* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Final Report - Research
Cardiovascular disease wikipedia , lookup
Remote ischemic conditioning wikipedia , lookup
Lutembacher's syndrome wikipedia , lookup
Quantium Medical Cardiac Output wikipedia , lookup
History of invasive and interventional cardiology wikipedia , lookup
Management of acute coronary syndrome wikipedia , lookup
Coronary artery disease wikipedia , lookup
Dextro-Transposition of the great arteries wikipedia , lookup
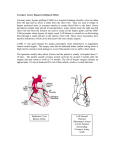
Improving CABG Surgery Lucas Burton Amir Durrani Benjamin Hoagland Santosh Tumkur Department of Biomedical Engineering Vanderbilt University School of Engineering April 23, 2003 ADVISORS: Thomas Ryan, PhD Fellow at Ethicon Inc. Jia Hua Xiao, PhD Principle Engineer at Ethicon, Inc. INSTRUCTOR: Paul King, PhD, PE Associate Professor of Biomedical and Mechanical Engineering Vanderbilt University School of Engineering TABLE OF CONTENTS 1. ABSTRACT 3 2. INTRODUCTION 2.1. OVERVIEW OF CORONARY ARTERY BYPASS SURGERY 2.2. CURRENT TREATMENTS 2.3. DESIGN GOALS 3 4 5 3. METHODOLOGY 3.1. TIMELINE 3.2. DESIGN AND PROTOTYPE 3.3. TESTING 7 7 9 4. RESULTS 4.1. 4.2. 4.3. 4.4. 4.5. ANASTOMOSIS DEVICE DESIGN IMPROVED CABG LOCALIZED STABILIZATION DEVICE MATERIAL SPECIFICATION SAFETY ANALYSIS ECONOMIC ANALYSIS 10 11 12 14 14 5. CONCLUSION 15 6. RECOMMENDATIONS 15 7. REFERENCES 16 8. ACKNOWLEDGMENTS 17 APPENDIX* A. B. C. D. E. CALCULATIONS DEVICE SCHEMATICS DEVICE PICTURES INNOVATION WORKBENCH RESULTS DESIGNSAFE REPORT * Click on the hyperlink to view the above appendices. 2 1. ABSTRACT Coronary artery disease is the single leading cause of death in the United States today. Coronary Artery Bypass Grafting (CABG) utilizes a vessel to carry blood around an obstruction in the coronary artery. The two main types of bypass surgery that are currently being used are conventional (arrested heart) surgery through use of a heart-lung machine and beating heart (off-pump) surgery. Our design goals concentrate on improving the off-pump procedure. First, we improved the design of the suction foot for the localized stabilizing device. In this novel design, we augmented the lateral stabilization of the grafting site. Additionally, we present a device that will aid in the attachment of the harvested vessel to the coronary artery. Second, we focused on a device that would eliminate or reduce the number of sutures required to attach the two vessels together using BioGlue® from Cryolife, Inc. The intravascular vessel connector anastomosis device consists of a hollow, semi-cylindrical base with a hollow stem attached at 45o. In this revolutionary technique, the upper shaft is inserted into the vessel to be grafted and secured with BioGlue®. or sutures placed in a circular fashion. The device is then inserted into an incision in the coronary artery, with the base designed to be longer than the incision. Once the device is inserted, an outward force is applied to stabilize the two vessels in close proximity so they can be easily sutured or joined with BioGlue®. Our localized stabilization device consists of two suction fingers with two chambers each. The base is mounted on a track, which expands up to 1cm via a turnbuckle mechanism located on the posterior end of the device. The device will be attached to the Ethicon’s FLEXSITE® stabilizer arm which allows it to be locked in place on the surface of the heart, around the coronary artery. The turnbuckle is then rotated away from the surgical field to spread the fingers of the device and introduce lateral stabilization of the grafting site. The efficiency of CABG will be greatly improved by integrating our vessel connector device, localized stabilizer device, and BioGlue® to reduce the complexity, difficulty, and time required to perform CABG, thus benefiting both the surgeon and the patient. 2. INTRODUCTION 2.1 OVERVIEW OF CORONARY ARTERY BYPASS GRAFTING Seven million Americans suffer from coronary heart disease, the most common form of heart disease. Coronary artery disease is the single leading cause of death in the United States today (1, 2). More than 95% of all coronary artery disease is due to arteriosclerosis, clogging of the coronary arteries by cholesterol or calcium deposits (3). The coronary arteries deliver a constant flow of blood to the heart muscle providing it with a necessary supply of oxygen and nutrients. When these arteries narrow or clog, they cannot provide adequate blood flow to the heart, resulting in coronary heart disease, which includes myocardial infarction (heart attack) and other diseases. Treatments for coronary artery disease include medication, angioplasty, and surgery. Medications used to treat coronary artery disease include blood thinners and drugs that decrease the amount of work required of the heart muscle by lowering pulse rate and blood pressure. Angioplasty is an invasive procedure that uses catheters introduced from the groin to 3 work on the inside of the coronary arteries. Angioplasty procedures include the use of stents, balloons, rotoblators, athrectomies, and lasers, to remove clots and increase blood flow within the coronary arteries. The major type of surgery used to treat coronary artery disease is coronary artery bypass grafting (CABG). Despite advances in the non-surgical treatments, there are many situations in which surgery is necessary and beneficial (4). The history of CABG began in 1876 when it was established that chest pain could be attributed to the interruption of coronary blood supply and that heart attacks occurred when at least one coronary artery becomes blocked (5). In 1910 Alexis Carrell presented a paper to the American Surgical Association describing CABG, and in 1950 the first myocardial revascularization was performed by Arthur Vineburg (6, 7). By 1968 Dr. Rene G. Favolaro had achieved restoration of coronary blood flow in 171 patients using CABG while working at the Cleveland Clinic (8). Today, 350,000 CAGB surgeries are performed in the United States each year with 600,000 performed worldwide (1). The CABG procedure utilizes a vessel to carry blood around the obstruction in the coronary artery. The vessels used for CABG are the saphenous vein from the leg or the internal thoracic artery from the inside of the chest. During the procedure, an incision is made down the front of the chest through the sternum exposing the heart and aorta. This incision is called a median sternotomy. The saphenous vein is attached at one end to the aorta and at the other end to the coronary artery downstream of the blockage. When using the internal thoracic artery, it is only disconnected at one end, and that end is then attached to the coronary artery downstream of the blockage (9). The average patient can leave the hospital approximately six days after the procedure, if there are no major postoperative problems. Patients normally take another two to three weeks to regain their strength and normal body habits, including appetite, sleep patterns, etc. (10). 2.2 CURRENT TREATMENTS The two main types of bypass surgery that are in use are conventional (arrested heart) surgery and beating heart (off-pump) surgery. Typically, a patient who has undergone conventional bypass (on-pump) surgery is discharged from the hospital several days after surgery. According to the year 2000 Medicare claims data (11), the average length of hospitalization for CABG surgery was approximately 8 days. During conventional bypass surgery, patients are placed on a heart-lung machine, which oxygenates and pumps the blood. When the patient is placed on the “bypass” heart-lung machine, the blood is routed outside of the body through the heart-lung machine. During this process, the patients may require transfusions to replenish blood volume, red blood cells, or platelets. In a recent study, it was found that more than half of the patients receiving the conventional CABG surgery required some blood products (12). Arrested heart bypass surgery also requires considerable resources in terms of number of staff, products and equipment, medications, and other items that will impact the total cost of the procedure. It should also be noted that in the medical literature, surgeons refer to “high risk patients”-patients whom they consider poor candidates for conventional bypass surgery because they are too ill, or have preexisting medical conditions that make exposure to the heart-lung machine too risky. These “high risk patients” may include the elderly, and those with renal problems, diabetes, previous history of strokes or heart attacks, etc. Patients who are unable to take part in the conventional bypass surgery may 4 still undergo successful CABG surgery via a method known as beating heart bypass surgery. Today’s evolving surgical innovations allow surgeons to safely and effectively suture coronary artery bypass grafts in place on the surface of the heart without hindering its ability to beat and pump blood. These innovations have not fully replaced the conventional CABG procedures, but they are being increasingly used, as further technologies are developed to support them. With beating heart bypass surgery patients may be discharged from the hospital more quickly. A recent report in the medical literature noted a 32% shorter length of hospital stay for off-pump CABG patients (13). The beating heart bypass surgery is performed on a beating heart through an incision down the middle of the chest. Positioning and stabilization instruments lift and hold the heart and then stabilize a portion of the heart’s surface where the bypass graft will be sutured in place, all as the heart continues to efficiently beat and pump blood. Some stabilizers use gentle suction to address the primary challenge in beating heart surgery, making it possible to access all surfaces of the heart while reducing motion of the small area of the surface tissue where the surgeon is sewing the bypass graft. For this procedure, the heart-lung machine is not used. However, it should be noted that on occasion, a surgeon might convert to the use of the heart-lung machine during the procedure if the patient’s condition becomes unstable. One of the benefits of using the off-pump CABG surgery method is the reduction in use of blood products that would otherwise be required if the heart-lung machine was utilized, thus fewer patients require transfusions when undergoing beating heart surgery. This is an important consideration for areas where banked blood may be in short supply or where transmission of bloodborne diseases is a concern. A study in the New England Journal of Medicine by Newman, et al., found measurable and persistent neurocognitive decline (i.e. memory loss, decline in thinking skills) in patients who had conventional arrested heart bypass surgery utilizing the heart-lung machine (13). In two separate studies, postoperative neurocognitive function test scores were significantly better in the groups of beating heart surgery patients than in the groups who underwent conventional CABG surgery (14, 15). Beating heart surgery potentially costs less than conventional surgery because cardiopulmonary bypass equipment is not used, and fewer blood products are needed. Another study found a cost reduction per patient in groups that underwent off-pump CABG surgery. This cost reduction was directly correlated to shorter ICU use and hospital stay. It also showed significant reduction in the need for blood products and in postoperative complications (16). Beating heart surgery, which avoids the use of the heart-lung machine, may make it possible for “high-risk patients” to have bypass surgery. Lastly, a study by Arom, et al., found that off-pump CABG surgery carries a significantly lower mortality rate in the high-risk population than conventional CABG surgery (17). 2.3 DESIGN GOALS The main objective of this design process was to find ways to improve the current off-pump CABG surgery procedures. Through discussions with various surgeons, engineers, and university faculty members, we identified the two bottlenecks that hinder control CABG efficiency. One bottleneck was associated with insufficient stabilization at the anastomosis site. The other, more pervasive, bottleneck dealt with the difficulties 5 related with the suturing of a harvested vessel to a coronary artery. The degrees of freedom associated with effective stabilization increase dramatically if the suturing process is either made easier for the surgeon or eliminated fully via using a vessel connector anastomosis device. The goals of our “out of the box” approach to this design were to provide effective vessel stabilization, simplify cardiac positioning, and minimize hemodynamic deterioration. Small size, strength, and flexibility are the general parameters that our group plans to work with for this next generation model. Our product will provide stabilization and greater access to the coronary arteries, simplifying cardiac positioning. Currently, surgeons have been using various heart stabilizing systems made by Guidant Inc., Medtronic Inc. and Ethicon Inc. These stabilizers are designed to address some of the significant challenges a heart surgeon may face while performing beating heart bypass surgery. These challenges include preserving the natural hemodynamic function of the heart as it is held and positioned for access to hard-to-reach lateral and posterior vessels, and then stabilizing it for precise suturing of the bypass graft. These stabilizers also offer improved visualization and less movement of the surgical site while still letting the heart pump blood normally. To use the stabilizers, one must first use a heart positioner. The positioner is a single-use, retractor-based device that features a silicone multi-appendage suction cup, an articulating arm and a mounting clamp that is attached to the chest spreader. The suction cup conforms to the surface of the heart at the apex. This way, the surgeon can position and hold the beating heart to ensure access to the coronary arteries that require bypassing, including those located in the posterior section of the heart. As soon as the heart is positioned correctly, the surgeon can then place the foot of stabilizer parallel to the target artery to reduce motion at the site where suturing of the bypass graft will occur. Stabilizers come equipped with some feet that use suction, and some that rely on mechanical stabilization alone. Stabilizer feet that use a vacuum, utilize a pull tension to keep the grafting site stable but fall prey to losing contact with the surface of the heart if one of the vacuum chambers should come loose. Conversely, the feet that do not make use of the vacuum employs a push tension that helps keep the anastomosis site from moving. Once the heart is stabilized adequately, the surgeons must then address the challenges of creating and maintaining a bloodless field and providing continuous perfusion to the heart muscle throughout the procedure. We felt that for this project, we could introduce an innovative design of the suction foot for the stabilizing device. For this novel design, we hoped to improve the lateral stabilization of the grafting site. In order to accomplish this, we hoped to introduce a turnbuckle mechanism, which would spread the tissue one centimeter further. We also set out to improve the vacuum system. By implementing a new vacuum mechanism, vacuum suction in the foot of the device would remain in contact with the tissue despite losing one of the vacuum lines. As a group, we decided that designing a device to aid in the attachment of the harvested vessel to the coronary artery could be beneficial to the surgeon during bypass surgery. We had to make certain that the device would eliminate the suturing process or reduce the number of sutures required to attach the two vessels together. In addition, the device needs to be made from a biocompatible material that would withstand the pressures associated with a beating heart. The device needs to be small enough to be 6 inserted into a coronary artery and must not impede blood flow between the vessels. The edges of the device must be rounded so as not to puncture the inner wall of the vasculature. Finally, a BioGlue® made by Cryolife, Inc. may be used along with the device to help in the attachment with the coronary artery. If the BioGlue® attachment is successful, the need for sutures will be eliminated. 3. METHODOLOGY 3.1 TIMELINE Our project, improving coronary artery bypass surgery, was done between the months of December 2002 and April 2003 as shown in Table 1. Research on the procedure and possible ways to improve the off-pump CABG surgery began in December. We consulted doctors and engineers and observed both on-pump and offpump surgeries. After research and consultations, some ideas for possible improvements were put together and feedback was obtained from advisors. In mid-January, the methods for improvement were finalized and the preliminary designs for the anastomosis device and improved stabilizer foot were delineated. The month of February marked the beginning of prototyping. After the initial prototypes were built, the decision of the final material to be used in the completed designs of both of our devices was made. By March, preliminary testing of the crude models was begun. Additionally, research and design modifications were conducted to further tweak the design of both the anastomosis device and improved stabilizer foot. The project was completed in mid-April leaving time for completion of the final report and poster presentation. Table 1: Project Timeline December January February March April Research Advisor Feedback Material Decision Design Prototype Testing 3.2 DESIGN AND PROTOTYPE The design problem originally asked the team to “think out of the box” in order to develop a more effective stabilization system. We began by acquainting ourselves with the terrain of the chest cavity during a beating heart CABG surgery. We attended two beating heart CABG operations and observed a standard on-pump procedure, paying special attention to the cardio-thoracic surgeons’ hand movements and taking note of how he interacted with the stabilization system and the beating heart system as a whole. We consulted the Journal of Cardio-thoracic Surgery regarding beating heart surgery and read numerous related journals in this research stage. Once we successfully grasped the 7 subject matter, we consulted with surgeons and their assistants on the existing problems that plague current stabilizers. Brainstorming sessions were held regularly and were controlled by mandating the introduction of at least three ideas from each group member. An engineering approach to the problem involved logical assumptions. We wanted to stop the motion of a patch of cardiac tissue that is about or 4.5cm x 2cm (this is the anastomosis site). There were five ways that we could have approached this problem, or various combinations of the five together could work to stabilize cardiac tissue locally for optimal performance. 1. Pushing on the site from either the outside or the inside of the heart to inhibit motion (balance the forces and moments). 2. Pulling on the site from either the outside or the inside of the heart to inhibit motion (balance the forces and moments). 3. Localized chemical methods (injection or other even less invasive means) that would act on a small area of the heart to inhibit motion. 4. Localized electrical inhibition—something similar to shunting around a Purkinje fiber, if that’s at all possible in the heart… 5. Apparent— motion is a perception of the mind—we would utilize a strobe light or some other device that would apparently stabilize the anastomosis site. Eventually, our sessions with engineers and consultations with faculty were instrumental in identifying the two essential bottlenecks associated with beating heart CABG surgery. The most obvious of these is the problem of insufficient stabilization. However, a less obvious bottleneck is the lack of suturing efficiency. We reasoned that either reducing the number of sutures or eliminating the suturing process by replacing it with a user-friendly and efficient method would simultaneously handle both bottlenecks. The need for increased stabilization hinges on the difficulty associated with the suturing process; therefore, replacing suturing with an adhesive based procedure that relies on a vessel connector would alleviate the need for more effective stabilization than current devices provide. Once we realized that a vessel connecting anastomosis device needed to be designed, we held brainstorming sessions to design a novel anastomosis device. The meetings resulted in the evolution of the device toward the final design presented in this paper. PVC and clay models were constructed during meetings to provide visual feedback to team members. Considerations during the design phase included: minimal occlusion of the blood flow, biocompatibility, strength of materials, comfortable stent angles with respect to the heart surface, damage to vessel walls, lateral suturing, and assurances that the harvested vessel would remain fastened to the stent. Originally, our base was a flexible oval shaped thin polyurethane material that would permit suturing. Eventually, we decided that an extended base that was cylindrical along its longitudinal axis would not occlude the flow and adhere to the vessel wall with greater ease. We also discussed creating the device out of a bio-absorbable material, but this option was dismissed due to its tendency toward forming emboli. The reason that Polyetheretherketone™ (PEEK™) was chosen as the primary polymer for the vessel connector can be attributed to PEEK™’s ability to withstand a significant amount of tensile and compressive stresses; furthermore, PEEK™ is biologically inert. Ideally, the 8 second bottleneck would also be handled with the introduction of an efficient adhesive that would replace the suturing via concurrent use with the vessel connector; Cryolife’s BioGlue® assumes this role in our ideal effective beating heart CABG system. After handling the primary bottleneck, we chose to improve the stabilization by refining the current turnbuckle based foot model. This included identifying the main problems in the current foot design and then replacing them with improvements. Therefore, we introduced a parallel port vacuum system that would not falter during surgery as the current series port vacuum system does. Lateral tautness was emphasized to increase local stabilization. An easy access turnbuckle system on the posterior end of the stabilizer foot would provide surgeons the means to increase lateral tension by spreading the cardiac tissue beneath the suction. Another advantage to this novel design was an introduction of a degree of freedom with respect to the width of the view window; the turnbuckle mechanism provided a method of adjustment for the view window width. 3.3 TESTING Foot Design Pressure Approximation We began on the assumption that the anastomosis site would be 3.5cm by 2cm (symbolically l h ). Moreover, the heart is simplified as a fluid filled sphere, of a diameter d, possessing a wall thickness t. The pressure of the fluid is FL and distributes over the inner surface of the beating heart. The muscular walls of the simplified heart exert a force to counter the beating motion of the heart; this force will be denoted as Fm, which is correctly associated with a resistive material force that occurs in similar systems. The pressure associated with the resistive muscle force (σh) is the Hoop stress in this problem. Solving for the Hoop stress: d 2 FL P 4 Fm dt h d 2 d 4 Pd h 4t Introduction of the stabilizer into the environment permits us to define a new force, the suction force that we desire (Fs) for sufficient stabilization. A pressure gradient exists across the heart wall during relaxation and contraction; where the pressures associated with the former state are collectively P2 and the pressures associated the latter state are collectively P1. The force of the blood at the anastomosis site will be labeled Fp. A force balance is set up to determine an adequate Fs, where the inherent resistive muscle force is set equal to the sum of Fp and Fs: h dt P Fm 2l h t h F p l h P Fs Ps l h 9 2l ht h Pl h Ps l h Ps ~ 2P or approximately 240 mmHg therefore, this heart contact member is configured to facilitate the use of negative pressure to engage the surface of the heart. For images of simplified heart model, see appendix. Vessel Connector Anastomosis Device Material Calculations The surface area of the device is 196.1mm2 (1.961e-4m2). We shall approximate the device as a flat plane of the same surface area. Based on previous experimentation, the maximum pressure inherent in the beating heart system that will affect the fidelity of the device is 560mmHg or 7.46e4 N/m2. Using this upper pressure limit will permit us to compensate via both tensile and compressive strengths. The base will apply an axial force against the wall of the vessel, at an angle vertical to the longitudinal axis of the protruding, beveled shaft. Moreover, the base is designed to apply a radial force at a horizontal angle transverse to the shaft’s longitudinal axis, against the wall of the target vessel, to secure the device to the target vessel. 4. RESULTS 4.1 ANASTOMOSIS DEVICE DESIGN The vessel connector design, as seen in Figure 1, serves as an anastomotic stent for connecting a graft vessel to a target vessel, particularly for use in coronary artery bypass grafting. The design comprises a small vessel anastomotic stent for use on a target vessel, which has a small diameter roughly equivalent to that of the coronary artery. The base of the device consists of a semicircular cylindrical piece with a diameter of 2mm and length of 10mm. The wall thickness is 0.1mm. Along the 10mm side of the base, the edges are rounded at a radius of 1.5707mm. A stem, 2mm in diameter, is attached to the base at an angle of 45o relative to the base. A hole passes from the cylindrical stem to the base. Figure 1. Anastomosis Device At the attachment point between the base and stem, the edges are rounded with the 135o attachment sloped to the base at an inverse radius of 3mm and the 45o attachment point sloped to the base at an inverse radius of 0.6mm. This allows for a smooth attachment of the harvested vessel to the coronary artery. The stem is a hollow cylinder with a 2mm diameter and 0.1mm wall thickness. The stem’s length is approximately 10mm. On the stem are two rings with semicircular cross-sections 1mm in diameter. The rings are separated by approximately 2mm, midway down the stem and slope to the surface at an inverse radius of 1mm. The area between the rings provides a site for securing the harvested vessel to the device with 10 lateral sutures. At the top of stem is a ring, also with a cross-section of 1mm that slopes to the surface of the stem at an inverse radius of 3mm. The purpose of this ring is to create a seal to prevent blood flow from escaping outside the device to the thoracic cavity. All of the edges of the device are rounded so as not to puncture the internal wall of the vasculature. The shaft will be inserted into the vessel that is to be grafted onto the coronary artery and secured in place using BioGlue® or suture. The suture will be placed in a circular fashion around the vessel between the raised beveled rings. This will prevent the vessel from detaching from the device. Once this vessel is securely attached, the semicircular base will be inserted into the coronary artery. The base is designed to be longer than the length of the incision so that it will not slip out. Once the device is inserted into the coronary artery an outward force, perpendicular to the artery surface, will be applied to the device so that the base will be brought into contact with the upper inside surface of the coronary artery. The two vessels are now stabilized in close contact with one another and can be easily sutured together or attached with BioGlue®. 4.2 IMPROVED CABG LOCALIZED STABILIZATION DEVICE The improved localized stabilization device, as seen in Figure 2, includes a modification to the suction foot stabilizer produced by Ethicon, Inc. Its main advantage over existing models is that it provides improved lateral stabilization to the site of anastomosis. The chief addition is a turnbuckle to the posterior end of the suction foot to allow the foot to be spread up to approximately one centimeter beyond the initial location. The turnbuckle is spread using a finger or thumb and can be easily rotated with gloved surgeon hands. Moreover, due to the threaded nature of the Figure 2. Localized Stabilization Device turnbuckle, once in place, the feet will remain in their location until the turnbuckle is reversed. The turnbuckle will allow the foot to expand along a track an additional centimeter beyond its initial configuration. The turnbuckle has a silicone covering to allow easy movement by the gloved, surgeon’s hand. The localized stabilizer foot device will attach to Ethicon’s FLEXSITE® arm for use in local stabilization of the surgical site during coronary artery bypass surgery. When attached to the FLEXSITE® arm, the foot can be rotated and swiveled nearly 360 o in two planes. Once placed in the desired position, the grip of the stabilizer arm is released, which releases a cable that tenses both the arm and the foot in place. The entire FLEXSITE® Stabilizer becomes rigid including the position of the foot. The current design of Ethicon’s suction foot incorporates a malleable base with suction areas on both fingers of the foot. Once placed on the heart, the foot cannot be manipulated or adjusted. The suction pulls the tissue away from the heart’s surface to stabilize the site. In reference to the suction system, Ethicon’s current suction foot 11 utilizes two suction cells on both of the suction fingers for a total of four suction cells. A common 5mm silicone surgical tube feeds the foot from a vacuum source collection reservoir. This vacuum tube splits into two 2mm tubes that feed each of the fingers of the foot. A small, 0.5mm tube made into the inside of each finger supplies each of the suction cells of the foot. An additional improvement is the addition of an improved vacuum system. This improvement will eliminate the current system’s problem where the entire device becomes dislodged when one cell looses tissue contact. The reason for this problem is that the vacuum lines feeding each of the cells have a high resistance when attached to the tissue. When one cell becomes dislodged, its resistance drops severely which causes the vacuum to pull most of its suction through its line. This greatly reduces the suction created in the other cells, and they dislodge. Our system will have each of the cells fed by an independent vacuum line from the vacuum source collection reservoir. This “sink” will allow each of the cells to have a constant suction even if one of the other cells dislodges from the tissue. An analogy can be drawn between the old system and a circuit with a voltage source and four resistors, of equally high resistance connected back to the voltage source. When one of the resistors is changed to low resistance, most of the current flows through that resistor. Our design can be compared to a voltage source connected to four resistors in parallel and then grounded. The current flowing through each of the resistors is constant regardless of the resistance of the other resistors. Our design will assure a stable surgical field even if one of the suction cells becomes dislodged. Our design maintains the low-profile nature of the current suction system and continues to provide the high visibility boasted by the Ethicon’s current foot device. Moreover, since our foot fits on to Ethicon’s current FLEXSITE stabilizer arm, surgical setup is easy and fast. Our design can be easily integrated into Ethicon’s system and has the potential to join Ethicon’s line of foot designs 4.3 MATERIALS Anastomosis Device Material The lower limit of Polyetheretherketone’s™ (PEEK™) tensile strength rests at 70MPa or 52.5e4mmHg. Indicating that PEEK™ is not reinforced will suffice for the vessel connector, but the degrees of freedom increase dramatically with carbon fiber reinforcement. With 30% carbon reinforcement the connector would experience optimum wear resistance and load carrying capability (90e4mmHg). The following calculation indicates that our device, composed of PEEK™, demonstrates a tensile strength well above the 560mmHg pressure constraint: 52.5e4 mmHg/ 560 mmHg = 937.5 therefore, our design should be able to withstand nearly a thousand times more tensile strength than the pressures the CABG environment could generate. PEEK™ polymer is extensively used in medical applications. It combines strength, purity, chemical resistance and ease of processing with superb sterilization resistance and a lack of interaction with biological systems. Moreover, PEEK™ is 12 widely regarded as the only material, which is easily processed and has the ability to be steam-sterilized repeatedly without a significant reduction in physical properties. Based on the cost of PEEK™ (approximately $90 US/kg) at a density of 1.26g/cm3, the material costs associated with production of 1000 devices (at 20.22mm3 volume) amount to approximately $2.80. Table 2: Polyetheretherketone™ Properties Tensile (73 °F) Strength Flexural (73 °F) Strength Pa Pa Compressive Strength (10% Deflection) Pa 9.99e7 1.70e8 1.17e8 Shear Strength (73 °F) Limiting PV Pa Pa/FPM 5.30e7 1.38e8 Cryolife’s BioGlue® Adhesive The glue is of high strength, stronger than sutures, and forms a non-toxic bond in 30 seconds or less. Wet conditions do not impede the formation of the bond. The compositions provide strong and rapid bonding to a range of substances of both natural and synthetic origin. BioGlue® provides rapid bonding to vascular and cardiac tissues, but strong bonds are also formed to leather, rubber, Dacron®, Teflon®, as well as metals. This enables the adhesive to be used for the attachments of surgical grafts and devices, as well as for wound closure, trauma repair, and hemostasis. This particular combination of properties is ideally suitable to the CABG environment (18). The adhesive compositions are the results of cross-linking on a surface or surfaces to be bonded of a mixture comprised of - Part A.- a water soluble proteinaceous material of about 27-53% by weight of the mixture; Part B.- di- or polyaldehydes present in a weight ratio of one part by weight to every 20-60 parts of protein present by weight in the mixture and water, optionally containing non essential ingredients to make up the balance of the composition. The final cross-linked bonding compositions are water insoluble, rubbery or leathery proteinaceous solids substantially free of aldehydes, and adherent to the substrate to be bonded with a tear strength of at least 75g/cm2. Bonding is accomplished by combining the two-part system (the parts being referred herein as Part A and B, respectively), and allowing the mixture to react on the surface or surfaces that are being joined. Rapid bond formation follows, generally requiring less than one minute to complete. The resulting adhesion is strong, generally providing bonds with tear strengths of 400-600g/cm2. Tear strengths of 1300g/cm2 have been recorded (18). Localized Stabilization Device Materials Based on previous experimentation, the maximum pressure inherent in the beating heart system that will affect the fidelity of the device is 560mmHg or 7.46e4N/m 2. Metals, which are biocompatible and meet the strength requirements for this application include: titanium (tensile strength of 2.76e8N/m2 at room temperature), tantalum (tensile strength range from 2.41e8N/m2 to 4.83e8N/m2 at room temperature), and surgical stainless steel (4.48e8N/m2). Superelastic materials such as nickel titanium alloys are reasonable materials to use in the foot as well. 13 4.4 SAFETY ANALYSIS Throughout the entire design process, all possible safety problems that might arise were considered. To aid in this process, we used the Designsafe™ program to help us limit or solve all the possible problems. We determined that there might be some safety issues regarding the stabilizer foot. One of the problems that could arise is the possibility that the stabilizer foot could cause a tear in the tissue of the heart when it spreads. To overcome this, we designed the foot to only spread a distance (~1cm) that would not overexert the tissue to a point where it would tear. The other main safety problem with the stabilizer foot is the vacuum system. The older version of the device had four chambers in which the two pairs of chambers were essentially connected in series with one vacuum tube each. Therefore, if one of the vacuum tubes malfunctioned, then the others would loose suction and contact with the heart’s surface would be lost affecting the heart hemodynamics and suture insertion during the surgical procedure. In order to bypass this problem, we designed a stabilizer vacuum system in a parallel fashion in which each of the four chambers is fed by a separate vacuum tube that stems from the main vacuum source. The anastomosis device presented us with more challenging safety concerns. The device was to be used invasively and thus the patient’s health was our first priority. We make certain our final design did not possess any sharp edges, which could puncture the inner wall of the vasculature. Another issue with the anastomosis device was biocompatibility. In order to use this device, it had to be compatible with the patient’s body. After researching possible materials, we decided that the use of PEEK™ would be our best option, but we have not excluded the possibility of using titanium, stainless steel or tantalum. PEEK™ as well as the other materials would still require extensive testing before being used in surgical practices. PEEK™ was chosen for the following properties: high tensile and flexural strength, high fatigue limit, high impact strength, low flammability, and its inability to interact with a magnetic field. The use of PEEK™ also addresses the potential problem of the device breaking and causing an embolus. Due to PEEK’s™ superior tensile strength and high fatigue limit, the forces incurred in the heart would not be sufficient to break the vasoconnector device. The task of proper placement of the anastomosis device in the heart would fall on the surgeon. It is his or her responsibility to have the proper training with the device to use it correctly. We designed this device on the assumption that the surgeons’ who use it have learned the proper protocol on installing the device. We feel that the design of the device is very straightforward and user friendly and should not pose any significant problems to the user. 4.5 ECONOMIC ANALYSIS If we use PEEK™ as the polymer of choice for our vessel connector device, we can determine a fairly accurate cost of making our device. We found that PEEK™ has a cost range from $90-$110 per kg and a density of 1.26g/cm3. Using these figures and the fact that our device has a volume of 20.22mm3, the material cost of making 1000 of the vasoconnectors is approximately between $2.30 and $2.80. It is very inexpensive to 14 make a great quantity, due to its very small size. It must be noted that there could be significant costs in the development and testing of the device to ensure proper function. As of now, we only have a scaled up (10x) prototype of our anastomosis device built. In some places, this prototype would cost up to $360. Working through Vanderbilt University and the NCIIA, we had our prototype built by the University of Pittsburgh. When this device nears its final stages of development, more resources would be needed to conduct rigorous analyses of the structural integrity and performance characteristics of the device. For testing purposes we would need to get FDA approval to conduct trials with animal specimens before the device is to be utilized in humans. With 150,000 off-pump CABG surgeries performed worldwide every year, there is a significant need for this device. The device is inexpensive to produce and easy to use. It may eliminate the need for sutures and make the surgical procedure less difficult. Using this device might also make the CABG procedure less expensive. 5. CONCLUSION We have successfully designed an anastomosis device that will reduce or eliminate the suturing required to perform the CABG procedure by providing a simple, quick and effective method to bring two vessels in close proximity to one another. Elimination of suturing during the procedure can be achieved through use of BioGlue® in conjunction with our vessel connector device. We have also developed an improved foot stabilizer device enhances lateral tension and provides a superior suction system to provide greater local stabilization at the anastomosis site. The efficiency of CABG will be greatly improved by integrating our vessel connector device, our foot stabilizer device, and BioGlue® to reduce the complexity, difficulty, and time required to perform CABG, thus benefiting both surgeon and patient. 6. RECOMMENDATIONS Future Testing (Materials and Methods) The anastomosis device should eliminate or reduce the amount of suturing during the anastomosis procedure via cooperation with the novel stabilizer foot design and Cryolife’s BioGlue®. BioGlue® is an adhesive composition comprised of cross-linked proteinaceous material. In Vitro Either bovine or swine hearts must be procured and the coronary arteries should be rinsed with saline-heparin solution. Segments of human saphenous veins will be used as the conduit vessels. Anastomoses will be created. In brief, the saphenous vein and the left anterior descending coronary (LAD) are to be positioned in close proximity. The BioGlue® adhesive should be applied via its standard applicator gun and tip. The saphenous vein will be slipped over the beveled shaft of the vessel connector/anastomosis device and more BioGlue® will be applied to the proximal region of the shaft where the saphenous vein meets the artery surface. After a sufficient period of time (30 seconds ensure 65% bonding strength) has passed, an incision of reasonable size is to be created in the coronary recipient, the vessel connector base will be inserted into the artery and 15 pulled outwards in order to ensure contact between the device base and the artery wall. The proximal end of the vein graft, as near to the device as possible, should be connected to a pressure-transducing box. Once flow through the vein graft is established via saline injection, the vein graft and the coronary artery will be perfused with a large syringe connected in a Y fashion to the proximal vein graft and the pressure transducer, and saline solution will be forcefully injected to simulate a pressure of at least 300mmHg (19). In Vivo After about 50 anastomoses, in vitro, are conducted, a long-term animal model needs to be established. Yearling goats or mongrel dogs are possible experimental animals. The left anterior descending coronary artery should be dissected and proximal and distal sites of the future arteriotomy will be occluded via elastic snares. Direct anastomosis will be performed between the LITA (left internal thoracic artery) and a 5mm coronary arteriotomy on the beating heart; the novel localized stabilization foot design detailed above will provide localized stabilization that will promote a comfortable working environment for the surgeons. The ITA (internal thoracic artery graft) will be fitted over the vessel connector shaft in the same manner as was detailed in the in vitro section. Five cc BioGlue® will applied and allowed to dry for 2 min. The distal elastic snare should be partially lifted to allow slow progressive retrograde filling of the vessels with some extra vascular oozing. The distal elastic snare must then be removed and the ITA-graft opened. Definitive proximal occlusion of the LAD upstream will exclude competitive flow from the native coronary (20). Additionally, we would have to perform extensive stress and material testing of the anastomosis device and foot and investigate other possible materials for the anastomosis device. We could also explore other possible vessel connector designs or improvements and obtain further feedback from doctors. 7. REFERENCES 1. “Coronary Heart Disease.” 2002 Heart and Stroke Statistical Update, Dallas, Texas. American Heart Association, 2001. http://www.americanheart.org, 2. “Facts About Coronary Heart Disease.” National Institutes of Health. http://nhlbi.nih.gov, (27 July 2001). 3. “The Heart and Coronary Artery Disease.” Hall-Garcia Cardiology Associates. http://www.hgcardio.com, (27 July 2001). 4. http://www.heartpoint.com/treatcoronaryartdis.html 5. Westaby S, Landmarks in Cardiac Surgery. Isis Medical Media, Oxford, 1997. 187 6. Ibid., 230 7. Borst, H & Mohr, F, History of Coronary Artery Surgery – A Brief Review; J Thorac Cardiov Surg 2001; 49: 195-198 8. Ibid, 196-197 9. http://www.sts.org/doc/3705 10. http://www.enter.net/~fsadr/cabg.htm 16 11. Hanvik B., Public Relations, Medtronic, Inc. Minneapolis, Minnesota. 12. Puskas JD, et al. Clinical outcomes, angiographic patency, and resource utilization in 200 consecutive off-pump coronary bypass patients. Annals of Thoracic Surgery 2000;71:1477-1484. 13. Newman, et al. Longitudinal assessment of neurocognitive function after coronary-artery bypass surgery. New England Journal of Medicine 2001;344(6):295-402. 14. Diegler A, et al. Neuromonitoring and neurocognitive outcome in off-pump versus conventional coronary bypass operation. Annals of Thoracic Surgery 2000;69:1162-1166. 15. BaskerRhao B., et al. Evidence for improved cerebral function after minimally invasive bypass surgery. Journal of Cardiovascular Surgery 1998;13:27-31. 16. Boyd W., et al. Off-pump surgery decreases postoperative complications and resource utilization in the elderly. Annals of Thoracic Surgery 1999;68:14901494. 17. Arom K., et al. Safety and efficacy of off-pump coronary artery bypass grafting. Annals of Thoracic Surgery 2000;69:704-710 18. U.S. PATENT DOCUMENT 5,385, 606 A 1/1995 Kowanko 19. Gundry, S. R., Black, K. and Izutanii, H. Sutureless artery bypass with BioGlue® anastomoses: preliminary in vivo and in vitro results. Journal of Thoracic and Cardiovascular Surgery, 2000, 120, 473–477. 20. Van Nooten, Y. Van Belleghem, L. Foubert, K. François, F. Caes, H. Van Overbeke and Y. Taeymans, An experimental model of coronary anastomosis without suturing. Cardiovascular Surgery, 2003, 11, 80-84. 9. ACKNOWLEDGMENTS We would like to thank the following people for all their help in our design project. Without their help, most of this would not have been possible. Thomas Ryan, PhD Paul King, PhD, PE Jia Hua Xiao, PhD Walter Merrill, MD James Greelish, MD V. Anilkumar, PhD E. Duco Jansen, PhD Carol Rubin, PhD Robert J. Bayuzick, PhD Bruce Hoagland, MBA Randall Ryan 17