or Ventilation / perfusion scan Pulmoner angiography (gold standard) Chest Radiography Negative chest radiogram is a common presentation so does’t exclude the diagnosis 80% Abnormal chest radiograph but nonspecific Early: Peripheral regional oligemia (Westermark’s sign) (7%) A prominant pulmonary hilus with little tapering of vessels (Fleischner’s sign) (15%) Later: Peripheral wedge shaped densities (Hampton’s hump) (35%) Plate like atelectasis Diaphragmatic elevation (%24) Pleural effusion (%48) Pulmonary infarct Linear atelectasis, pleural effusion Frontal chest radiograph obtained from a patient with an acute pulmonary embolism. The left pulmonary artery is enlarged (small arrow), and a wedge-shaped peripheral opacity is present at the left costophrenic angle (large arrow) ABG Analysis Hypoxemia, hypocapnia and respiratory alcalosis PaO2 <%80 PaO2 may be normal in submassive embolism if no underlying pulmonary disease is present (A-a)O2 gradient is increased in almost all the patients ECG Abnormalities of ECG are nonspecific Acute right ventricular strain in massive embolism Sinus tachicardia Negative T wave and/or ST segment depression in leads V1-3 S1Q3T3 patern (Deep S wave in lead D1, deep Q wave in lead D3, inverted T waves in D3) Right bundle branch block (complete or incomplete) P-pulmonale Changes can be similar to MI Standard laboratory tests Nonspecific changes WBC can be slightly elevated LDH, bilirubine can be slightly elevated D-Dimer (fibrin degradation product) can be elevated ELISA or Latex agglutination Sensitivity % 95-97 but specificity is low <500 ng/ml PE can be excluded if there is also low clinical probability Elisa is more sensitive but slow compared to Latex ECHOCARDIOGRAPHY (Doppler) Can be performed rapidly at the bedside Features that suggest acute massive PE include A dilated, hypokinetic right ventricle With the absence of right ventricular hypertrophy Distortion of the interventriculer septum toward the left ventricle Tricuspit regurgitation the elevation of pulmonary artery pressure Identified trombi in the central pulmonary arteries Absence of significant pathologic left ventricular conditions Spiral Computed Tomography Angiography (SCTA) May demonstrate or exclude other abnormalities in the lung Bolus contrast is used for the visualization of the pulmonary vasculature Filling defects are diagnostic Sensitivity and specificity is around 90% up to subsegmental defects Partial filling defect in right middle lobe and lover lobe artery Wedge shaped infiltration on the right upper lobe posterior segment Ventilation-Perfusion Scintigraphy Detection of the perfusion abnormalities subsequent to the embolic event Classically to display that a segment distal to an obstructing embolus is not perfused but is still ventilated 99Tc is usually used for perfusion and 133Xe for ventilation scaning. The two studies are analysed together. In clinical practice the results of V/Q scintigraphy are interpreted together with the clinical estimate of the likelihood of acute PE A normal V/Q virtually excludes clinically relevant PE Patient with multiple embolisms in both lungs: segmental mismatch defect in left lung was detected by both SPECT (A and B) and planar scintigraphy (C and D). Defects are marked by arrows in B and D. Subsegmental mismatch defects are present in right lung. CT angiography found thrombotic clots in branches of middle lobe artery and both lower lobe arteries Pulmonary Angiography (gold standard) Detects emboli in the subsegmental or even more peripheral arteries Unfortunately it is invasive and there is lack of availability in an urgent investigation Can be used if V/Q scan is nondiagnostic and the clinical probability is high Mortality %0,5 Major complications %0,4 Suspected PE High clinical suspect D dimer (+) Low clinical suspect V-Q scan/ BT Nondiagnostic D dimer (-) exclude High probability/filling defect Hipotension Severe hypoxemia P.angio Treatment PE Stabile clinical condition Bilateral lower extremity (USG, IPG, CV, MRI) Serial examination Or angio PE (-) No treatment DVT (-) or nondiag . DVT (+) Treatment ATS Clinical Practice Quideline-1999 Deep Venous Thrombosis (DVT) Compression ultrasound Doppler ultrasonography Venography (gold standard) Treatment of PTE and DVT Supportive treatment Anticoagulant therapy Oxygen Intravenous fluid Vasopressor agents Resuscitary measures depending on the clinical status of the patient Unfractionated heparin (UFH) Low molecular weight heparin (LMWH) Oral anticoagulants (Warfarin, rivaroxaban) Thrombolytic treatment Surgical treatment UFH Binds to AT-III Anticoagulant factors are secreted from vascular endothelial cells Inhibits platelet aggregation Its effect is prophlactic for the recurrences, not thrombolytic Administration of UFH APTT monitorisation should be performed 5000 UI bolus + 25.000-35.000 UI/24 hr. Or 1000 IU/hr continious iv infusion aPTT check in 6 hours (x1,5-2,5), platelet count in 3-5 days Probability of recurrent VTE 5.4% and major hemorrhagia 1.9%. Body Weight-Based Dosing of Intravenous Heparin Initial dosing: Loading 80 U/kg 18 U/kg/hr (APTT in 6 hrs) APTT(s) (x normal) Dose Change Additional (U/kg/h) Action <35 (1.2 x) 35-45 (1.2-1.5x) 46-70 (1.5-2.3x) 71-90 (2.3-3.0x) >90 (<3x) +4 +2 0 -2 -3 Next APTT (h) Rebolus 80 U/kg Rebolus 40 U/kg 0 0 Stop infusion 1 h * APTT check during first 24 hr, there after once a day 6 6 6* 6 6 bed rest until heparin is therapeutic elastic stockings until patient becomes ambulatory ( post-thrombotic syndrome) Oral anticoagulant can be given as warfarin (Coumadin)5 mg/day on the first 24 hours, when prothrombin time (PT) becomes x22.5 (INR 2-2.5)heparin can be stopped Antidode of UFH is protamine sulphate Antidote of warfarin is Vitamin K Complications and side effects of heparin Hemorrhagia (major % 0.5-3, fatal %0-0,8) Thrombocytopenia (The risk is lover in LMWH but if the condition occurs due to autoantiplatelet antibodies it is a fatal complication. Heparin should be stopped and an alternative anticoagulant (Hirudin etc) should be given) Osteopenia Reversible condition and the risk is high in prolonged use of the drug Alopecia Cutaneous rush Hypersensitivity reactions Urticeria, konjonctivitis, rhinitis, asthma, angioneurotic edema Contraindications for heparin Active hemorrhagia Recent cerebrovascular hemorrhagia History of major hemorrhagia from gastrointestinal, genitourinary or respiratory system LMWH Weight adjusted fixed dose subcutaneous application is possible without laboratory monitoring Safer and better biopharmacology HIT, osteopenia complications are less As plasma half life becomes longer in renal failure and morbid obesity anti Xa should be monitored in this group Low-Molecular-Weight Heparin Drug Treatment Dose Ardeparin 130 anti-Xa U/kg bid (Normaiflo) Dalteparin 120 anti-Xa U/kg bid (Fragmin, ) Enoxaparin 1-1.5 mg/kg bid (Lovenox, Clexane) (1 mg 100 anti-Xa units) Danaparoid (Orgaran) Tinzaparin 175 IU/kg once a day (Innohep) Warfarin Oral agent Inhibits the synthesis of protrombin, Protein C, S,f II, VII, IX, X’un related to vitamin K Plasma half life 42 hr Monitorization PT(x2-3), INR(2-3) 5 mg/day is started in the first 24 hours of treatment Rivaroxaban (New treatment) Inhibitor of Factor Xa Oral (Xarelto) 2x15 mg (3 weeks,) followed by 1x20 mg Laboratory monitoring not needed Plasma half life 5-9 hours Dose should be reduced in renal failure Side effects anemia, dizzinesss, vomiting, hemoragia Dabigatran Trombin inhibitor Oral (Pradaxa) 2x150 mg Laboratory monitoring not needed Peak efficacy 1-4 hours after ingestion Plasma half life 12-14 hr but can be longer in renal failure, old age No antidote!! Can not be used in pregnancy, Dyspepsia, hemoragia Indications for thrombolytic treatment Massive Pulmonary Embolism Hypotension Deep hypoksemia Right ventricular disfunction/iskemia Cardiovascular collaps Thrombolytic treatment should be performed immediately Can be performed in the first 14 days Thrombolytics Drug Loading dose Infusion dose Treatment duration 250 000 IU 30 min 100 000 IU/hr 24 hours 4400 UI 10 min 4400UI/kg/hr 12 hours - 100 mg 2 hours - Streptokynase Urokynase rt-PA Complications Hemorrhagia (intracranial 1-2%) Fever, alergic reactions,nausea, vomiting, myalgia, headache Contraindications Cerebral surgery or hemorrhagic attack within the last 2 months Active intracranial disease Uncontrolled hypertension Hemorrhagic diathesis Infective endocarditis Pregnancy Hemorrhagic rethinopathy Pericarditis Aneurism Treatment duration Reversible risk factor, first event, age<60 : 3-6 months Reversible risk factor, first event, age>60: 6-12 months First event, unknown risk factor: 6-12 months Recurrent event: >12months- life long Irreversible risk factor, first event: >12 months- life long Vena Cava Filters If there is a contraindication for anticoagulation If a complication due to anticoagulation occurs Failure: new DVT or PE under treatment Major or minor hemorragia Trombocytopenia Tissue necrosis Drug reactions For prophylaxsis Thrombectomy, embolectomy are the other surgical options Primary Prevention Determined by the thrombotic risk of the clinical situation in conjunction with the patients profile of risk factors Ortopedic surgery (post-traumatic) ICU Neurosurgery carry the highest risk Prophylaxis LMWH or UFH can be used in low doses LMWH’s can be used preoperatively safely Prophylaxis should be continued up to 4 weeks after surgery (min 10-14 days) Rivaroxaban 1x10 mg, Dabigatran 1x220 mg can be used Non medical Prophylaxis Graduated compression stockings İntermittent pneumatic compression Foot impulse pumps Early mobilization Can be used for patients who have contraindications to anticoagulants.