* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Formulation Development Services
Compounding wikipedia , lookup
Pharmacogenomics wikipedia , lookup
Neuropharmacology wikipedia , lookup
Pharmaceutical industry wikipedia , lookup
Prescription drug prices in the United States wikipedia , lookup
Prescription costs wikipedia , lookup
Drug interaction wikipedia , lookup
Pharmacognosy wikipedia , lookup
Theralizumab wikipedia , lookup
Drug design wikipedia , lookup
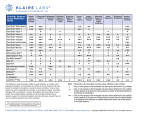
Formulation Development Not all test articles are freely soluble or readily bioavailable. Utilizing a combination of our formulation expertise and historical knowledge of vehicle suitability, as well as the close collaboration with our in vivo scientists, Charles River can ensure an informed lead candidate selection process and a formulation suitable for your preclinical programs. Formulation Development • pH solubility profiling • Dose suitability • Compatibility We offer a comprehensive package of formulation discovery and development activities in a vast array of formulation types. From preformulation investigations and screening to formulation validations, stability assessments, compatibility and assessment of suitability with dosing apparatus, our experience is unmatched. Extensive global Analytical Chemistry groups utilizing state-of-the-art instrumentation are available to support our formulation development services. Successful formulation development requires knowledge of the physicochemical characteristics of the drug. To achieve this goal, we carry out preformulation screening prior to formal formulation development experiments. Since ionizable entities in a drug molecule can significantly alter the solubility profile and the nature of its interaction with the cell membrane and the target site, it is important to assess the relative solubility of ionizable drug molecules at various pH levels. Data derived from such experiments can provide the basis for optimizing the final formulation, and optimum solubility can often facilitate higher drug absorption. pH Solubility Profile of Piroxicam Absorbance vs. pH of Piroxicam pH Absorbance (at 353 nm) Formulation B (400 mg/mL of Piroxicam, in 15% Labrasol in 0.1 Molar PBS pH 4.5) Formulation C (400 mg/mL of Piroxicam, in 15% Labrasol in 0.1 Molar PBS pH 1.2) 0.5 Formulation Types • Oral -- Solutions -- Suspensions -- Emulsions -- IV -- Intramuscular -- Intraperitoneal -- Subcutaneous • Topical -- Creams -- Ointments Formulation A (400 mg/mL of Piroxicam, in 15% Labrasol in 0.1 Molar PBS pH 6.8) 0.6 • Analytical chemistry • Parenteral pH Solubility Profiling 0.7 • Bioavailability enhancement • Inhalation -- Nebulizer solutions -- Powder blends 0.4 0.3 0.2 0.1 0 0 1 3 5 7 9 Time (hours) 11 13 [email protected] www.criver.com © 2014, Charles River Laboratories International, Inc. Dose Suitability Preclinical formulations, especially those used for oral dosing, are often formulated using high concentrations of test article. As a result, the physical characteristics of the formulation may not be readily predictable. It is important to assess the stability, ease of preparation, suitability and other physical characteristics of a formulation during dosing. This information adds confidence and aids in minimizing complications and delays in future preclinical studies. Compatibility Identifying the compatibility (or incompatibility) of a new drug molecule with the appropriate container and dosing apparatus is essential for developing a good experimental design. The identification and resolution of such issues as early as possible ensures a successful study with minimal time delays. Bioavailability Enhancement Poor drug solubility often correlates with poor bioavailability. The use of cosolvents, solubilizing excipients and biological enhancers can improve both solubility and drug absorption. However, a critical balance must exist between improving solubility and minimizing unnecessary side effects. By working closely with our Discovery and Toxicology in vivo scientists, formulations are optimized for improved results and minimal toxicity of formulation components. Potential Effect of Formulation Excipients on Bioavailability Plasma Concentration vs. Time 0.09 O Plasma concentration (μg/ml) 0.08 CH3 O N S 0.07 NH N 0.06 O 0.05 OH 0.04 0.03 0.02 0.01 0 0 5 10 15 Time (hours) 20 25 Analytical Chemistry Support The extensive experience and resources available within the Analytical Chemistry groups at Charles River serve as the backbone for validating and analyzing the produced formulations. It is essential to have reliable and robust analytical methods in the development stages of any preclinical study. Our Analytical Chemistry groups comprise scientific, technical and support staff with many years of experience and state-of-the-art instrumentation that can meet all your analytical needs. [email protected] www.criver.com © 2014, Charles River Laboratories International, Inc.