* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Atomic Structure PP
Survey
Document related concepts
Transcript
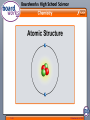
1 of 6 © Boardworks Ltd 2008 Atomic Structure For some time, people thought that atoms were the smallest particles and could not be broken into anything smaller. Scientists now know that atoms are actually made from even smaller particles called protons, neutrons and electrons. proton neutron electron How are these particles arranged inside the atom? 2 of 6 © Boardworks Ltd 2008 Atomic Structure The protons and neutrons are located in the dense core at the center of the atom. This is called the nucleus. The nucleus has a positive charge (protons are positive and neutrons are neutral and don’t have a charge). The electrons are spread out around the edge of the atom. They orbit the nucleus in layers called shells. Electrons have a negative charge. 3 of 6 © Boardworks Ltd 2008 Labeling the atom 4 of 6 © Boardworks Ltd 2008 Atomic Number The atoms of any particular element always have the same number of protons. For example: hydrogen atoms always contain 1 proton carbon atoms always contain 6 protons magnesium atoms always contain 12 protons. The number of protons in an atom is known as the atomic number and is the smaller of the two numbers shown in most periodic tables. 5 of 6 © Boardworks Ltd 2008 What is the atomic number? What are the atomic numbers of these elements? sodium 11 iron 26 tin 50 fluorine 6 of 6 9 © Boardworks Ltd 2008 Atomic Number On very rare occasions, the number of protons can change, which means that it is now a different element. This happens with radioactive decay and nuclear bombs/reactors. 7 of 6 © Boardworks Ltd 2008 Mass Number Electrons have a mass of almost zero, which means that the mass of each atom results almost entirely from the number of protons and neutrons in the nucleus. The sum of the protons and neutrons in an atom’s nucleus is the mass number. It is the larger of the two numbers shown in most periodic tables. To calculate how many neutrons there are in an atom, simply subtract: mass number minus atomic number (protons). Atoms Protons Neutrons Mass number hydrogen 1 0 1 lithium 3 4 7 aluminum 13 14 27 8 of 6 © Boardworks Ltd 2008 What’s the mass number? mass number = number of protons + number of neutrons What is the mass number of these atoms? Atoms Protons Neutrons Mass number helium 2 2 4 copper 29 35 64 cobalt 27 32 59 iodine 53 74 127 germanium 32 41 73 9 of 6 © Boardworks Ltd 2008 How many neutrons? number of neutrons = mass number - number of protons = mass number - atomic number How many neutrons are there in these atoms? Atoms Mass number Atomic number Neutrons helium 4 2 2 fluorine 19 9 10 strontium 88 38 50 zirconium 91 40 51 uranium 238 92 146 10 of 6 © Boardworks Ltd 2008 Building a nucleus 11 of 6 © Boardworks Ltd 2008 Calculating Electrons Every atom has a neutral charge, so must have equal numbers of protons and electrons. To calculate electrons, just look at the atomic number (# of protons)! Atoms Protons Neutrons Electrons helium 2 2 2 copper 29 35 29 iodine 53 74 53 Atomic number is the number of protons rather than the number of electrons, because atoms can lose or gain electrons but do not normally lose or gain protons. 12 of 6 © Boardworks Ltd 2008 How many electrons per shell? The arrangement of electrons in shells is called the electron configuration. Each shell can hold a certain number of electrons. 1st shell holds a maximum of 2 electrons 2nd shell holds a maximum of 8 electrons 3rd shell holds a maximum of 8 electrons This electron arrangement is written as 2,8,8. 13 of 6 © Boardworks Ltd 2008 Making a Bohr Model Using Helium 1. Use the periodic table to determine how many protons, neutrons and electrons are in the atom. P=____ N=_____ E=_____ 2. Draw a circle and label the # of P and N in the inside of the circle P= N= 14 of 6 © Boardworks Ltd 2008 Making a Bohr Model Using Helium 3. Draw your 1st electron shell (with up to 2 electrons). P= 2 N= 2 P= 2 N= 2 4. If you need to add more electrons, you need to add more electron shells! Remember…2, 8, 8!!! 15 of 6 © Boardworks Ltd 2008 Calculate electron configurations 16 of 6 © Boardworks Ltd 2008 Which element? 17 of 6 © Boardworks Ltd 2008 What are the missing numbers? 18 of 6 © Boardworks Ltd 2008