* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Cyanide Geochemistry - University of Manitoba
Allotropes of carbon wikipedia , lookup
Spin crossover wikipedia , lookup
Metal carbonyl wikipedia , lookup
Activated carbon wikipedia , lookup
Coordination complex wikipedia , lookup
Metalloprotein wikipedia , lookup
Stability constants of complexes wikipedia , lookup
Hydroformylation wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
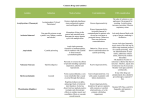
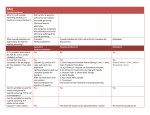
Cyanide Geochemistry Outline • • Introduction to Cyanide Cyanide in the beneficiation of gold – – – – Heap Leach Process Cyanide tank leach and CIP circuits Optimum Conditions for CN leaching Extraction of gold from the CN solution • (a) Merrill Crowe Process • (b) CIP Process • • • Cyanide Analysis Toxicity Degradation mechanisms to reduce toxicity – – – – – – – • • • • • 1. Volatolization 2. Complexation 3. Adsorption 4. Oxidation to Cyanate 5. Formation of Thiocyanate, SCN6. Hydrolysis 7.Biodegradation Cyanide degradation in a Heap Leach Cyanide degradation in Mill Tailings Examples of Cyanide Spills Summary References Introduction to Cyanide • • • • • • • • • 1.4 m tonnes CN produced annually 13% CN is used for the extraction of Au and Ag: 460 of 875 Au/Ag mines use CN 87% used in production of paint, adhesives, cmputer electronics, fire retardants, cosmetics, dyes, nylon, Plexiglas, rocket propellant and pharmaceuticals Cocaine CuCN.9(C17H19O3N.HCN).7HCN Novocaine CuCN.9(C17H20O2N.HCN).HCN Codeine CuCN.4(C18H21O3N.HCN).3HCN Nicotine CuCN.2(C10H14 N2.HCN).1.5HCN Morphine CuCN.9(C17H19O3N.HCN).7HCN Caffeine 4CuCN.(C8H10O2N4.HCN) Natural Cyanide • Cyanide is naturally produced by both fauna and flora. • Humans have <0.217 g/l SCN in saliva, <0.007 g/l SCN in urine and <0.006 g/l in gastric juices. • Cyanogenic bacteria generate cyanide from glycine. NH2CH2COOH = HCN + CO2 + 2H2 Cyanide in the beneficiation of gold • 0.05% NaCN solution is used to extract Au and Ag from ore • Au dissolves by two processes occurring simultaneously on its surface. Cathode • At one end of the metal, the cathodic zone, oxygen takes up electrons and undergoes a reduction reaction. O2 + 2 H2O + 2 e- => H2O2 + 2 OHAnode • At the other end, the anodic zone, the metal gives up electrons and undergoes an oxidation reaction. Au => Au+ + eAu+ + 2CN- => Au(CN)2- • And then form strong complexes by Elsener’s/ Adamson’s 1st reaction: 4Au + 8NaCN + O2 + 2H2O = 4NaAu(CN)2 + 4NaOH Or Adamson’s 2nd reaction 2Au + 4NaCN +2H2O = 2NaAu(CN)2 + H2O2 + 2NaOH Heap Leach Process Cyanide tank leach and CIP circuits Optimum Conditions for CN leaching • The rate of Au dissolution is determined by the rate at which the dissolved oxygen and/or the cyanide ions permeate or diffuse through the Nernst layer (~0.05 mm) which surrounds the surface of Au. – CN tanks must be aerated by agitation or by pumping air through. • Increasing the temperature of the leach solution will promote the dissolution of Au, but as the temperature increases, the solubility of oxygen decreases. – The optimal temperature is 60 to 80º C. • Other metallic species from ore minerals, e.g. sphalerite (ZnS), chalcocite (Cu2S), chalcopyrite (CuFeS2), bornite (FeS.2Cu2S.CuS), will form complexes with CN. – Therefore more CN is needed than for just Au complexation. – The tailings will contain these complexes. Extraction of gold from the CN solution (a) Merrill Crowe Process • Merrill Crowe process discovered and patented by Charles Washington Merrill around 1900, thenrefined by Thomas B. Crowe, working for the Merrill Company • Zinc replaces Au in the NaAu(CN)2 complex, as it has a higher affinity for CN- than gold NaAu(CN)2+ Zn = NaZn(CN)2 + Au • Au precipitates as a solid. • Early zinc precipitation systems simply used a wooden box filled with zinc chips. They were very inefficient and much of the dissolved gold remained in solution. • The Merrill-Crowe process works better than the early zinc boxes because it uses zinc powder and reduces the amount of dissolved oxygen. (b) Carbon in Pulp (CIP) • Carbon in Pulp was introduced in 1985, • Granular activated carbon particles (burnt coconut shells) have a high porosity, each pore is about 10-20 Å and the surficial area is >1000 m2/g. • The carbon particles are much larger than the ground ore particles. • The activated carbon and cyanided pulp are agitated together. • Au(CN)2 becomes adsorbed onto the charged surface of the activated carbon. • The loaded activated carbon is mechanically screened to separate it from the barren ore pulp • The gold adsorbed on the activated carbon is recovered from the carbon by elution with a hot caustic aqueous cyanide solution. • The carbon is then regenerated and returned to the adsorption circuit • The gold is recovered from the eluate using either zinc cementation or electrowinning. • The gold concentrate is then smelted and refined to gold bullion that typically contains about 70 - 90% gold. • The bullion is then further refined to either 99.99% or 99.999% fineness using chlorination, smelting and electro-refining. CIP circuit Cyanide Analysis CN is difficult to analyze because of the difference in solubility of the various complexes. 1. Weak acid dissociable (WAD) cyanide. • Most often used as it measures the cyanide which would be easily leached in mildly acidic conditions including free cyanide and weakly complexed cyanide (with Cd and Ni). • The WAD technique is least susceptible to interference and overestimation. • There are two methods of analysis: • a) Reflux distillation for one hour in mild acid, buffered with acetate to pH of 4.5. HCN collected and measured by titration • b) Picric Acid titration • 2. Cyanide amenable to chlorination • Analyses the same compounds as WAD and is accepted by the US EPA. • A two step process measures CN evolving before and after chlorination 3. Total Cyanide: • Reflux for one hour in strong acid which dissociates most complexes and measure HCN which is absorbed in NaOH solution. • Analytical interferences from oxidizing agents, sulphides, sulphates, thiocyanate, nitrate, nitrite, carbonate, thiosulphates. TOXICITY • Cyanide binds to the active Fe atom in cytochrome c oxidase and inactivates oxidative respiration. • Cyanide may be inhaled ingested or absorbed through the skin but does not accumulate in the body. • HCN and CN- are acutely toxic if inhaled or ingested and result in convulsions, vomiting, coma and death. • Lethal doses (LD 50) of KCN or NaCN: 1.1-1.5 mg/kg of body weight. • Lower long term concentrations result in neuropathy, optical atrophy, pernicious anaemia. • Cyanide complexes are not as toxic as free cyanide and their toxicity depends on ability of the gut to break down the complex and absorb the free cyanide. • Ferric ferrocyanide is used as an antidote to thallium poisoning. Degradation mechanisms to reduce toxicity 1. Volatilization Reaction between cyanide and water produces HCN gas CN- + H2O = HCN + OH- • • At pH < 8.3 HCN is the dominant species. Therefore cyanide leaching operation is kept at a pH over 10. • HCN is a colourless liquid or gas: with a boiling point of 25.7oC. • Reaction is dependant on pH (<pH7 99% will be HCN), cyanide solubility, HCN vapour pressure, and CN concentration in solution. Degradation mechanisms to reduce toxicity 2. Complexation 72 complexes with varying solubilities are possible from 28 elements. These rapid reactions immediately remove CN- from solution. • Complexes may absorb on organic and inorganic surfaces or precipitate as insoluble salts with Fe, Cu, Ni, Mn, Pb, Zn, Cd, Sn, Ag. • Complex may dissociate in acid conditions but may persist for hundreds of years. 2a. Neutral Cyanide Compounds Soluble compounds NaCN, KCN and Ca(CN)2, Hg(CN)2 dissolve in water to give cyanide anions NaCN = Na+ + CNCa(CN)2 = Ca2+ + 2CNInsoluble Neutral Cyanide Compounds Zn(CN)2, Cd(CN)2, CuCN, Ni(CN)2, AgCN 2b Charged metal CN complexes Cyanide complexes form in order of increasing number of CN ligands with successively higher CN concentration • Weak Complexes: • [Zn(CN)4]2-, [Cd(CN)3]-, [Cd(CN)4]2• Moderately Strong Complexes: • [Cu(CN)2]-, [Cu(CN)3]2-, [Ni(CN)4]2-, [Ag(CN)2]• The rate of dissolution depends on pH, temperature, intensity of light, and bacteria • Weak and moderately strong cyanide complexes will break down at pH 4.5 so will register in the weak acid dissociable (WAD) cyanide analysis. Strong Complexes • [Fe(CN)6]4-, [Co(CN)6]4-, [Au(CN)2]-, [Fe(CN)6]3- form at pH l<9.0 and can form insoluble salts with other species. • Ferrocyanide [Fe2+(CN)6]4- (hexaferrocyanate, red) and ferricyanides [Fe3+(CN)6]3- (hexaferricyanates, yellow) are very stable in the absence of light (<100s of years) but dissociate in UV to form CNand hence HCN Fe(CN)64- + H+ = Fe(CN)53- + HCN • • • • The transformation of Fe3+ to Fe2+ leaves CN content constant. This oxidation/reduction couple is pH dependent. Reaction is very slow so most mine wastes have both species. When both Fe2+ and Fe3+ are present the compound is a deep “Prussian” blue. Degradation mechanisms to reduce toxicity 3. Adsorption • Adsorption of CN- on Fe, Al and Mn oxides and hydroxides and on clays. • Clays with high anion exchange capacity are most effective e.g. clays containing kaolinite, chlorite, gibbsite or Al or Fe oxyhydroxides • Clays with high cation exchange capacity (CEC) are less effective at scavenging CNe.g. montmorillonite. Degradation mechanisms to reduce toxicity 4. Oxidation to Cyanate • • Cyanide can be oxidized to less toxic cyanate HCN + 0.5O2 = HCNO • From the phase diagram, cyanate should be the dominant form under environmental conditions but this requires strong oxidants e.g. ozone, H2O2, plus UV, bacteria or a catalyst. • Adsorption onto organics or carbonaceous material which causes CN to become oxidized Degradation mechanisms to reduce toxicity 5. Formation of Thiocyanate, SCN• In neutral to basic solution • From oxidation products of sulphides such as chalcopyrite, chalcocite, pyrrhotite not pyrite and sphalerite. • From polysulphides Sx2- + CN- = Sx-12- + SCN- • From thiosulphates S2O32- + CN- = SO32- + SCN• SCN- behaves like a pseudohalogen and forms insoluble salts with Ag, Hg, Pb, Cu, Zn. • Complexes may react with SCN- to form even more stable compounds Degradation mechanisms to reduce toxicity 6. Hydrolysis HCN + 2H2O = NH4COOH (ammonium formate) HCN + 2H2O = NH3 + HCOOH (formic acid) • Slow reaction, 2% per month • Dependent on pH. Degradation mechanisms to reduce toxicity 7. Biodegradation • Aerobic degradation in unsaturated zones is 25 times more effective than in saturated zones HCN + O2 = 2 HCNO HCNO + 0.5 O2 + H2O = NH3 + CO2 • Anaerobic degradation in the saturated zones CN + H2S = HCNS + H+ HCN + HS = HCNS + H+ • The toxic limit for effective anaerobic degradation is 2 mg/L. • Bacteria can be used in a bioreactor to decrease CN content e.g. Landusky heap leach remediation Cyanide degradation in a Heap Leach • Cyanide decreases from >250 mg/l in leach solution to 130 mg/l in rinsate and then decays to below detection limit. Cyanide degradation in Mill Tailings • Most CN is degraded by volatilization of HCN because the pH is lowered immediately from 10 by rainwater and uptake of CO2 from air and more slowly by oxidation of sulphides. • Between 3 and 6 months, WAD CN (from CIP process) has reduced by a factor of 100 to a few ppm. • There are slight difference between surface and deep waters and between winter and summer. • There is a need to consider transformation of CN between solid, liquid and gas phases. This may be dependent on type of soil, cations, weather, bacteria, depth and degree of oxygenation of pond. Examples of Cyanide Spills • Hungary-Romania-Slovakia-Ukrain: 1-11 February 2000cyanide spill in Szamos and Tisza rivers polluted the Danube • Australia February 8, 2000: BHP fined over cyanide pollution incident • Ghana: 23rd October 2004, and 16 June 2006 BHP fined over cyanide pollution incident at the Port Kembla steel-making operation near Wollongong. • Honduras: 3rd May 2006 In the Siria Valley in Honduras, are extensive. Cyanide and heavy metal contamination of several water sources in the area of the San Martin mine has been confirmed. • Romania: 30 January 2000 Baia Mare Mine • Kyrgystan: May 20 1998, a truck carrying sodium cyanide to Kyrgyzstan's Kumtor Gold Company (one-third owned and operated by a subsidiary of the Saskatchewan-based Cameco Corporation) overturned into the Barskoon River, spilling nearly two tonnes of deadly cyanide. Summary • Cyanide/ CIP is an efficient method to extract Au and Ag. • Most CN will convert to HCN in tailings ponds or heap leach and volatilize under increasing acidic conditions or be consumed by bacteria. • CN forms complexes of varying strengths and longevity with metals • The major environmental issues relate to spills from tailings ponds, trucks pipes before CN has decomposed. Cyanide spill kills fish and wildlife immediately but the major long term problems relate to heavy metal contamination, some coming from the decomposition of metal cyanide complexes. References • Filipek, L H., (1999) Determination of the Source and Pathway of Cyanide Bearing Mine Water Seepage, in The Environmental Geochemistry of Mineral Deposits Part B Case Studies and Research Topics Eds Filipeck, L.H. and Plumlee, G.S. • Meehan, S.M. (2000) The fate of cyanide in groundwater at gaswork sites in SE Australia, PhD thesis, University of Melbourne. • Smith, A.,(1994) The Geochemistry of Cyanide in Short Course Handbook on Environmental Geochemistry of Sulphide Mine-Wastes Ed. Jambor, J.L. and Blowes, D.W. MAC • Smith, A.C.S & Mudder, T.I. (1998) The Environmental Geochemistry of Cyanide in The Environmental Geochemistry of Mineral Deposits Part A Processes, Techniques and Health Issues, eds Plumlee and Logsdon. Review in Economic Geology Volume 6A, Society of Economic Geologists. (all 11. figures and tables) •