* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Human Herpesviruses Lecture Objectives
Survey
Document related concepts
Viral phylodynamics wikipedia , lookup
Public health genomics wikipedia , lookup
Hygiene hypothesis wikipedia , lookup
Focal infection theory wikipedia , lookup
Transmission (medicine) wikipedia , lookup
Infection control wikipedia , lookup
Gene therapy of the human retina wikipedia , lookup
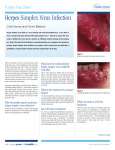
Canine distemper wikipedia , lookup
Canine parvovirus wikipedia , lookup
Marburg virus disease wikipedia , lookup
Henipavirus wikipedia , lookup
Vectors in gene therapy wikipedia , lookup
Transcript
Human Herpesviruses Jackie Parker, Ph.D. Department of Pediatrics, Infectious Diseases CHB 118B, 1600 6th Avenue South (behind Children’s Hospital) 996-7881 (office) 996-7875 (lab) [email protected] Lecture Objectives • Introduction to the Human herpesviruses • Virus replication • Human herpesviruses Herpes Simplex Viruses VZV EBV CMV HHV6,7,8 Simian B Virus *Clinical manifestations *Antiviral treatment *Disease in the Immunocompromised host 1 Herpesviruses: History • Herpes infections have been described since ancient Greece • Name derived from the Greek word “herpein”, which means “to creep” • Hippocrates and other Greek scholars used the word “herpes” to describe spreading cutaneous lesions caused by many types of herpesviruses Herpesviruses: General Features Family Herpesviridae Subfamilies Alpha, Beta, Gamma herpesvirinae Size 180-200 nm Envelope Yes Genome linear ds DNA ranges from ~120 kb (VZV) to ~230 kb (CMV) Genome Replicated Virus Assembly nucleus nucleus 2 Herpesviruses: Structural Components Viral glycoproteins, found embedded within the lipid envelope, are responsible for several functions, including receptor-mediated cellular entry. The tegument, a layer of proteins between the envelope and the underlying capsid. Tegument proteins are responsible for induction of viral gene expression and shutoff of host protein synthesis immediately following infection, in addition to virion assembly functions. Icosadeltahedral capsid dsDNA core Herpesviridae Subfamilies Alphaherpesvirinae (HSV-1, HSV-2, VZV) -variable host range -short reproductive cycle; rapid spread in culture -capacity to establish latent infections primarily (but not exclusively) in sensory ganglions Betaherpesvirinae (CMV, HHV-6) -restricted host range -long reproductive cycle; slow infection in culture -latent virus maintained in secretory glands, lymphoreticular cells, kidneys, and other tissues Gammaherpesvirinae (EBV) -members replicate in lymphoblastoid cells -latent or lytic infection, but without production of infectious progeny 3 Gene organization among representative herpesviruses (HSV) α (VZV) (HCMV) β (HHV-6B) γ (EBV) (HHV-8) Properties of Human Herpesviruses Common Name (Designation) Target cell type Latency Disease HSV-1 (HHV-1) Mucoepithelia Neuron Oral, ocular lesions; HSV-2 (HHV-2) Mucoepithelia Neuron Genital, ocular, disseminated Varicella Zoster virus, VZV (HHV-3) Mucoepithelia Neuron Chicken pox, shingles Epstein-Barr virus, EBV (HHV-4) B lymphocyte, epithelia B lymphocytes Infectious mononucleosis, Cytomegalovirus (CMV) Epithelia, monocytes, lymphocytes Monocytes, lymphocytes, dendritic cells numerous (HHV-5) Herpes lymphotrophic virus (HHV-6) T lymphocytes and others T lymphocytes and others Roseola in infants HHV-7 T lymphocytes and others T lymphocytes and others Some cases of roseola, possibly Pityriasis rosea HHV-8. Kaposi’s sarcoma associated virus (KHSV) B cells, Endothelial cells B lymphocytes, others? Tumors, Kaposi’s sarcoma, some B cell tumors Simian B virus (Herpes B) Mucoepithelia Neuron* encephalitis encephalitis, etc. tumors 4 HHV Infections in the Immunocompromised Patient In immune competent patients any clinical illness tends to be mild and self-limiting. HHVs cause severe and atypical clinical illness in immunocompromised patients. Patients may be immunocompromised for many different reasons, most of which are acquired rather than congenital. These include: Iatrogenic - the consequence of prescribed drugs such as corticosteroids or other immunosuppressants. Cancer - often associated with dysregulation of the immune system. Infection - patients with T-cell immunodeficiency (e.g. AIDS) generally have more severe and persistent HHV infections. It is also important to remember that the immune system changes through life: Neonates have an immature immune system and are particularly susceptible to severe HHV primary infection that may be fatal. Primary HHV infection in pregnancy can be severe • HHV infection of the developing fetus can cause severe congenital abnormalities associated with mortality and high morbidity. • Pregnant women are at increased risk of developing severe primary HHV infection that can be fatal. The immune system becomes less efficient with increasing age. • In particular, T-lymphocyte immunity is less effective in old age. Alpha herpesviruses • Herpes simplex virus type 1 (HSV-1) • Herpes simplex virus type 2 (HSV-2) • Varicella Zoster virus (VZV) 5 Herpes Simplex Viruses (HSV-1, HSV-2) • HSV-1 and HSV-2 are neurotrophic alpha-herpesvirus that only naturally occur in humans. There are no animal reservoirs. • Significant differences between genomes of HSV-1 and HSV-2, but both cause similar clinical illnesses • HSV-1 mostly infects oral mucosa, but can infect genital mucosa, whereas HSV-2 mostly infects genital mucosa, but can infect oral mucosa • HSV-1 seroprevalence exceeds 90%, whereas HSV-2 seroprevalence is ~18%. Herpes Simplex Viruses, cont. • Initially establish a productive infection in epithelial cells, gain access to the sensory nerve endings within the infected area, and travel by retrograde axonal flow to neuronal cells bodies within the respective dorsal root ganglia • Can establish a latent infection within sensory neurons and lytic replication in the nervous system is generally limited • Reactivates periodically in a fraction of the latently infected neuronal cells • The newly replicated virus is transported anterograde, usually to a site at or near the portal of entry into the body, where it may cause a localized lesion 6 ab UL b’a’a’c’ US ca Herpes simplex virus genome The "a" sequences are important in both circularization of the viral DNA, and in packaging the DNA in the virion. The 9,000 bp long repeat (red), which encodes 4 proteins, including an important immediate early regulatory protein (α0). Also includes the promoter of and most of the "gene" for the latency associated transcript (LAT). The long unique region (UL),108 kb long, encodes at least 65 distinct proteins (actually more because some ORFs are spliced and expressed in redundant ways). It contains genes for the DNA replication enzymes and the capsid proteins, as well as many other proteins. The 6,600 bp short repeats (yellow) encodes a transcriptional activator (α4) that acts along with α0 and α27 (in the UL) to stimulate the infected cell for all viral gene expression that leads to viral DNA replication. Three origins of replication: the oriL is in the middle of the UL region, while the oriS is located within the repeat sequences and thus, is present in two copies. The 13,000 bp unique short region (US) encodes 14 ORFs, a number of which are glycoproteins important in viral host range and response to host defense. Herpesvirus Replication (1) • Initial attachment is mediated by the interaction of viral envelope glycoproteins with cell surface receptors. • Tropism of some herpesviruses (e.g. EBV) is restricted as a result of the tissue-specific expression of their receptors. • The nucleocapsid is released into the cytoplasm through fusion of the viral envelope and plasma membrane. • Virally encoded enzymes and transcription factors are carried into the infected cell in the tegument of the virion. 7 Herpesvirus Replication (2) •The nucleocapsid docks with the nuclear membrane and delivers the viral genome into the nucleus, the site of viral gene transcription and replication. •VP16, a structural viral protein, in conjunction with cellular proteins Oct-1 and complex forming factor (CFF), interacts with sequence-specific elements within the promoters of five immediate early (IE) (alpha) genes: ICP0, ICP4, ICP22, ICP27 and ICP47. •Transcription of the viral genome and viral protein synthesis proceeds in a coordinated and regulated manner in the following three phases: •Immediate early proteins(α genes), which are necessary for the regulation of gene transcription and takeover of the cell •Early proteins (β genes), includes more transcription factors and enzymes, including the DNA polymerase •Late proteins (γ genes), which includes the viral structural proteins http://www.dbc.uci.edu/~faculty/wagner/hsvresrch.html Herpesvirus Replication (3) The viral genome is transcribed by the cellular DNA-dependent ribonucleic acid (RNA) polymerase and is regulated by viral encoded and cellular nuclear factors. The interplay of these factors determines whether the proteins necessary for a lytic, persistent or latent infection are produced. 8 Latency: HSV-1 and HSV-2 • The virus establishes latency in sensory neurons. • Some neurons harboring latent virus express an RNA designated as Latency Associated Transcript (LAT). • This RNA does not play a role in the establishment or maintenance of the latent state. • Viral proteins have not been demonstrated in neurons harboring latent virus. • DNA is maintained in episomal form (circular nonintegrated) • Establishment of latency may involve a cellular function. Reactivation from latency • Not all neurons harboring virus reactivate at once. • Reactivation is more frequent than the appearance of clinical lesions: the phenomenon is known as “shedding” which may occur in a significant fraction of infected individuals. • The stimulus for reactivation may differ from one individual to another: e.g., fever, physical, emotional stress, intake of hormones, menstruation, etc. Immunosuppression exacerbates the lesions caused by reactivated virus. • Upon reactivation, the virus multiplies in the sensory neuron and is transported to a site at or near the portal of entry. On rare occasion, the reactivated virus causes ocular or CNS infections. 9 HSV-1 and HSV-2: Establishment of Latency and Reactivation Clinical Manifestations of Herpes Simplex Infections 10 Diseases Caused by Herpes Oral HSV Simplex Viruses • Orolabial herpes (almost all type 1) Primary – HSV gingivostomatitis Recurrent – herpes labialis (cold sores, fever blisters) • Ocular infections • Cutaneous herpes • Genital herpes (mostly type 2) Primary Recurrent • Herpes proctitis, perianal herpes (type 2) • HSV encephalitis Diseases Caused by Herpes Oral HSV Simplex: Orolabial • • • • Mostly caused by HSV-1 In primary herpetic gingivostomatitis, typical clear lesions first develop, followed by ulcers with white appearance Infection initially on the lips and spreads to all parts of the mouth and pharynx Spectrum of severity of clinical illness ranging from a few intraoral ulcers and no systemic features, to severe oral ulceration. Clinical symptoms include: Painful vesicles that burst to leave ragged ulcers Drooling, from increased saliva production due to inflammation Systemic symptoms including fever, cervical lymphadenitis, etc. • Risk of autoinoculation to other sites (eye, skin) highest during the acute phase of the primary infection Herpetic whitlow Herpetic keratitis • Disease is more severe in immunosuppressed people 11 Diseases Caused by Herpes Simplex: Orolabial (2) • HSV-1 latency in trigeminal ganglia • Reactivation triggers include: UV exposure, fever, menses, trauma, etc. • Reactivation from trigeminal ganglia often results in cold sore development at or near initial site of infection • prodrome of itching, tingling; cluster of painful vesicles • External lesions • Lips (90%) • Nose, chin, or cheeks (10%) • Intra-oral lesions can also occur Diseases Caused by Herpes Oral HSV Simplex Viruses: Oral Herpes simplex cold sore Gingivostomatitis in an AIDS patient Herpetic gingivitis 12 Herpes Labialis: Epidemiology in the US • Cold sores affect 1 in 5 people • ~98 million cases each year • 35%-60% Caucasians: HSV-1 serology + • Seroprevalence dependent on age, socioeconomic status, and geographic location • Primary infection occurs almost exclusively before age 4 Usually asymptomatic • 20%-40% of individuals have recurrent outbreaks In only 1% recurrence is severe Herpes Labialis – Principles Of Antiviral Treatment • Treat early (during prodrome) – rapid natural evolution of disease • Short duration of therapy – period of viral replication is brief (2-3 days), then controlled by host immunity • High dose systemic therapy – deliver high drug concentrations to tissue, prevent extension of HSV to uninfected cells Ref: Spruance et al, J infect Dis 161:185, 1990 13 Topical Antiviral Therapy for Herpes Labialis • Penciclovir 1% cream (Denavir®) Reduced duration of lesion pain Accelerated lesion healing Apply topically q2h FDA approved September, 1996 • Acyclovir 5% In PEG ointment – not effective In MAC cream Diseases Caused by Herpes Simplex Viruses: Whitlow • Direct contact with body secretions • Infection occurs through small cuts or abrasions on the fingers, hand or wrist • HSV-1 or HSV-2 • Often seen in health care workers treating patients with HSV infections • Thumb-sucking children are also more susceptible Herpetic whitlow 14 Diseases Caused by Herpes Simplex Viruses: Bell’s Palsy • Acute onset of unilateral facial nerve palsy • Multiple causes; many of the “idiopathic” cases may be associated with HSV reactivation • Small studies with prednisone and/or acyclovir have yielded variable results – larger studies ongoing • Many experts recommend valacyclovir + prednisone – optimal dose & duration not known Diseases Caused by Herpes Simplex Viruses: Encephalitis • Reactivation of HSV-1; 1250 cases/yr in US of HSE • Presentation: AMS, fever, HA, personality change, seizures • Diagnosis: MRI – temporal lobe lesion CSF – PCR for HSV DNA • Therapy: Acyclovir 10-15 mg/kg q8h x 21d • Outcome (with therapy): mortality 13% at 1 mo., 28% at 18 mo, 38% have no/mild impairment at 6 mo 15 Diseases Caused by Herpes Simplex Viruses: Genital Herpes • Mostly likely caused by HSV-2 • 18-20% of US population is HSV-2 seropositive • Primary infection may be more severe, may be associated with systemic syndromes, or may be asymptomatic. • Variable frequency of recurrences – about 90% will have at least 1 recurrence/year • Vesicles/ulcerations on genitals or perigenital skin • Key to natural history – asymptomatic viral shedding Antiviral Therapy for Primary Genital HSV • • • Acyclovir 200 mg po 5x daily for 10 days Valacyclovir 1000 mg po bid for 10 days Famciclovir* 125 mg po tid for 10 days *not FDA approved for this indication 16 Oral HSV Diseases Caused by Herpes Simplex Viruses: Ocular Infections • HSV-1 usually associated with ocular infections • Leading cause of blindness in the United States • Typically is limited to one eye • A number of manifestations, including blepharitis, conjunctivitis, keratis, retinitis • Long-term (12 month) suppressive therapy with an oral antiviral drug (acyclovir) reduces rates of recurrence of HSV keratitis and rates of recurrence of ocular complications Oral HSV Diseases Caused by Herpes Simplex Viruses: Ocular Infections 17 Antiviral Drugs All anti HSV drugs currently licensed are dependent on being activated by viral enzymes. Herpes Simplex Viruses: Antiviral Therapy and Drug Resistance nucleoside analogs Drugs are activated by a viral enzyme, thymidine kinase Only activated in herpes-infected cells, few side effects as a consequence. The best known nucleoside analog used to treat herpes simplex virus infections is acycloguanosine (acyclovir); other approved drugs include famciclovir and valacyclovir. Drug resistant herpes mutants do arise with all of these, although resistant strains are usually less virulent than the wild type. Drugs act only against replicating virus (incorporated into DNA during synthesis); are ineffective against latent virus. 18 Herpes Simplex Viruses: Antiviral Therapy and Drug Resistance acyclovir acyclo GMP acyclo GTP Herpes Simplex Viruses: Antiviral Therapy and Drug Resistance Mechanisms of Action of Acyclovir: Inhibition of DNA Synthesis & Chain Termination 19 Activation of acyclovir (ACV) and gancyclovir (GCG) by viral enzymes Anti-CMV drugs Anti-HSV drugs DNA DNA Inhibits polymerase GCG-TP Inhibits polymerase; DNA terminator ACG-TP ACG-DP GCG-DP ACG-MP GCG-MP GCG Viral protein kinase Viral Thymidine kinase ACG Wide substrate range Varicella-Zoster Virus (VZV) • Primary infection causes chickenpox (varicella) • Reactivated virus causes herpes zoster, or shingles • Spreads primarily through the respiratory route • Local replication occurs in respiratory tractleads to formation of skin lesions over the entire body • Latency is established in dorsal root or cranial nerve ganglia • Antibody immunity limits viremic spread of VZV • Cell-mediated immunity important for limiting progression and resolving disease 20 Varicella-Zoster Virus (VZV) shingles chickenpox VZV in newborn Varicella-Zoster Virus (VZV): treatment • For children with chickenpox, normally supportive care is sufficient • Acyclovir, famciclovir and valacyclovir are used to prevent disseminated infection in immunosuppressed patients • Protection for immunosuppressed patients also may come from varicella-zoster immunoglobulin (VZIg). This can prevent viremic spread, but is ineffective for patients with active varicella or herpes zoster disease. • An FDA-approved live virus vaccine is available for VZV 21 Epstein-Barr Virus (EBV) • First discovered as the causative agent for African Burkitt’s lymphoma (AfBl). • Later identified as causative agent for infectious mononucleosis (“kissing disease”) when serum from infected individual was found to contain antibody to AfBl. • Tissue tropism is determined by expression of its receptor, which is same receptor for the C3d component of the complement system (a.k.a. CR2 or CD21). Infection is limited to only those cell types which express this receptor. • Encodes more than 70 proteins • Has both semi-permissive and permissive replicative cycles EBV Gene Expression Semi-permissive replication • In latently infected lymphocytes, a few unintegrated copies are replicated every time the cell divides. Early immediate genes are expressed, including the EBV nuclear antigens, two latent membrane proteins (LMPs), and two small RNA molecules (EBER-1 and EBER-2). • The LMPs are oncogenes that stimulate growth of and immortalize the B cell. Permissive replication • In epithelial cells or B cells permissive for EBV replication, expression of ZEBRA protein activates early genes, resulting in expression of the polymerase and DNA replication and rest of the lytic cycle. 22 EBV Pathogenesis: B cell transformation • Taken up by CD21+ B lymphocytes following virus replication in pharyngeal epithelial cells and shedding in saliva • Infected B cells become protected from undergoing apoptosis; cell is transformed • B cell transformation changes the interaction of the cell with other immune system components EBV Pathogenesis: Burkitt’s lymphoma • Tumor of the jaw and face found in children, with evidence of EBV DNA and tumor antigens in the tumor cells • Tumor cells are monoclonal and show a characteristic translocation between chromosomes 8 and 14, which brings the c-myc gene next to the gene for the Ig heavy chain. • Result is elevated transcription of c-myc • Biopsy shows large multinucleated cells 23 EBV Pathogenesis: Burkitt’s lymphoma EBV Pathogenesis: Nasal Pharyngeal Cancer • Tumor of the epithelium and upper respiratory tract • Occurrences seen in south China, Alaska, Tunisia, east Africa • Genetic predisposition may be involved, or possibly environmental co-factor 24 EBV Pathogenesis: Oral Hairy Leukoplakia • An unusual manifestation of a productive EBV infection in HIV-infected patients with AIDS • Characterized by a nonpainful white plaque along the lateral tongue borders. • May spread to contiguous sites such as the mouth floor, tonsil, and pharynx. EBV Pathogenesis: Infectious mononucleosis • Characterized by malaise, lymphadenopathy, tonsilitis, enlarged spleen and liver, and fever, which may persist for more than a week. • A rash may also develop • Resolution usually occurs in 1 to 4 weeks • Complications can include neurological disorders, including meningitis, encephalitis, myelitis and Guillain-Barre syndrome. 25 EBV Diagnosis and Treatment • Blood smears showing atypical lymphocytes in infectious mononucleosis • Serological tests are also available for diagnosis • No drugs are currently available to treat Epstein-Barr virus, since it lacks a thymidine kinase • A vaccine is under development Cytomegalovirus (CMV) • Largest genome of all herpesviruses • Only replicates in human cells • Productive infection seen in macrophages and fibroblasts, whereas latent infection is set up in several cell types, including T lymphocytes and stromal cells of the bone marrow. • Found in a significant proportion of the population. • Spread mediated through most secretions, including saliva, urine, vaginal and semen. • Maternal to fetal spread can occur during pregnancy 26 CMV and Disease: Congenital Infection • Most common viral cause of congenital disease. • Up to 1/40 newborns in the United States are infected by the virus. • During primary infection of the mother, the virus can spread via the placenta to the fetus. • Abnormalities include microcephaly, rash, brain calcification and hepatosplenomegaly. • Can result in hearing loss or retardation • Neonates can also receive virus through blood transfusion, in which the titer of virus is much higher. CMV: Infection in the immunocompromised host • Pneumonia and pneumonitis, which is fatal if not treated • In 16-19% of patients with terminal AIDS, CMV produces a hemorrhagic, necrotizing retinitis. • In 10% of AIDS patients, CMV colitis or esophagitis may develop 27 CMV: Diagnosis • Most infections are asymptomatic and are undetected • Hallmark of infection is the cytomegalic cell, which is an enlarged cell that contains a dense, central “owl’s eye” basophilic intranuclear inclusion body • Viral antigens can be detected using immunofluorescent antibodies, ELISA, or PCR CMV: Antiviral Therapy • Ganciclovir, valganciclovir, cidofovir and foscarnet have all been approved by the FDA for the treatment of specific diseases resulting from CMV infection in immunosuppressed patients • Progression of CMV retinitis may be delayed in the short term by intravenous ganciclovir or foscarnet. Both are equally effective; however, foscarnet is not as well tolerated. • Acyclovir is not effective for CMV infections • A vaccine is currently under development 28 HHV-6 and HHV-7 • Human herpesvirus 6 (roseola) has two variants, 6A and 6B. • Is found worldwide and in the saliva of >90% of adults. • Almost all children are infected by the age of two, and the infection is life-long. • Replicates in T and B cells • HHV-6B causes exanthem subitum, otherwise known as roseola infantum. • Symptoms include fever and sometimes upper respiratory tract infection and lymphadenopathy. • 14 day incubation period. • After fever subsides, a macropapular rash appears on trunk and neck that lasts a few days. • Human herpesvirus 7 is similar to HHV-6 and may be responsible for some cases of exanthem subitum. • HHV-7 binds to CD4 antigen and replicates in CD4+ T cells. HHV-6 and HHV-7, Clinical Manifestations Exanthem Subitum; Roseola Infantum 29 HHV-8 (Kaposi’s Sarcomaassociated herpesvirus, or KSHV) • Initially found in biopsy specimens of Kaposi’s sarcoma in AIDS patients • Like EBV, B cells are primary target, but also infects other cell types • HHV-8 is found in ~10% immunocompetent people in peripheral blood lymphocytes • Encodes several proteins with homology to human proteins that promote growth and prevent apoptosis of infected cell and surrounding cells. • Characteristic opportunistic infection associated with AIDS. Oral KSHV Clinical Manifestations • Over 50% of AIDS patients with skin KS also have oral KS, while 10-20% of AIDS patients have only oral KS • Oral KS typically found on the hard and soft palates, but also can develop on the gingiva, the anterior palate, the tongue and the buccal mucosa • Characterized by one or more generally flat areas, with a reddish brown or deep purple discoloration • A hyperplastic, or nodular, variety of KS can also be seen, mostly on the gingiva or tongue. Hyperplastic KS will frequently bleed. • As disease progresses, and color can change to darker reddish brown, even violet 30 KSHV Clinical Manifestations A. Flat Kaposi's sarcomas involving the hard and soft palates. B. Nodular gingival Kaposi's sarcoma (arrows). C. Multiple skin Kaposi's sarcomas in a patient with advanced AIDS. Treatment of KSHV Oral Lesions • Large intraoral lesions of KS can be treated with systemic chemotherapeutic single-agents such as vinblastine and vincristine. • Radiation therapy is also used in large lesions (after prior use of chlorhexidine gluconate rinse to diminish severity of glossitis and mucositis that are side effects of radiation) • Small intraoral lesions can be treated with surgical and laser excision. Nonsurgical treatment is also effective, such as intralesional injections of vinblastine sulfate. 31 Herpesvirus simiae (B virus) • Indigenous to Old World monkeys, such as macaques. • Transmitted to humans via monkey bites or saliva, or infected cell tissues • 75% of human cases result in death • Most survivors have serious neurologic damage. • Both acyclovir and ganciclovir are recommended for therapy, but efficacy is unknown (some evidence for efficacy in the literature). The take home message: 32