* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Evaluation of novel affinity bio-beads for use in the production of
Survey
Document related concepts
Transcript
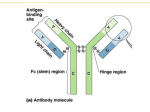
Evaluation of novel affinity bio-beads for use in the production of plasma derived albumin. Sergio Pagliazzi, Karl McCann, Jose Martinez, Tracy Thompson, Sara Ladd, Joseph Bertolini. CSL Behring, Camp Rd. Broadmeadows, Victoria PolyBatics, Ltd. BioCommerce Centre, Palmerston North, New Zealand Introduction Results The purification of albumin at CSL Behring from human plasma involves a combination of chromatographic steps with a final gel filtration polishing step used to purify material derived from ion exchange chromatography. This requires multiple cycles to process a batch and offers an opportunity to improve process efficiency. The aim of the project was to find an alternative to this process step. Polybind-Z Polybind-Z was able to remove up to 99% of IgG present in the albumin feedstock up to a loading a resin to feedstock ratio of 1:400, while for IgA and IgM was significantly lower. An affinity chromatography process involving the use of bio-beads, Polybind-Z and Polybind-L, manufactured by Polybatics, NZ was evaluated to remove the main residual proteins IgG, IgA and IgM. Bio-beads are produced in bacteria and consist of specific ligands incorporated onto a polyester backbone. The specific resins evaluated were Polybind-Z with a specific affinity for IgG, and Polybind-L, specific for the kappa light chain and able to interact with IgG, IgA and IgM. Structure of biobeads produced by Polybatics, NZ, showing ligands on the surface of the polyester scaffold Polybind-L Polybind-L had a broader affinity for immunoglobulins. It bound 70% of IgG and IgA at a low resin to volume ratio (1:10) with increasing drop through evident with further loading. Thus at a 1:400 resin to volume ratio, only 30% of IgG and IgA bound to resin, indicating a progressive saturation of binding sites. This resin had greater specificity and capacity for IgM with up to 70% removal at a resin to volume ratio of 1:200. Method Resins were tested to investigate their IgG, IgA and IgM binding capacity and specificity. Concentrated and pH adjusted albumin process intermediate was mixed with Polybind-Z or Polybind-L beads at various resin to feedstock ratios. One gram of resin was used in all studies. After thoroughly mixing the solution of resin and feedstock, the mixture was separated by centrifugation at 17,000x g. The supernatant was tested for unbound IgG, IgA and IgM. Application in bioprocessing Further process development would need to focus on establishing an appropriate means of removing the biobeads from the treated intermediate. As centrifugation is not practical in any large scale application, the use of depth filtration coupled with use of a filter aid has provided promising results in initial studies. Conclusions 1. The capability of two novel affinity resins (Polybind-Z and Polybind-L) produced by bacteria and incorporated with specific ligands for IgG, IgA and IgM was examined for the capability of removing these residual proteins an albumin intermediate containing these residual proteins. 2. Polybind-Z exhibited a high binding capacity and specificity for IgG, but low specificity for IgA and IgM when used to purify an albumin intermediate. Polybind-L had specificity and capacity for IgM but had lower capacity for IgA and IgG. 3. Biobeads with specific ligands have the capability of removing significant amounts of trace immunoglobulin contaminants from a predominantly albumin matrix and offer the potential to be used in an alternative low cost method for polishing an in-process albumin intermediate.