* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Anticaog_update_R_Maclean
Prescription costs wikipedia , lookup
Neuropharmacology wikipedia , lookup
Electronic prescribing wikipedia , lookup
Drug interaction wikipedia , lookup
Pharmacokinetics wikipedia , lookup
Adherence (medicine) wikipedia , lookup
Theralizumab wikipedia , lookup
Dydrogesterone wikipedia , lookup
Pharmacogenomics wikipedia , lookup
Discovery and development of direct Xa inhibitors wikipedia , lookup
Discovery and development of direct thrombin inhibitors wikipedia , lookup
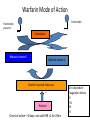
Anticoagulation Update Rhona Maclean [email protected] By the end of this session you will know: • What anticoagulant therapies are currently available • What anticoagulants can be used in particular clinical situations • Practical tips in using anticoagulants – Starting/ switching/ procedures • Current STH pathway for management DVT/PE Available Anticoagulant Therapies Heparins Vitamin K Antagonists NOACs Unfractionated Heparin Low molecular weight heparin Warfarin Dabigatran Fondaparinux Phenindione Sinthrome Rivaroxaban (Acenocoumarol) Apixaban Heparin and Fondaparinux Immediate onset of anticoagulation Warfarin Mode of Action Prothrombin Prothrombin precursor ---- Carboxylase Reduced vitamin K Oxidised vitamin K Vitamin K epoxide reductase Warfarin Onset of action- >5 days; not until INR >2 for 24hrs Vit K dependent Coagulation factors FII FVII FIX FX Mode of Action NOACs Novel Oral Anticoagulants (NOACs) Dabigatran Rivaroxaban Apixaban Half life 12-17h 5-13h 9-12h Administration Oral Oral Oral Renally excreted? +++ +/- +/- Heparin Induced Thrombocytopenia - - - Osteoporosis - - - Side effects Dyspepsia in 8-10% - - Monitoring? No No No Dietary/ drug interactions + + + Reversal agent? ? ? ? Drug Interactions with NOACs Increase anticoagulant effect Dabigatran Rivaroxaban Apixaban Amiodarone (caution) Azole Antimycotics Azole Antimycotics Verapamil (caution) HIV Protease Inhibitors HIV Protease Inhibitors Azole antimycotics Dronaderone Dronaderone Rifampicin Rifampicin Rifampicin Carbamazepine Carbamazepine Carbamazepine Phenytoin Phenytoin Phenytoin Phenobarbital Phenobarbital Phenobarbital St John’s wort St John’s wort St John’s wort Tacrolimus Cyclosporin HIV protease inhibitors Dronaderone Reduce anticoagulant effect Licensed and NICE approved indications for NOACs • Dabigatran: – VTE prevention after elective hip or knee replacement surgery – Stroke prevention in Atrial Fibrillation • Rivaroxaban: – – – – – VTE prevention after elective hip or knee replacement surgery Stroke prevention in Atrial Fibrillation Deep Vein Thrombosis treatment and secondary prevention Pulmonary Embolism treatment and secondary prevention Acute coronary syndromes • Apixaban: – VTE prevention after elective hip or knee replacement surgery – Stroke prevention in Atrial Fibrillation Risk of adverse events in patients with atrial fibrillation taking warfarin : Warfarin is an effective drug Optimal level of oral anticoagulant therapy Torn M et al, Arch Int Med 2009; 169:203-1209 Tips for achieving good warfarin control and avoid complications • Patient education and engagement • Encourage stable lifestyle • Minimise other drug changes/ choose drugs less likely to interact with warfarin • Use standardised induction regimen • Adjust warfarin dose cautiously (+/- 10% of weekly dose) when outside therapeutic range – Consider patient’s previous response • Don’t blindly follow CCDS (INR star) • Do INR if change of medication likely to interact or if unwell/ bleeding Unstable Warfarin Anticoagulation INR Results 20 16 12 8 4 0 09/09/2006 08/10/2006 06/11/2006 05/12/2006 03/01/2007 01/02/2007 03/01/2007 01/02/2007 Dosage 100 80 60 40 20 0 09/09/2006 08/10/2006 06/11/2006 05/12/2006 68 year old woman on warfarin for recurrent VTE INR 3.5 Rivaroxaban Warfarin Event rate/100 patient years Major and non major clinically relevant bleeding 14.9 14.5 Major bleeding 3.6 3.4 Critical bleeding 0.8 1.2 Intracranial bleeding 0.5 0.7 Clinical Benefit of Anticoagulants in AF Banerjee et al, T&H 2012 What anticoagulant to use in AF? • Warfarin – Pros • • • • • Experience Efficacious Long half life Reversal agent Use in renal failure – Cons • • • • • Requirement for monitoring Variable dosing requirements Not immediate onset of action Drug interactions/ lifestyle issues Bleeding Risk What anticoagulant to use in AF • NOACs – Pros • • • • • Immediate onset of action Once or twice daily medication- dosette boxes No (little) monitoring required Few drug interactions Risk major bleeding reduced, particularly intracranial – Cons • • • • New- as yet relatively unfamiliar Shorter half life- compliance issues? Lack of specific reversal agent- as yet… Cannot use in renal failure How to start NOAC in patient with AF • Counsel patient- it is an anticoagulant • Check baseline bloods (FBC, renal and liver biochem) • Start NOAC (Rivaroxaban with food- in morning?) • See patient for review in 3-4 weeks- any problems? • Check FBC, biochemistry, 3-12 monthly dependent on patient factors • Reinforce compliance whenever possible Which anticoagulant to use for VTE? • After a first VTE event, patients require minimum 3 months anticoagulation • If idiopathic or ongoing risk factors, consider longer term anticoagulant treatment (NICE) • Patients require immediate anticoagulation • If start warfarin, usually entails 8-15 anticoagulation visits in first 3 months of treatment EINSTEIN DVT and EINSTEIN PE pooled analysis: primary efficacy outcome Cumulative event rate (%) 3.0 Enoxaparin/VKA N = 4131 2.5 2.0 Rivaroxaban N = 4150 1.5 HR=0.89; p non-inferiority <0.0001 1.0 Mean time in therapeutic range = 61.7% 0.5 0.0 0 30 60 90 120 150 180 210 240 270 300 330 360 Time to event (days) Number of patients at risk Rivaroxaban 4150 4018 3969 3924 3604 3579 3283 1237 1163 1148 1102 1034 938 Enoxaparin/VKA 4131 3932 3876 3826 3523 3504 3236 1215 1149 1109 1071 1019 939 ITT population EINSTEIN DVT and EINSTEIN PE pooled analysis: major bleeding First major bleeding Cumulative event rate (%) 3.0 2.5 Rivaroxaban n/N (%) Enoxaparin/VKA n/N (%) HR (95% CI) p-value 40/4130 (1.0) 72/4116 (1.7) 0.54 (0.37–0.79) p=0.002 2.0 Enoxaparin/VKA N=4116 1.5 1.0 Rivaroxaban N=4130 0.5 0.0 0 30 60 90 120 150 180 210 240 270 300 330 360 Time to event (days) Number of patients at risk Rivaroxaban 4130 3921 3862 3611 3479 3433 2074 1135 1095 1025 969 947 499 Enoxaparin/VKA 4116 3868 3784 3525 3394 3348 1835 1109 1065 990 950 916 409 Safety population Rivaroxaban for VTE • Currently in patients with a first DVT without contraindications – Eg pregnancy, breastfeeding, renal failure, interacting drugs • 15mg bd for 3 weeks then • 20mg od for 10 weeks • ?Reduce dose in renal impairment – A reduction of the dose from 20 mg once daily to 15 mg once daily should be considered if the patient's assessed risk for bleeding outweighs the risk for recurrent DVT and PE • Consider duration of treatment – Reassess after 3 months • Idiopathic/ recurrent events – Long term rivaroxaban? LMWH for patients with VTE and cancer Lee A et al, NEJM 2003 LMWH for treatment VTE • Dalteparin 200iu/kg once daily – In pregnancy currently use bd dosing. • SPC caps dose at 18,000iu od if >83kg- good evidence that need to increase dose in those with high body weight- STH guideline • After 30 days reduce dose (as per BNF/ STH guideline) • Patients with cancer- watch weight and renal function- may need to adjust dose Summary: Anticoagulation for VTE • Patient choice • Rivaroxaban after a first VTE event (unless contraindicated), if unstable, side effects or contraindications to warfarin • LMWH if cancer, pregnant, interacting drugs with rivaroxaban – Once chemotherapy finished, consider switching from LMWH to alternative anticoagulant • Warfarin if renal failure, if recurrent events Mechanical Heart Valves • Do not use NOACs • Study of dabigatran terminated as excess bleeding and thrombosis in dabigatran groups compared with warfarin. • No other studies underway at present Converting from Warfarin to Rivaroxaban • Discuss with patient • Check that patient is not on interacting drugs • Baseline bloods- FBC, renal and liver biochemistry, INR • Make switch • Review in a couple of weeks for side-effects and to reinforce compliance • FBC and biochemistry every 3-12 months Converting from Warfarin to Rivaroxaban Stop VKA VKA VTE treatment INR < 2.5 Monitor INR Commence Xarelto at appropriate dose SPAF INR < 3.0 VTE INR ≤ 2.5 Xarelto SmPC SmPC for rivaroxaban L.GB.12.2012.1370 Date of preparation Jan 2013 28 Converting from Rivaroxaban to Warfarin Standard VKA dose Xarelto (dose according to indication) INR adapted VKA dose Xarelto can be stopped once INR > 2.0 INR testing before Xarelto administration Days Once Rivaroxaban is discontinued, INR testing may be done reliably at least 24hrs after the last dose Xarelto SmPC Preoperative management of Rivaroxaban anticoagulation Major surgery Minor surgery Postoperative Management of Rivaroxaban Anticoagulation Major surgery Surgery D +1 D+2 Prophylactic dalteparin once daily starting 6 – 8 hrs post op D+3 D+4 D+5 D+6 Stop dalteparin the day before restarting therapeutic rivaroxaban (at day 3 – 5, at earliest on day 3, depending on bleeding tendency). Check U&E/LFT. Do not restart therapeutic anticoagulation with epidural in situ. Minor surgery Start Rivaroxaban 24 hours after the procedure. Patients admitted on prophylactic doses of rivaroxaban (10 mg OD) may be re-started 6 8 hours post op. Patient pathway for VTE Diagnosed in A&E or as an inpatient Seen by thrombosis nurse specialist for assessment MDT Malignancy suspected 2 weeks O.P.A with consultant Unprovoked DVT 4 to 6 weeks O.P.A with consultant Provoked DVT Nurse-led pathway Conclusion • Warfarin and NOACs both efficacious in prevention stroke in AF and treatment VTE • Most important issue is patients who require anticoagulation have access to anticoagulant treatment