* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Presentation
Environmental persistent pharmaceutical pollutant wikipedia , lookup
Psychopharmacology wikipedia , lookup
Medical prescription wikipedia , lookup
Orphan drug wikipedia , lookup
Compounding wikipedia , lookup
Neuropharmacology wikipedia , lookup
Adherence (medicine) wikipedia , lookup
Drug design wikipedia , lookup
Pharmacokinetics wikipedia , lookup
Drug interaction wikipedia , lookup
Pharmacognosy wikipedia , lookup
Drug discovery wikipedia , lookup
Pharmacogenomics wikipedia , lookup
Pharmaceutical marketing wikipedia , lookup
Pharmaceutical industry wikipedia , lookup
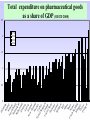
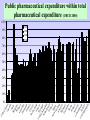
Perspectives and achievements with Rational Pharmacotherapy Meeting under Danish EU presidency Implementation of Rational Pharmacotherapy Copenhagen , November 6, 2002 WHO/EURO Kees de Joncheere, Regional Adviser Pharmaceuticals Overview of the presentation • • • • Pharmaceutical policies in Europe Rational use of medicines Comparing European countries Improving the prescribing and use of medicines • Concluding remarks Challenges for pharmaceutical policies in Europe • Equitable access for patients to effective, safe and good quality medicines • Enhancing appropriate use of medicines for better health outcomes • Ensuring value for money • Balance with industrial policy objectives Values underpinning health systems : equity, quality, solidarity, participation and accountability Pharmaceutical policies and the EU • EU regulatory framework and legislation • National responsibilities on pricing and reimbursement (“subsidiarity”) • Transparency directive • Industrial policy communication 1995 • Single market communication 1998 • Public health communication 2000 • Portugal 2000, EU MINE , DG5/health High Level committee, G 10 , … • Review 2000-1 Medicines and Public health in EU • Future of the European system for evaluations and supervision of medicinal products • Relevance of the added therapeutic value of medicinal products ( registration criteria ? Placebo or comparator?) • Aspects determining innovation and research • Rational use of medicinal products • Importance and evolution trends for the information systems • Increasing use of generic medicines • Adoption of EU public health action plan, 2002 Sw U itze ni rl te an d d S N tate o s D rwa en y m ar I r N el k et an he d r Au lan Lu st ds xe ral m ia b Ca ourg G nad er a m Sw any ed Au en str F N in ia ew la Ze nd a ni Ic lan te e l d d an K d in g Fr dom a Be nce lg iu m Ita l Sp y ai Cz J ec a n h pa Re n p P o ub rtu lic H ga un l g G ary re e T u ce rk ey Total expenditure on pharmaceutical goods as a percentage of total expenditure on health (OECD 2000) 45 40 35 30 0 1970 1980 1990 1996 25 20 15 10 5 a Ire rk la N nd Lu or xe wa m y Sw bo itz urg N erl et a n he d r A land us s tra A lia us tr N Fin ia ew la Ze nd a Sw land ed U ni T en te ur d ke Ki y ng d U Ca om ni na te da d S Ge tate rm s a Ic ny el Be and lg iu m Ita l Ja y pa Sp n Cz F ain ec ra h nc Re e p H ubl un ic ga G ry re Po ece rtu ga l m D en 2.5 Total expenditure on pharmaceutical goods as a share of GDP (OECD 2000) 2 1970 1980 1990 1996 1.5 1 0.5 0 Link between total expenditure on pharmaceutical goods per capita and GDP per capita at purchasing power parities Total expenditure on pharmaceutical goods, $/capita PPP (OECD 2000) 400 350 JPN FRA ISL BEL ITA DEU 300 PRT GRC 250 CZE USA CAN AUT ESP 200 HUN GBR SWE FIN AUS NZL NLD 150 LUX CHE NOR DNK IRL 100 50 5000 10000 15000 20000 GDP per capita $ ppp 25000 30000 35000 te d St Ca ates na da Ita Be ly lg i Fi um n A land us t D rali en a m A ark us t G ria Sw re it z e c e er la Fr nd an ni Po ce te rtu d Ki gal N ng et do he m rla Ic nds el an N Ja d ew p Ze an a Sw land e H den un Ge gar rm y an Sp y a Ire in la N nd Lu or xe wa m y b Cz T our ec ur g h ke Re y pu bl ic U ni Public pharmaceutical expenditure within total pharmaceutical expenditure (OECD 2000) 100% 90% 80% 1970 1980 1990 1996 70% 60% 50% 40% 30% 20% 10% 0% Rational use of medicines • • • • • Right patient with right indication Right medicine Right dose/administration Right information Right moment to stop or change Inappropriate use leads to • Adverse effects • Sub-optimal outcomes • Waste of resources ( money, health professionals and patients time) Why are drugs not used rationally ? • • • • • • • • Lack of training and knowledge Marketing practices Financial incentives for irrational use Availability problems Patient expectations Prescribing as a means to finish the consultation Health systems and services effects … Main discrepancies in the EU pharma markets • • • • • • • Number of licensed medicines Reimbursement classes Legal categories Price differentials Licensed indications and dosages Utilisation Medicines withdrawn from a country still used in others 15 different systems for 15 Member States An overview of the European differences C01EB15 - Trimetazidine Trade Names UK DNK + Presentations Average Reimbursement Expenditure by health systems (€) Rank by cost (EURO) withdrawn from the market 1 1 34% 103,382 85,371,000 FRA + 16 31 78% ITA + 1 1 0% 0 PLN + 1 2 0% 0 46 Source: EURO-Medicines database 4 An overview of the European differences Approved Indications of Trimetazidine oral forms (tablets and solution) FRA ITA IRL DNK Angina pectoris Ischemic eye diseases Ischemic ear diseases (symptomatic treatment) Meniere’s disease (DK only) Source: EURO-Medicines database + + + + - + + + + prescriptions PPI Prescribing - Diagnosis at first prescription 3500 GI Misc. 3000 Non specific Abdominal Pain 2500 Non Ulcer Dyspepsia 2000 1500 1000 Hiatus Henia/Reflux Unlicensed Indications Oesophagitis Ulcer unspecified Indication licensed since 1991 Gastric 500 Duodenal 0 1991 1995 Source of Data : General Practice Research Database (GPRD) DEPARTMENT OF MEDICINES MANAGEMENT Tel: 01782 583444 Fax: 01782 713 586 E-mail: [email protected] Measuring and monitoring the use of medicines • Describe and compare existing situations and patterns • Identify differences and potential problems • Monitor results of interventions • Lessons drawn from other countries` experiences From drug utilisation to costeffective intervention (1) • Drug utilisation studies tend to be descriptive, aggregated data : WHAT? • Indicator studies more focused on rational drug use: WHAT? HOW MUCH? • Qualitative studies WHY? From drug utilisation to costeffective intervention (2) • Intervention studies HOW MUCH? WHY? (intervention) HOW MUCH NOW? Conclusion DOES IT WORK? IS THE INTERVENTION EFFECTIVE? • Management studies IS THE INTERVENTION REPRODUCABLE? IS IT COST-EFFECTIVE? Examples of indicators (WHO/INRUD) Prescribing indicators • Average number of drugs per encounter (<2) • Percentage of drugs prescribed by generic name (close to 100%) • Percentage of encounters with an antibiotic prescribed (<30%) • Percentage of encounters with an injection prescribed (<10%) • Percentage of drugs prescribed from EDL or formulary (close to 100%) Examples of indicators (EU countries ) • Rate indicators – – – – – NSAIDs antibiotics benzodiazepines anti-ulcer drugs inhaled corticosteroids • Choice indicators – – – – – inhaled corticosteroids/beta agonists quinolones/total antibiotics short/long acting NSAIDs PPI/total anti-ulcer trimethoprim/cotrimoxazole Examples of indicators • Appropriateness – drugs of limited value – agreement with clinical practice guidelines • ASA after AMI • ACE-s in CHF • Statins in secondary prevention • Economic – choice of branded/generic agents – cheaper drug classes – compliance with budgets • Errors – drug-drug interactions – duplication (drug or class) – dosage (con`t) Challenges on monitoring drug use • Pharmaco-epidemiology is often concerned with links between exposure and outcomes (usually adverse) • Drug prescribing data-sets have other uses – Budget tracking and forecasting – Examining trends in drug use – Assessing appropriateness of drug selection and accuracy and safety of prescribing • Clear need for going beyond basic statistics and for developing better analytical methods and benchmarking tools Challenges for monitoring drug use • Intercountry variability as ‘learning device’ • Improving networking of researchers and policy makers • Gap between sales/aggregated consumption data and individual patient data (privacy!) Financial measures on improving prescribing and containing costs Fixed and indicative budgets for prescribers Budgeting for regions, PHC groups with pay-back mechanisms Price regulations and reference prices Patient co-payment Financial incentives to pharmacies for better dispensing Differential reimbursement rates Promote generics Managerial measures on improving prescribing and containing costs Positive and negative lists Disease management Restrict distribution and prescription Prescribing support systems Regulate marketing, approved indications and commercial information Practice guidelines Improving prescribing and containing costs : educational and informational measures Formularies University training and continuing information EBM - evidence based practice guidelines Pharmaceutical care Prescriber information Drug committees in hospitals and primary care, coordinated approach Feedback and discussion of drug use data Behavioural approaches Patient information Promoting rational prescribing Effective interventions • Combination of strategies • Participation and ownership by health professionals • Follow-up to avoid relapse • Feedback and active discussion • Best practices Promoting rational prescribing proven effective interventions • Standard treatment guidelines, when evidencebased, developed with end-users, with active dissemination and follow-up • Essential Medicines lists, • Hospital Drugs and Therapeutic Committees • Undergraduate training • Discussion groups with feedback of prescribing data • “Academic detailing” • Comprehensive approach, with all components Promoting rational prescribing interventions that need more testing Mixed results, probably effective: • Pharmacists interventions • Public education • Financial incentives and reimbursement measures Mixed results, probably ineffective: • Drug information bulletins and other printed materials used in isolation • Arbitrary prescription limitations • Traditional stand-up lecturing Promoting rational use : involving patients and sharing experience Patient • experience of illness • social circumstances • attitude to risk • values • preferences Picker Institute Health professional • diagnosis • disease aetiology • prognosis • treatment options • outcome probabilities Conclusions • Room for improving drug use, e.g minimising risk and improving outcomes • Important role for health professionals , patients and coordinated team approach • Need for better evidence and information on interventions that work