* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Fig. 11 - JACC: Cardiovascular Imaging
History of invasive and interventional cardiology wikipedia , lookup
Lutembacher's syndrome wikipedia , lookup
Hypertrophic cardiomyopathy wikipedia , lookup
Quantium Medical Cardiac Output wikipedia , lookup
Pericardial heart valves wikipedia , lookup
Marfan syndrome wikipedia , lookup
Turner syndrome wikipedia , lookup
Artificial heart valve wikipedia , lookup
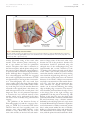
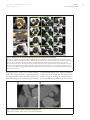
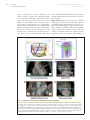
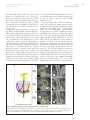
JACC: CARDIOVASCULAR IMAGING VOL. 6, NO. 2, 2013 © 2013 BY THE AMERICAN COLLEGE OF CARDIOLOGY FOUNDATION PUBLISHED BY ELSEVIER INC. ISSN 1936-878X/$36.00 http://dx.doi.org/10.1016/j.jcmg.2012.12.005 STATE-OF-THE-ART PAPER Standardized Imaging for Aortic Annular Sizing Implications for Transcatheter Valve Selection Albert M. Kasel, MD,* Salvatore Cassese, MD,* Sabine Bleiziffer, MD,† Makoto Amaki, MD, PHD,‡ Rebecca T. Hahn, MD,§ Adnan Kastrati, MD,* Partho P. Sengupta, MD‡ Munich, Germany; and New York, New York The safety and efficacy of transcatheter aortic valve replacement procedures are directly related to proper imaging. This report revisits the existing noninvasive and invasive approaches that have concurrently evolved to meet the demands for optimal selection and guidance of patients undergoing transcatheter aortic valve replacement. The authors summarize the published evidence and discuss the strengths and pitfalls of echocardiographic, computed tomographic, and calibrated aortic balloon valvuloplasty techniques in sizing the aortic valve annulus. Specific proposals for 3-dimensional tomographic reconstructions of complex 3-dimensional aortic root anatomy are provided for reducing intermodality variability in annular sizing. Finally, on the basis of the sizing approaches discussed in this review, the authors provide practical recommendations for balloonexpandable and self-expandable prostheses selection. Strategic use of echocardiographic, multislice computed tomographic, and angiographic data may provide complementary information for determining the anatomical suitability, efficacy, and safety of the procedure. (J Am Coll Cardiol Img 2013; 6:249 – 62) © 2013 by the American College of Cardiology Foundation T ranscatheter aortic valve replacement (TAVR) provides an alternative treatment option for inoperable and high– surgical risk patients with symptomatic severe aortic stenosis (1– 4). On the basis of evidence from a recent randomized trial (5,6), balloon-expandable prostheses (Edwards SAPIEN; Edwards Lifesciences, Irvine, California) received U.S. Food and Drug Administration approval for the U.S. market (7). Similarly, there is extensive evidence for self- expandable prostheses (CoreValve ReValving System; Medtronic, Inc., Minneapolis Minnesota), and randomized trials are expected to be completed in the next few years (3,4,8,9). The safety and efficacy of the TAVR procedure are directly related to proper imaging, which is based on patient selection and procedural guidance. However, as opposed to surgical aortic valve replacement, in which surgeons can directly observe the adaptation of the prosthesis to the aortic root while suturing, From the *Clinic for Cardiology and Cardiovascular Diseases, Deutsches Herzzentrum, Technische Universität, Munich, Germany; †Clinic for Cardiovascular Surgery, Deutsches Herzzentrum, Technische Universität, Munich, Germany; ‡The Zena and Michael A. Wiener Cardiovascular Institute, Mount Sinai School of Medicine, New York, New York; and §Columbia University Medical Center/New York Presbyterian Hospital, New York, New York. Dr. Kasel is a proctor for Edwards Lifesciences and Medtronic, Inc. Dr. Bleiziffer is a proctor for Medtronic and JenaValve, and is a steering committee member of the Engager Pivotal Study. All other authors have reported that they have no relationships relevant to the contents of this paper to disclose. Manuscript received November 26, 2012; revised manuscript received December 20, 2012, accepted December 21, 2012. 250 Kasel et al. Aortic Annular Sizing for TAVR TAVR does not permit the prediction of interaction between the sutureless transcatheter prostheses and surrounding structures before implantation. Therefore, an in-depth understanding of aortic root anatomy has become pivotal over the past few years (10). Thus, imaging techniques have garnered growing attention because they allow more precise measurements of the annulus and aortic root for proper definition of the spatial orientation of the aortic valve complex. This is important because these features guide the eligibility of patients for TAVR and allow adequate device sizing (11). Herein, we propose 3 techniques for sizing an aortic valve annulus before TAVR and discuss their implications for the currently available selection of prostheses. Although the exact utility and success of these approaches await prospective confirmation, the step-by-step approaches highlighted here should serve primarily as a guide for the methodological approaches available for sizing the aortic valve annulus for patients undergoing TAVR. ABBREVIATIONS AND ACRONYMS Studying the Aortic Valve Anatomy to Match it with an Adequately Sized Prosthesis MDCT ⴝ multidetector computed tomography TAVR ⴝ transcatheter aortic valve replacement TEE ⴝ transesophageal echocardiography In TAVR, “sizing” can be defined as the choice of prosthesis within a range of available sizes to ensure that it is best accommodated into the native aortic root. This sizing is dependent on the observation of anatomy-device interaction and represents one of the most important predictors of a successful procedure (3,4,12). During TAVR, the size of the aortic valve annulus is used as a standard measurement for quantitative assessment of the site of implantation. By definition, an annulus should be in the form of a ring. However, previous reports have questioned the concept of a distinct anatomic aortic annulus given the structure of the aortic root and semilunar-shaped cusps (10). Second, the close anatomical continuity of the left ventricular outflow tract into the ascending aorta precludes clear identification of the individual anatomic components (13). Arguably, the diameter of the aortic root varies considerably, and it depends primarily on the direction in which the diameter is measured. Piazza et al. (10) reported that the aortic root has a 3dimensional structure in which 3 main circular JACC: CARDIOVASCULAR IMAGING, VOL. 6, NO. 2, 2013 FEBRUARY 2013:249 – 62 rings, planes, and a crownlike ring can be recognized, and all of these originate from the aortic valve leaflets, which are attached throughout the length of the aortic root. The aortic valve annulus typically represents the tightest part of the aortic root and is defined as a virtual ring with 3 anatomical anchor points at the base of each of the attachments of the aortic leaflets (Fig. 1). The size of a transcatheter valve prosthesis traditionally relied on the dimensions of the aortic annulus during systole, with the native leaflets and their hinge points providing the first resistance and anchoring force to the prosthesis. The ability to provide the correct measurement of the aortic valve annulus is essential to avoid undersizing and oversizing of transcatheter heart valves. Undersizing of the aortic valve annulus could lead to the selection and deployment of a smaller prosthesis, which could result in paravalvular regurgitation (14) and valve embolization (15). In contrast, oversizing can lead to underexpansion of the prosthesis, with possible reduced valve durability, conduction disturbances leading to permanent pacemaker insertion, or annular rupture (16). In general, transcatheter prostheses are designed to be deployed in annuli that are slightly smaller than the prostheses. This “controlled” oversizing is essential for anchoring a sutureless prosthesis (11). Because of the particular characteristics of transcatheter prostheses, a complete assessment of the anatomy of the aortic valve complex is required during pre-procedural decision making. To date, 3 imaging techniques have been predominantly used to perform sizing before TAVR: echocardiography, multidetector computed tomography (MDCT) imaging, and intraoperative balloon sizing. Imaging Tools for Aortic Annular Sizing Transesophageal echocardiography (TEE). In clinical practice, patient eligibility and the determination of prosthesis size are currently based largely on echocardiography, which is an essential imaging tool for all patients undergoing TAVR (17,18). Given the increased understanding of the anatomy of the aortic valve, a single-dimensional measurement is no longer accepted as the sole determinant of transcatheter valve sizing. However, 2-dimensional transthoracic echocardiography is the first step to assess aortic valve stenosis JACC: CARDIOVASCULAR IMAGING, VOL. 6, NO. 2, 2013 FEBRUARY 2013:249 – 62 Kasel et al. Aortic Annular Sizing for TAVR Figure 1. Normal Anatomy of the Aortic Annulus The aortic annulus accounts for the tightest part of the aortic root (A) and is defined as a virtual ring (green line) with 3 anatomical anchor points at the nadir (green points) of each of the attachments of the 3 aortic leaflets (B). LCC ⫽ left coronary cusp; NCC ⫽ noncoronary cusp; RCC ⫽ right coronary cusp. severity and initial sizing of the aortic valve annulus. Three reference planes intersecting at 90° to one another are used for quantitative analysis. The plane of the valve is referred to as the transverse plane, and the planes orthogonal to the transverse plane define the sagittal (anteriorto-posterior) and coronal (left-to-right) reference planes. Although direct comparison of transthoracic echocardiography and TEE has suggested that systolic sagittal plane measurements on TEE are approximately 1 mm larger than on transthoracic echocardiography, this may result from image quality rather than from inherent differences in techniques (19,20). With either modality, in the long-axis view, the annular dimension measured on the sagittal plane is the shorter one, while that measured on the coronal plane is the longer one. Thus, it is of a paramount importance to ascertain the cross-sectional anatomy of the annulus, which can be performed in the shortaxis view or more accurately using 3-dimensional imaging. The guidelines of the American Society of Echocardiography (21) and the consensus document on TAVR (11) recommend TEE before TAVR if there are issues regarding aortic root anatomy, aortic valve annular diameter, or the number of cusps. With TEE, the size of the aortic valve annulus at the level of basal attach- ment or hinge points of the aortic valve cusps dictates the size of the prosthesis. Annular size measurement should be performed using the enlarged view of the midesophageal long axis (approximately 110° to 140°, referred to as the “3-chamber view”) during the early systolic phase of the cardiac cycle. In this projection, the left ventricular chamber, outflow tract, and ascending aorta should be aligned along their long axes to ensure that the sagittal plane bisects the maximal diameter of the annulus. Oblique measurements may lead to overestimation of the annular dimensions (Fig. 2A). Once the aforementioned anatomic structures are aligned, the aortic valve annulus can be measured following the trailing edge–to–leading edge convention. The measurement should be performed from the edge of sinus to the hinge point of the right coronary cusp perpendicular to the long axis of the aorta (Fig. 2B). Because in this long-axis view, the plane passes posteriorly between the commissure situated within left and noncoronary cusps, a distinct anatomic landmark (such as the hinge point of a cusp) cannot be clearly delineated. Proper attention is required to avoid measurement of bulky commissural calcium that is often present within the sinus of Valsalva, leading to overestimation of the actual annular size. The measurement must exclude ectopic calcifications, with the points measured outside the calcifi- 251 252 Kasel et al. Aortic Annular Sizing for TAVR Figure 2. Aortic Annular View With Transesophageal Echocardiography Midesophageal long-axis zoomed-up view (“3-chamber view”) during the early systolic phase of the cardiac cycle (A). The left ventricular chamber (LV), the outflow tract (OT), the aortic valve (AV), and the ascending aorta (AA) are aligned. An orthogonal plane is considered as a reference (B, dotted yellow line). The 4 edges of the aortic sinus (yellow points) are the landmarks: the sinotubular junction defines the upper reference plane, and the insertion point of the aortic valve leaflets on the outflow tract defines the lower reference plane. The aortic valve annulus is measured as the distance between the hinge point of the right cusp and the edge of the sinus at the side of the commissures between the left and noncoronary cusps, including calcifications (red line with arrowheads). It is useful to select the landmarks by moving frames in cine loop back and forth to rule out artifacts (C). JACC: CARDIOVASCULAR IMAGING, VOL. 6, NO. 2, 2013 FEBRUARY 2013:249 – 62 cations. In some cases, real-time imaging may help distinguish side-lobe and reverberation artifacts from real structures (Fig. 2C). Although measurements from 2 to 5 beats can be averaged, it is important to remember that cyclic variation in the location of the sagittal plane occurs with respirations or within the RR cycle. An enlarged short-axis view on TEE illustrates the aortic valve opening and location and extent of calcifications. The new generation of ultrasound scanners offers multiplanar imaging with simultaneous acquisition of short-axis and long-axis planes, a feature that could be helpful for finding a plane that bisects the annulus, especially when patients are examined with limited echocardiographic windows. Two-dimensional TEE possesses the inaccuracy observed in monoplanar displays. Furthermore, 2-dimensional TEE has some specific drawbacks due to the unique measurements required for TAVR. Although the operator can obtain a symmetric visualization of the cusps, their measurement could prove challenging (20,22,23). Although 3-dimensional TEE lacks adequate standardization and is not yet routinely available, there is some evidence suggesting that this method is a valid alternative for more precise pre-procedural measurements in the setting of TAVR, potentially reducing the possibility of sizing errors (24 –27). We propose a 3-step approach, referred to as the “turnaround rule,” to easily define aortic valve annular size (Fig. 3). Two-dimensional TEE is performed to obtain a short-axis view of the aortic valve. Three-dimensional TEE is then performed over zoom mode to acquire loops with the narrowest possible depth, with adjustment of lateral width and elevation width for obtaining a volume containing the whole aortic root, the left ventricular outflow tract, and part of the ascending aorta; volume rates of at least 10/s will allow an accurate assessment of early systole. The loop is then assessed with a 3-dimensional commercially available software package with standard short-axis views of the aortic valve (Fig. 4) in mid systole used as a reference frame. First, the transverse and sagittal and coronal orthogonal planes are oriented along the aortic root such that all planes intersect at the center of the opened valve, with the sagittal and coronal planes aligned parallel to the long axis of the ascending aorta. Second, the orthogonal planes are rotated to identify the most caudal attachments of the aortic valve leaflets (hinge points). It is important to remember that the Kasel et al. Aortic Annular Sizing for TAVR JACC: CARDIOVASCULAR IMAGING, VOL. 6, NO. 2, 2013 FEBRUARY 2013:249 – 62 Figure 3. Alignment of Aortic Root Planes With 3-Dimensional Transesophageal Echocardiography The colors used through selected images (lines and planes) reflect the 3-dimensional schematic reconstructions. The center axis of the left ventricular outflow tract (LVOT) is chosen as a reference with the transverse plane (1) placed parallel to the sinotubular junction (red dots). The planes orthogonal to the transverse plane are rotated (2) for delineating the hinge point of the left and right coronary sinuses. The transverse plane is moved toward the left ventricular outflow (3) to arrive at the level of the hinge points of the aortic leaflet insertion. The orthogonal plane is further rotated (4) to ensure that the transverse plane also crosses through the hinge point of the noncoronary cusp (NCC). The longest (D1) and the shortest (D2) diameters, the circumference, and the area of the annulus are measured. Abbreviations as in Figure 1. hinge-point plane refers to the virtual annular plane. The transverse plane is repositioned from the aorta toward the ventricle until it reaches the level of the hinge points. In the setting of severely calcified and immobile cusps, bulky calcifications of the cusps may extend into the plane of the annulus in systole, making an accurate measurement more difficult. In addition, acoustic artifacts Figure 4. Artifacts With Computed Tomographic Angiographic Scans Image distortion caused by motion artifacts is depicted (A, B, red arrows). 253 254 Kasel et al. Aortic Annular Sizing for TAVR such as side lobes or acoustic shadowing may reduce accuracy. Finally, the orthogonal planes are repeatedly rotated (the turnaround rule) to ensure that the hinge points of the aortic valve leaflets are transected by the transverse plane. The annulus is typically oval in its appearance, with the minimal dimension in the sagittal plane and the maximal dimension in the coronal plane. These measurements, as well as the perimeter and annular area, can then be measured on the transverse plane. A mean annular diameter (the mean JACC: CARDIOVASCULAR IMAGING, VOL. 6, NO. 2, 2013 FEBRUARY 2013:249 – 62 of the minimal and maximal diameters) or a mean annular diameter (from the perimeter or area) can be calculated. MDCT imaging. Over the past few years, MDCT imaging has become an essential tool for providing detailed and reliable description of the complex 3-dimensional aortic root anatomy in patients undergoing TAVR. Indeed, tomographic images of the aortic annulus, commissures, and sinuses of Valsalva, where the coronary arteries originate, provide an in-depth understanding of Figure 5. Alignment of Aortic Root Planes With Multislice Computed Tomography The colors used through selected computed tomography images (lines and contours) reflect the 3-dimensional schematic reconstructions (top). The center axis of the left ventricular outflow tract and ascending aorta is chosen as a reference with the 3 planes locked in a 90° angle. The crosshair are moved in the middle of the aortic root to align the longitudinal axis in coronal and sagittal planes (A, B, bottom, orange/blue lines). The transverse plane is aligned at the level of the valve (C, valve plane), dragging this plane down from the aorta towards the ventricle (A, B, bottom, white arrows) until the most caudal attachment of the aortic valve leaflets (hinge points) come into view (D, transverse plane). The transverse plane is the basis for the alignment of the hinge point plane which refers to the virtual annulus plane in which no valve structure is visible. A ⫽ anterior; L ⫽ left; P ⫽ posterior; R ⫽ right. Kasel et al. Aortic Annular Sizing for TAVR JACC: CARDIOVASCULAR IMAGING, VOL. 6, NO. 2, 2013 FEBRUARY 2013:249 – 62 the framework within which the aortic valve leaflets are suspended (20,28 –31). These preoperative assessments are required because of the lack of direct valve visualization when the procedure is performed. Consensus regarding TAVR recommends the use of MDCT systems with at least 64 detectors and spatial resolution of 0.5 to 0.6 mm (11). Suggested scanning protocols for annular sizing during TAVR include electrocardiographically synchronized (gated) imaging of the aortic root, which is important to avoid motion-induced artifacts (Figs. 4A and 4B). Image reconstruction can be performed in the desired phase of the cardiac cycle, and in contrast to transesophageal echocardiographic measurements, MDCT imaging does not report any significant differences with respect to diameters, which are acquired during the systolic or diastolic phase (28). We recommend using the phase of the cardiac cycle with the best image quality. Reconstruction of the annulus should be performed orthogonally in relation to the central axis of the left ventricular outflow tract; this permits correct understanding of minimal and maximal diameter, circumfer- ence, and area, while emphasizing the presence of a noncircular aortic valve annulus (reported in up to 40% of patients who may need TAVR) (20,24,30,32,33). The turnaround rule is useful to define the aortic valve annular size with the help of simple multiplanar rendering software (with the 3 planes locked at a 90° angle), and it is integrated in almost all of the workstations used for cardiac imaging. First, the crosshairs should be moved to the middle of the aortic root, and the longitudinal axis in the coronal and sagittal plane should be aligned (double oblique planes) (Figs. 5A and 5B). Second, the transverse plane should be aligned at the level of the valve (valve plane) by dragging this plane down from the aorta toward the ventricle until the most caudal attachments of the aortic valve leaflets (hinge points) come into view (Figs. 5C and 5D). Finally, the transverse plane should be rotated around its own axis to align the longitudinal planes (coronal oblique and sagittal oblique planes) to ensure that the hinge points of the aortic valve leaflets individually touch the transverse plane (Figs. 6A to 6D). Ideally, when the orientation of the transverse Figure 6. Spatial Reconstruction of the Aortic Annulus With the “Turnaround Rule” Scheme (A) and computed tomographic images (B to D): the transverse plane is turned clockwise around its own axis to align one at a time the nadirs of the aortic valve leaflets with the transverse plane in 3 steps (1–3). The distances to the coronary ostia can be measured in the longitudinal axis at a right angle to the hinge point plane (B, C, red arrows). Abbreviations as in Figure 1. 255 256 Kasel et al. Aortic Annular Sizing for TAVR plane is correct, all 3 aortic valve leaflets appear or disappear simultaneously by moving this plane in a caudal or cranial fashion. If the planes need to be realigned to minutely adjust the transversal plane, it should be ensured that all 3 cusps touch the plane again while the transverse plane is turned around 1 more time. Once the hinge point plane is aligned, the measurement can be performed in this plane or 1 section below (in the upper outflow tract) because of the excellent spatial resolution associated with previous generation of MDCT systems. However, it must be remembered that in the current setting of clinical practice, although imaging refinements are available, the proper identification and alignment of the plane on which the virtual ring is situated might be difficult because of heavy calcifications or extremely oval annuli. Both of these issues could lead to distortion of the aortic root. In these cases, patients would benefit most from multimodality imaging (20,22,34,35). JACC: CARDIOVASCULAR IMAGING, VOL. 6, NO. 2, 2013 FEBRUARY 2013:249 – 62 Once alignment has been performed, it is possible to trace a polygonal line that circumscribes the aortic valve annulus (visible at the transverse plane level) to determine its area and perimeter. Minimal and maximal dimensions can be manually determined through the center point of this annulus, with the mean diameter being the mean of these 2 measures or calculated using the area or circumference (Fig. 7). Bioprostheses are supposed to have a circular cross-section. In this respect, the diameter derived from this ideal circumference is calculated as: (perimeter of the traced polygon)/, while the area is calculated as: [2 ⫻ ✓(area of traced polygon in mm2/)] (36,37). The distance of the valve plane from the coronary ostia should be measured in the longitudinal axis at right angle to the hinge point plane (Figs. 6B and 6C). The importance of MDCT measurements performed at the transverse plane level and the potential drawbacks associated with erroneous measures are described in Figure 8. Figure 7. Aortic Annular Measurements at Hinge-Point Plane (A) Minimal and (B) maximal dimensions are drawn manually through the center point of the aortic valve annulus. Kasel et al. Aortic Annular Sizing for TAVR JACC: CARDIOVASCULAR IMAGING, VOL. 6, NO. 2, 2013 FEBRUARY 2013:249 – 62 Figure 8. Errors During Measurements at the Level of the Aortic Root False measurements, as well as correct aortic annular definition at the hinge-point plane, are illustrated. Several lines of evidence have demonstrated that MDCT measurements of the virtual basal ring (mean diameter, circumference, and area) are more reproducible than echocardiography (20,29,33). The use of MDCT measurements could result in changes in strategy in a greater number of patients compared with TEE (20) because of its close correlation with direct surgical measures (38,39) and a high degree of reproducibility (37). Annular sizing and the assessment of aortic root orientation would allow the prediction of the aortic root angle before the procedure (40,41). This might potentially decrease the number of aortograms required during the procedure and therefore reduce the procedure time and need for contrast, thereby improving the precision of bioprostheses deployment (40). Calibrated balloon aortic valvuloplasty. Although there have been continual advances in imaging techniques, the characteristics of degenerative aortic valve stenosis can lead to very challenging scenarios in the setting of TAVR. Furthermore, because of conflicting measurements obtained with multimodal imaging, annular sizing using 2 different prosthesis sizes, asymmetric calcifications, or eccentric leaflets, there can be uncertainties regarding the selection of an optimal prosthesis for TAVR (20,42). In those cases, balloon aortic valvuloplasty (43) after proper calibration (44 – 46) could be used as a tool for direct annular sizing and correct prosthesis selection in the Figure 9. Aortic Annular Sizing Through Balloon Valvuloplasty and Simultaneous Aortography Indirect signs of proper measurements: the lack of movement of the balloon within the aortic valve (yellow line with arrowhead), the possible waist of the balloon at the level of the annulus (red arrows), the residual contrast medium regurgitation between the maximally inflated balloon and the hinge points of the valve (blue arrow), and the calcified leaflets splayed against coronary ostia (green arrow). 257 258 Kasel et al. Aortic Annular Sizing for TAVR JACC: CARDIOVASCULAR IMAGING, VOL. 6, NO. 2, 2013 Figure 10. Recommendations for Balloon-Expandable and Self-Expandable Prosthesis Selection Commercially available prostheses are presented (Edwards SAPIEN [A] and SAPIEN XT [B]. FEBRUARY 2013:249 – 62 JACC: CARDIOVASCULAR IMAGING, VOL. 6, NO. 2, 2013 FEBRUARY 2013:249 – 62 Kasel et al. Aortic Annular Sizing for TAVR Figure 10. Continued Edwards Lifesciences; CoreValve ReValving System [C], Medtronic, Inc.) with ideal measurements and requested oversizing. For Edwards SAPIEN and SAPIEN XT prostheses, the 23-mm and 25-mm balloons provided from the manufacturer can be used for sizing. AsAo ⫽ ascending aorta; BAV ⫽ balloon aortic valvuloplasty; CoV ⫽ CoreValve; CT ⫽ computed tomography; MD ⫽ mean diameter; SXT ⫽ Edwards SAPIENT XT; TEE ⫽ transesophageal echocardiography. aRange provided by the manufacturer. bRange suggested according to the authors’ personal experience. cComputed tomographic confirmation is required. catheterization laboratory. For instance, previous reports have suggested that balloon aortic valvuloplasty could directly influence the change in the size of the prosthesis in up to 25% of patients (45). As illustrated in Figure 9, during rapid pacing, balloon valvuloplasty performed simultaneously with contrast injection at the level of the ascend- ing aorta can provide an indirect confirmation of adequate sizing through several signs: first, the lack of balloon movement within the aortic valve; second, the waist of the balloon present at the level of annulus; and third, the absence of regurgitation of the residual contrast medium between the maximally inflated balloon and hinge points of the valve. Moreover, during balloon valvulo- Figure 11. Annular Calcifications and Bioprosthesis Oversizing Annulus without calcification (A), annulus with moderate calcifications (B), and annulus with severe or diffuse calcifications (C). 259 260 Kasel et al. Aortic Annular Sizing for TAVR JACC: CARDIOVASCULAR IMAGING, VOL. 6, NO. 2, 2013 FEBRUARY 2013:249 – 62 plasty, operators can reliably predict the final position of the splayed calcified leaflets to exclude the potential risk for coronary ostia occlusion after TAVR. Prosthesis Selection after Aortic Annular Sizing Once definitive measurements have been made, the prosthesis with the correct size that adequately fits the annular diameter should be selected on the basis of charts provided by manufacturers and one’s own experience (Figs. 10A to 10C). Because balloon-expandable valves are designed to reach a definite diameter and form after deployment, they can change the shape of the annulus remarkably. In contrast, self-expandable prostheses are more prone to accommodate native anatomies. Thus, for TAVR, especially with balloon-expandable prostheses, procedural success and inherent risks are highly dependent on proper annular sizing. In this regard, it must be acknowledged that balloon-expandable transcatheter prostheses (Edwards SAPIEN, available only in the United States, and SAPIEN XT, available only in Europe) are available in 3-mm step sizes (namely, 23 and 26 mm for the Edwards SAPIEN valve and 23, 26, and 29 mm for the SAPIEN XT valve), while self-expandable transcatheter prostheses are available in 3-mm step sizes up to 29 mm, with a newly available 2-mm step size of 31 mm. This aspect could represent a problem when measurements of the aortic annulus are in the overlapping area between 2 different prosthesis sizes. In such cases, attention must be paid to the extent of calcification of the leaflets to avoid the risk for aortic root rupture. In cases of annuli without calcification (Fig. 11A), the valve size should be at least ⱖ1 REFERENCES 1. Webb JG, Wood DA. Current status of transcatheter aortic valve replacement. J Am Coll Cardiol 2012;60: 483–92. 2. Genereux P, Head SJ, Van Mieghem NM, et al. Clinical outcomes after transcatheter aortic valve replacement using valve academic research consortium definitions: a weighted meta-analysis of 3,519 patients from 16 studies. J Am Coll Cardiol 2012; 59:2317–26. 3. Genereux P, Head SJ, Wood DA, et al. Transcatheter aortic valve implantation 10-year anniversary: re- mm bigger than the annular size. However, in cases of moderate calcification (Fig. 11B), the valve size should be ⱖ0.5 mm bigger than annular size, and for severe or diffuse calcification (Fig. 11C), the chosen valve size could be nearly equal to the annular size. With the currently available bioprostheses, up to 97% of patients are anatomically suitable for TAVR, suggesting that a wide array of bioprostheses will permit the management of patients with severe aortic stenosis who are at high or prohibitive risk for surgery (47– 49). Conclusions As technology and imaging rapidly evolve, refinements to TAVR procedures are expected to diminish the uncertainty that still surrounds their widespread application, potentially leading to an increase in frequency resembling that seen with percutaneous coronary intervention over the past 2 decades. As the procedures become simpler and safer, it is more likely that they will be performed more often. Therefore, clear and easily available 3-dimensional imaging and device ameliorations, in addition to increasing the experience of operators, will play a pivotal role for TAVR, especially in the setting of patient eligibility and prosthesis selection. The overall aim is to avoid the serious consequences due to undersizing or oversizing in this clinical scenario. Reprint requests and correspondence: Dr. Albert M. Kasel, Deutsches Herzzentrum, Technische Universität, Lazarettstrasse 36, Munich, Germany. E-mail: markus.kasel@ googlemail.com. OR Dr. Partho P. Sengupta, Mount Sinai School of Medicine, One Gustave L. Levy Place, Box 1030, New York, New York 10029. E-mail: partho. [email protected]. view of current evidence and clinical implications. Eur Heart J 2012;33: 2388 –98. 4. Genereux P, Head SJ, Wood DA, et al. Transcatheter aortic valve implantation: 10-year anniversary. Part II: clinical implications. Eur Heart J 2012;33:2399 – 402. 5. Leon MB, Smith CR, Mack M, et al. Transcatheter aortic-valve implantation for aortic stenosis in patients who cannot undergo surgery. N Engl J Med 2010;363:1597– 607. 6. Smith CR, Leon MB, Mack MJ, et al. Transcatheter versus surgical aortic-valve replacement in high- risk patients. N Engl J Med 2011; 364:2187–98. 7. U.S. Food and Drug Administration. Edwards SAPIEN Transcatheter Heart Valve (THV)-P100041. Available at: http://www.fda.gov/ MedicalDevices/ProductsandMedical Procedures/DeviceApprovalsand Clearances/Recently-Approved Devices/ucm280840.htm. Accessed January 4, 2013. 8. Buellesfeld L, Gerckens U, Schuler G, et al. 2-year follow-up of patients undergoing transcatheter aortic valve implantation using a self-expanding valve prosthesis. J Am Coll Cardiol 2011;57:1650 –7. JACC: CARDIOVASCULAR IMAGING, VOL. 6, NO. 2, 2013 FEBRUARY 2013:249 – 62 9. Ussia GP, Barbanti M, Petronio AS, et al. Transcatheter aortic valve implantation: 3-year outcomes of selfexpanding CoreValve prosthesis. Eur Heart J 2012;33:969 –76. 10. Piazza N, de Jaegere P, Schultz C, Becker AE, Serruys PW, Anderson RH. Anatomy of the aortic valvar complex and its implications for transcatheter implantation of the aortic valve. Circ Cardiovasc Interv 2008;1:74 – 81. 11. Holmes DR Jr, Mack MJ, Kaul S, et al. 2012 ACCF/AATS/SCAI/STS expert consensus document on transcatheter aortic valve replacement. J Am Coll Cardiol 2012;59:1200 –54. 12. Jilaihawi H, Chin D, Spyt T, et al. Prosthesis-patient mismatch after transcatheter aortic valve implantation with the Medtronic-CoreValve bioprosthesis. Eur Heart J 2010;31: 857– 64. 13. Anderson RH. Clinical anatomy of the aortic root. Heart 2000;84:670 –3. 14. Detaint D, Lepage L, Himbert D, et al. Determinants of significant paravalvular regurgitation after transcatheter aortic valve: implantation impact of device and annulus discongruence. J Am Coll Cardiol Intv 2009;2:821–7. 15. Tay EL, Gurvitch R, Wijeysinghe N, et al. Outcome of patients after transcatheter aortic valve embolization. J Am Coll Cardiol Intv 2011;4:228 –34. 16. Blanke P, Reinohl J, Schlensak C, et al. Prosthesis oversizing in balloonexpandable transcatheter aortic valve implantation is associated with contained rupture of the aortic root. Circ Cardiovasc Interv 2012;5:540 – 8. 17. Vahanian A, Alfieri O, Al-Attar N, et al. Transcatheter valve implantation for patients with aortic stenosis: a position statement from the European Association of Cardio-Thoracic Surgery (EACTS) and the European Society of Cardiology (ESC), in collaboration with the European Association of Percutaneous Cardiovascular Interventions (EAPCI). Eur Heart J 2008;29:1463–70. 18. Jayasuriya C, Moss RR, Munt B. Transcatheter aortic valve implantation in aortic stenosis: the role of echocardiography. J Am Soc Echocardiogr 2011;24:15–27. 19. Moss RR, Ivens E, Pasupati S, et al. Role of echocardiography in percutaneous aortic valve implantation. J Am Coll Cardiol Img 2008;1:15–24. 20. Messika-Zeitoun D, Serfaty JM, Brochet E, et al. Multimodal assessment of the aortic annulus diameter: implications for transcatheter aortic valve implantation. J Am Coll Cardiol 2010;55:186 –94. 21. Zamorano JL, Badano LP, Bruce C, et al. EAE/ASE recommendations for the use of echocardiography in new transcatheter interventions for valvular heart disease. J Am Soc Echocardiogr 2011;24:937– 65. 22. Tuzcu EM, Kapadia SR, Schoenhagen P. Multimodality quantitative imaging of aortic root for transcatheter aortic valve implantation: more complex than it appears. J Am Coll Cardiol 2010;55:195–7. 23. Blanke P, Siepe M, Reinohl J, et al. Assessment of aortic annulus dimensions for Edwards SAPIEN Transapical Heart Valve implantation by computed tomography: calculating average diameter using a virtual ring method. Eur J Cardiothorac Surg 2010;38: 750 – 8. 24. Ng AC, Delgado V, van der Kley F, et al. Comparison of aortic root dimensions and geometries before and after transcatheter aortic valve implantation by 2- and 3-dimensional transesophageal echocardiography and multislice computed tomography. Circ Cardiovasc Imaging 2010;3:94 –102. 25. Altiok E, Koos R, Schroder J, et al. Comparison of two-dimensional and three-dimensional imaging techniques for measurement of aortic annulus diameters before transcatheter aortic valve implantation. Heart 2011;97: 1578 – 84. 26. Santos N, de Agustin JA, Almeria C, et al. Prosthesis/annulus discongruence assessed by three-dimensional transoesophageal echocardiography: a predictor of significant paravalvular aortic regurgitation after transcatheter aortic valve implantation. Eur Heart J Cardiovasc Imaging 2012;13:931–7. 27. Janosi RA, Kahlert P, Plicht B, et al. Measurement of the aortic annulus size by real-time three-dimensional transesophageal echocardiography. Minim Invasive Ther Allied Technol 2011;20:85–94. 28. Tops LF, Wood DA, Delgado V, et al. Noninvasive evaluation of the aortic root with multislice computed tomography implications for transcatheter aortic valve replacement. J Am Coll Cardiol Img 2008;1:321–30. 29. Schoenhagen P, Tuzcu EM, Kapadia SR, Desai MY, Svensson LG. Threedimensional imaging of the aortic valve and aortic root with computed tomography: new standards in an era of transcatheter valve repair/implantation. Eur Heart J 2009;30:2079 – 86. 30. Akhtar M, Tuzcu EM, Kapadia SR, et al. Aortic root morphology in patients undergoing percutaneous aortic valve replacement: evidence of aortic root remodeling. The Journal of thoracic and cardiovascular surgery 2009; 137:950 – 6. 31. Schoenhagen P, Numburi U, Halliburton SS, et al. Three-dimensional Kasel et al. Aortic Annular Sizing for TAVR imaging in the context of minimally invasive and transcatheter cardiovascular interventions using multi-detector computed tomography: from pre-operative planning to intra-operative guidance. Eur Heart J 2010;31:2727– 40. 32. Leipsic J, Gurvitch R, Labounty TM, et al. Multidetector computed tomography in transcatheter aortic valve implantation. J Am Coll Cardiol Img 2011;4:416 –29. 33. O’Brien B, Schoenhagen P, Kapadia SR, et al. Integration of 3D imaging data in the assessment of aortic stenosis: impact on classification of disease severity. Circ Cardiovasc Imaging 2011;4:566 –73. 34. Delgado V, Kapadia S, Schalij MJ, Schuijf JD, Tuzcu EM, Bax JJ. Transcatheter aortic valve implantation: implications of multimodality imaging in patient selection, procedural guidance, and outcomes. Heart 2012;98:743–54. 35. Bloomfield GS, Gillam LD, Hahn RT, et al. A practical guide to multimodality imaging of transcatheter aortic valve replacement. J Am Coll Cardiol Img 2012;5:441–55. 36. Schultz CJ, Moelker A, Piazza N, et al. Three dimensional evaluation of the aortic annulus using multislice computer tomography: are manufacturer’s guidelines for sizing for percutaneous aortic valve replacement helpful? Eur Heart J 2010;31:849 –56. 37. Jilaihawi H, Kashif M, Fontana G, et al. Cross-sectional computed tomographic assessment improves accuracy of aortic annular sizing for transcatheter aortic valve replacement and reduces the incidence of paravalvular aortic regurgitation. J Am Coll Cardiol 2012;59:1275– 86. 38. Dashkevich A, Blanke P, Siepe M, et al. Preoperative assessment of aortic annulus dimensions: comparison of noninvasive and intraoperative measurement. Ann Thorac Surg 2011;91: 709 –14. 39. Kempfert J, Van Linden A, Lehmkuhl L, et al. Aortic annulus sizing: echocardiographic vs. computed tomography derived measurements in comparison with direct surgical sizing. Eur J Cardiothorac Surg 2012;42:627–33. 40. Gurvitch R, Wood DA, Leipsic J, et al. Multislice computed tomography for prediction of optimal angiographic deployment projections during transcatheter aortic valve implantation. J Am Coll Cardiol Intv 2010;3:1157– 65. 41. Kurra V, Kapadia SR, Tuzcu EM, et al. Pre-procedural imaging of aortic root orientation and dimensions: comparison between x-ray angiographic planar imaging and 3-dimensional multidetector row computed tomography. J Am Coll Cardiol Intv 2010;3:105–13. 261 262 Kasel et al. Aortic Annular Sizing for TAVR 42. Cerillo AG, Mariani M, Glauber M, Berti S. Sizing the annulus for transcatheter aortic valve implantation: more than a simple measure? Eur J Cardiothorac Surg 2012;41:717– 8. 43. Cribier A, Savin T, Berland J, et al. Percutaneous transluminal balloon valvuloplasty of adult aortic stenosis: report of 92 cases. J Am Coll Cardiol 1987;9:381– 6. 44. Babaliaros VC, Liff D, Chen EP, et al. Can balloon aortic valvuloplasty help determine appropriate transcatheter aortic valve size? J Am Coll Cardiol Intv 2008;1:580 – 6. 45. Babaliaros VC, Junagadhwalla Z, Lerakis S, et al. Use of balloon aortic valvuloplasty to size the aortic annulus JACC: CARDIOVASCULAR IMAGING, VOL. 6, NO. 2, 2013 FEBRUARY 2013:249 – 62 before implantation of a balloonexpandable transcatheter heart valve. J Am Coll Cardiol Intv 2010;3:114 – 8. 46. Cerillo AG, Mariani M, Berti S, Glauber M. Sizing the aortic annulus. Ann Cardiothorac Surg 2012;1:245–56. 47. Jilaihawi H, Bonan R, Asgar A, et al. Anatomic suitability for present and next generation transcatheter aortic valve prostheses: evidence for a complementary multidevice approach to treatment. J Am Coll Cardiol Intv 2010;3:859 – 66. 48. Jilaihawi H, Chakravarty T, Weiss RE, Fontana GP, Forrester J, Makkar RR. Meta-analysis of complications in aortic valve replacement: comparison of Medtronic-CoreValve, Edwards-SAPIEN and surgical aortic valve replacement in 8,536 patients. Catheter Cardiovasc Interv 2012;80: 128 –38. 49. MacDonald I, Pasupati S. Transcatheter aortic valve implantation: know the differences between the currently available technologies. Eur Heart J 2010;31:1663–5. Key Words: aortic stenosis y computed tomography y echocardiography y patient selection y transcatheter aortic valve implantation.