* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download PHARMACOKINETICS
Discovery and development of proton pump inhibitors wikipedia , lookup
Specialty drugs in the United States wikipedia , lookup
Polysubstance dependence wikipedia , lookup
Compounding wikipedia , lookup
Plateau principle wikipedia , lookup
Orphan drug wikipedia , lookup
Psychopharmacology wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Drug design wikipedia , lookup
Neuropharmacology wikipedia , lookup
Pharmacognosy wikipedia , lookup
Drug discovery wikipedia , lookup
Pharmaceutical industry wikipedia , lookup
Prescription costs wikipedia , lookup
Pharmacogenomics wikipedia , lookup
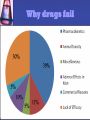
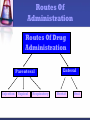
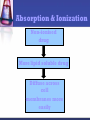
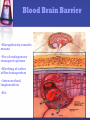
PHARMACOKINETICS “What the body does to the drug” Pharmacokinetics (PK) The study of the disposition of a drug The disposition of a drug includes the processes of ADME Absorption Distribution Metabolism Excretion Toxicity Elimination ADMET DRUG R&D Drug discovery and development •10-15 years to develop a new medicine •Likelihood of success: 10% •Cost $800 million – 1 billion dollars (US) Why drugs fail Importance of PK studies Patients may suffer: Toxic drugs may accumulate Useful drugs may have no benefit because doses are too small to establish therapy A drug can be rapidly metabolized. Routes Of Administration Routes Of Drug Administration Parenteral Injection Topical Respiratory Enteral Rectal Oral Absorption The process by which drug proceeds from the site of administration to the site of measurement (blood stream) within the body. Necessary for the production of a therapeutic effect. Most drugs undergo gastrointestinal absorption. This is extent to which drug is absorbed from gut lumen into portal circulation Exception: IV drug administration IV vs Oral I.V Drug Oral Drug Immediately Delayed completely incomplete The Process Absorption relies on Passage through membranes to reach the blood passive diffusion of lipid soluble species. The Rule of Five formulation Poor absorption or permeation are more likely when: There are more than 5 H-bond donors. The molecular weight is over 500. The LogP is over 5. There are more than 10 H-bond acceptors. Absorption & Ionization Non-ionised drug More lipid soluble drug Diffuse across cell membranes more easily First Pass Metabolism Destroyed in gut Dose Not absorbed Destroyed by gut wall Destroyed by liver to systemic circulation Bioavailability: the fraction of the administered dose reaching the systemic circulation Determination of bioavailability A drug given by the intravenous route will have an absolute bioavailability of 1 (F=1 or 100% bioavavailable) While drugs given by other routes usually have an absolute bioavailability of less than one. The absolute bioavailability is the area under curve (AUC) non-intravenous divided by AUC intravenous . Toxicity The therapeutic index is the degree of separation between toxic and therapeutic doses. Relationship Between Dose, Therapeutic Effect and Toxic Effect. The Therapeutic Index is Narrow for Most Cancer Drugs 100× 10× Distribution The movement of drug from the blood to and from the tissues DISTRIBUTION Determined by: • partitioning across various membranes •binding to tissue components •binding to blood components (RBC, plasma protein) •physiological volumes DISTRIBUTION All of the fluid in the body (referred to as the total body water), in which a drug can be dissolved, can be roughly divided into three compartments: intravascular (blood plasma found within blood vessels) interstitial/tissue (fluid surrounding cells) intracellular (fluid within cells, i.e. cytosol) The distribution of a drug into these compartments is dictated by it's physical and chemical properties TOTAL BODY WATER Vascular Extravascular Intracellular 3L 9L 28 L 4% BW 13% BW 41% BW Distribution Apparent volume of distribution (Vd) = Amt of drug in body/plasma drug conc VOLUME OF DISTRIBUTION FOR SOME DRUGS DRUG Vd (L) cocaine 140 clonazepam 210 amitriptyline 1050 amiodarone ~5000 Factors affecting drugs Vd Blood flow: rate varies widely as function of tissue Muscle = slow Organs = fast Capillary structure: •Most capillaries are “leaky” and do not impede diffusion of drugs •Blood-brain barrier formed by high level of tight junctions between cells •BBB is impermeable to most water-soluble drugs Blood Brain Barrier •Disruption by osmotic means •Use of endogenous transport systems •Blocking of active efflux transporters • Intracerebral implantation •Etc Plasma Protein Binding Many drugs bind to plasma proteins in the blood steam Plasma protein binding limits distribution. A drug that binds plasma protein diffuses less efficiently, than a drug that doesn’t. Physiochemical propertiesPo/w The Partition coefficient (Po/w) and can be used to determine where a drug likes to go in the body Any drug with a Po/w greater than 1(diffuse through cell membranes easily) is likely be found throughout all three fluid compartments Drugs with low Po/w values (meaning that they are fairly water-soluble) are often unable to cross and require more time to distribute throughout the rest of the body Physiochemical PropertiesSize of drug •The size of a drug also dictates where it can go in the body. •Most drugs : 250 and 450 Da MW •Tiny drugs (150-200 Da) with low Po/w values like caffeine can passively diffuse through cell membranes •Antibodies and other drugs range into the thousands of daltons •Drugs >200 Da with low Po/w values cannot passively cross membranes- require specialized protein-based transmembrane transport systems- slower distribution •Drugs < thousand daltons with high Po/w values-simply diffuse between the lipid molecules that make up membranes, while anything larger requires specialized transport. Elimination The irreversible removal of the parent drugs from the body Elimination Excretion Drug Metabolism (Biotransformation) Drug Metabolism The chemical modification of drugs with the overall goal of getting rid of the drug Enzymes are typically involved in metabolism Metabolism Drug More polar (water soluble) Drug Excretion METABOLISM •From 1898 through to 1910 heroin was marketed as a nonaddictive morphine substitute and cough medicine for children. Bayer marketed heroin as a cure for morphine addiction •Heroin is converted to morphine when metabolized in the liver Phases of Drug Metabolism Phase I Reactions Convert parent compound into a more polar (=hydrophilic) metabolite by adding or unmasking functional groups (-OH, -SH, -NH2, COOH, etc.) eg. oxidation Often these metabolites are inactive May be sufficiently polar to be excreted readily Phases of metabolism Phase II Reactions Conjugation with endogenous substrate to further increase aqueous solubility Conjugation with glucoronide, sulfate, acetate, amino acid Mostly occurs in the liver because all of the blood in the body passes through the liver The Most Important Enzymes Microsomal cytochrome P450 monooxygenase family of enzymes, which oxidize drugs Act on structurally unrelated drugs Metabolize the widest range of drugs. CYP family of enzymes • Found in liver, small intestine, lungs, kidneys, placenta • Consists of > 50 isoforms • Major source of catalytic activity for drug oxidation • It’s been estimated that 90% or more of human drug oxidation can be attributed to 6 main enzymes: • CYP1A2 • CYP2D6 • CYP2C9 • CYP2E1 • CYP2C19 • CYP3A4 In different people and different populations, activity of CYP oxidases differs. Inhibitors and inducers of microsomal enzymes Inhibitors: cimetidine prolongs action of drugs or inhibits action of those biotransformed to active agents (pro-drugs) Inducers: barbiturates, carbamazepine shorten action of drugs or increase effects of those biotransformed to active agents Blockers: acting on non-microsomal enzymes (MAOI, anticholinesterase drugs) Phase II Main function of phase I reactions is to prepare chemicals for phase II metabolism and subsequent excretion Phase II is the true “detoxification” step in the metabolism process. Phase II reactions Conjugation reactions Glucuronidation (on -OH, -COOH, -NH2, -SH groups) Sulfation (on -NH2, -SO2NH2, -OH groups) Acetylation (on -NH2, -SO2NH2, -OH groups) Amino acid conjugation (on -COOH groups) Glutathione conjugation (to epoxides or organic halides) Fatty acid conjugation (on -OH groups) Condensation reactions Glucuronidation Conjugation to a-d-glucuronic acid Quantitatively the most important phase II pathway for drugs and endogenous compounds Products are often excreted in the bile Phase I and II - Summary Products are generally more water soluble These reactions products are ready for (renal) excretion There are many complementary, sequential and competing pathways Phase I and Phase II metabolism are a coupled interactive system interfacing with endogenous metabolic pathways Excretion The main process that body eliminates "unwanted" substances. Most common route - biliary or renal Other routes - lung (through exhalation), skin (through perspiration) etc. Lipophilic drugs may require several metabolism steps before they are excreted ADME - Summary