* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
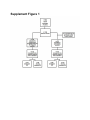
Download Is a Diabetes-Linked Amino Acid Signature associated with Beta
Basal metabolic rate wikipedia , lookup
Personalized medicine wikipedia , lookup
Genetic code wikipedia , lookup
Biosynthesis wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Metabolic network modelling wikipedia , lookup
Metabolomics wikipedia , lookup
Pharmacometabolomics wikipedia , lookup
DOI: 10.1161/CIRCGENETICS.113.000421 Is a Diabetes-Linked Amino Acid Signature associated with Beta BlockerInduced Impaired Fasting Glucose? Running title: Cooper-DeHoff et al.; Amino acids and ȕEORFNHU-induced IFG Rhonda M. Cooper-DeHoff, PharmD, MS1,2,3,4; Wei Hou, PhD6; Liming Weng, PhD1,2,3; Rebecca A. Baillie, PhD7; Amber L. Beitelshees, PharmD, MPH8; Yan Gong, PhD1,2,3; Mohamed H.A. Shahin, MS1,2,3; Stephen T. Turner, MD9; Arlene Chapman, MD10; John G. Downloaded from http://circgenetics.ahajournals.org/ by guest on May 6, 2017 Gums, PharmD1,3,5; Stephen H. Boyle, PhD11; Hongjie Zhu, PhD11; William R. Wikoff,, PhD12; Phar PharmD, armD ar mD,, Ph mD PhD D8; Ri Rim Rima ma ma Eric Boerwinkle, PhD13; Oliver Fiehn, PhD12; Reginald F. Frye, Ph 1,2,3,4* , ,4* Kaddurah-Daouk, PhD11*; Julie A. Johnson, Pharm m D1,2,3 1 Department mentt of Pharmacotherapy me Pharma m co oth t erap apyy and ap an nd Translational Trran Tran anslat atio at ionaal Re io R Research, seaarch, 2Ce Center Cent nter ffor nt o Pharmacogenomics, or Pharm rm rmacog m og gen enom o ics, om s,, 3Co College Coll llege of ll 4 5 Division D vision of Cardiovascular Card dio iov ovascul ullar Medicine, Med e icin in ne, De Department D eparrtm men nt ooff Community Comm Co munitty Health Heal a th h and ndd Family Fam mil ily y Medicine, Med Me dicine, College Pharmacy, Div Department D eparttmentt of of Preventive P even Pr ntive Medicine, Mediicin i e, College Col ollege ol ge of Medicine, M dii Me n University ne, Uni n versity of F loridda,, Gain i esville, F in L; 6De of Medicine, Florida, Gainesville, FL; 7 Rosa Ros sa & C Co. o. L LLC, L ,S LC San an Ca Carlos, arl rlos os,, CA os CA;; 8Depa Department De epa parttme ment n ooff Me Medicine, edicin ne, Sc School c of Stony Brook University, Stony Unniv iver ersi er s ty y, St S on ny Brook, Brook, k NY; NY Y; Ro Division ion o of Neph Nephrology p ro ph r logy and Hypertension, Hyp y ertension, Department Departmee of Medicine, Universi University Maryland, Baltimore, ity of Mary ryla ry l nd, Ba alt ltim imore, MD; 9Divisi Renal R nall Di na Divi Division, v siion vi on, Sc Sch School hool of Medicine Emory Internal Medicine, Clinic College Medicine, Rochester MN, d dicine, Mayo Clini nicc Co ni oll lleg egee of eg of M edic ed iccin ine, e, R oche oc oche hest ster err M N 100Re N, 11 Department tmentt off Ps P Psychiatry ychi hiiatry t aand nd dB Behavioral eh havio i ral Sc S Sciences, iences, D Duke uke U University niversity i Medical C Center, University, Atlanta, GA; Depart 13 ty y ooff Cali Ca California ali lifo fo orn r iaa D Davis avis G av avis Genome en enom nome ome Cent C Ce Center; ent nter er;; Un er Univ University iver iv ersi er sity ooff Ca sity Cali California lifo li forn fo rnia rn iiaa D Davis, avis av iss, Da D Davis, vis, CA; 13 Human Durham, NC;; 12University cs and cs and institute ins nsti titu tute te ooff Mo Mole Molecular lecu cula larr Me Medi Medicine, dici di cine ne,, University Univ Un iver ersi sity ity ooff Te Texa Texas xass He Heal Health alth th S Science cien ci ence ce C Center, ente en ter, r, H Houston, oust ou ston on,, TX Genetics *contributed equally as co-senior authors Correspondence: Rhonda M. Cooper-DeHoff, PharmD, MS Department of Pharmacotherapy and Translational Research & Center for Pharmacogenomics University of Florida Gainesville, FL 32610 Tel: 352-273-6184 Fax: 352-273-6121 E-mail: [email protected] Journal Subject Codes: [192] Glucose intolerance, [193] Clinical studies 1 DOI: 10.1161/CIRCGENETICS.113.000421 Abstract: Background - The five amino acid (AA) signature including isoleucine (Ile), leucine (Leu), valine (Val), tyrosine (Tyr), and phenylalanine (Phe) has been associated with incident diabetes and insulin resistance. We investigated whether this same AA signature, single nucleotide polymorphisms (SNPs) in genes in their catabolic pathway, were associated with development of impaired fasting glucose (IFG) after atenolol treatment. Methods and Results - Among 234 European American participants enrolled in the Pharmacogenomic Evaluation of Antihypertensive Responses (PEAR) study and treated with Downloaded from http://circgenetics.ahajournals.org/ by guest on May 6, 2017 atenolol for 9 weeks, we prospectively followed a nested cohort that had both metabolomics profiling and genotype data available, for the development of IFG. Wee as assessed association asse sess se ssed ss ed th thee as asso ocia between baseline circulating levels of Ile, Leu, Val, Tyr and Phe, as well BCAT1 ell as SN SNPs Ps iin n BC BCAT A and PAH with development levels d ve deve velo l pm lo pmen ent off IFG. All baseline AA le en eve vels l were stronglyy aassociated ssociated with IFG development. increment AAss was with nt. Each nt Each increm em men nt in n sstandard tand ta ndar nd ardd de ar ddeviation viiat atio on off the he five fiv ivee AA A was associated wa asssoci as ciiat ated ed dw itth thee following odds Ile 2.29 oddds ratio ratio and and n 95% 5% confidence confi fide fi dencce interval de in ntervvall fo for or IFG FG G bas based ased oon n fu fully ullly ad adjusted dju j stted m model: odeel:: Il (1.31-4.01),, Leu 1.80 (1.10 (1.10-2.96), Vall 1.77 Tyr 0-2 - .9 .96) 6)), Va V 777 ((1.07-2.92), 1..077-2.992)), Ty yr 2. 22.13 133 (1.20-3.78) (1.20 20-3 20 -3.778)) and Phe 2.04 (1.16-3 3.59). The compo ccomposite p site p value vallue was 2x 22x10 100-55. Those T ose wi Th with ith h PA PAH H ((rs2245360) rs22 2245 22 453660) AA AA genotype g noty ge ype p had the t highest incidence idence ide denc de ncee of IFG IFG (p (p fo forr tr tren trend=0.0003). end= d=0. d= 0 0003 0. 00003 0 )). Conclusions - Our data provide important insight into the metabolic and genetic mechanisms underlying atenolol associated adverse metabolic effects. Clinical Trial Registration - clinicaltrials.gov; Unique Identifier: NCT00246519 Key words: amino acids, impaired glucose tolerance, pharmacogenetics, metabolomics, betablocker 2 DOI: 10.1161/CIRCGENETICS.113.000421 Hypertension and being overweight or obese have previously been identified as risk factors for diabetes and frequently coexist.1, 2 Impaired fasting glucose (IFG), impaired glucose tolerance (IGT) and insulin resistance, important early components of cardiometabolic dysfunction, are also prevalent in those with hypertension 3 and significantly increase the risk for diabetes.4, 5 Eblockers, although very effective blood pressure (BP) lowering agents, are associated with adverse metabolic effects, including hyperglycemia, IFG, and diabetes, all of which are associated with adverse cardiovascular consequences long-term.6-8 While the mechanistic Downloaded from http://circgenetics.ahajournals.org/ by guest on May 6, 2017 underpinnings of Eblocker associated adverse metabolic effects are incompletely nco c mp pletely ly y under understood, erst er stooo we st bolic eeffects ffec ects ts iss pr pres e en in have previously shown that risk for Eblocker associated adverse metabolic present those with and andd w without ithout u abdominal ut abd bdominal obesity.6 bd Identification ntification nt tiffication of risk rissk factors facctorrs for diabetes fa diab betess has has been beenn a focus f cuss for decades, fo deecaades, with with tthe he ul uultimate ultim ltim goal of developing elopi strateg eloping strat strategies gie iess to del delay ellay ay or pr pprevent event onse onset s t of diabetes diaabe b tes in tho those hoos at highest risk. B hose Based on data from m oobservational bser bs erva er vati va tion ti onal on al and and rrandomized ando an domi do mize mi zedd cl ze clin clinical inic in ical ic al ttrials, rial ri alss, cl al clin clinical inic in ical ic al ch char characteristics arac ar acte ac teri te rist ri stic st icss kn ic know known ownn to ow increase risk for drug induced diabetes include age, ethnicity, race, body mass index, hypertension, stroke, among many others.9 Metabolomics, a rapidly growing field that enables mapping of global biochemical changes associated with disease or treatment,10 has been used successfully to map pathways implicated in mechanisms of variation of response to drugs.11 Recently, metabolomics data from an observational study of the Framingham Offspring cohort identified a small cluster of essential amino acids (AAs), including baseline levels of three branched-chain amino acids (BCAA), isoleucine (IIe), leucine (Leu) and valine (Val), and two aromatic amino acids (AAA), phenylalanine (Phe) and tyrosine (Tyr), as metabolomic risk factors associated with a significant and independent increased risk for diabetes.12 This same 5 AA cluster has also been identified as 3 DOI: 10.1161/CIRCGENETICS.113.000421 a predictor of insulin resistance in young adults.13 While the clinical characteristics that predict risk for drug induced diabetes are similar to those that predict diabetes of other etiologies,14, 15 there are currently no data available to assess whether the same profile of BCAA and AAA that predicted diabetes and insulin resistance risk might also predict risk for drug induced alterations in glucose status. Likewise, while branched chain amino-acid transaminase 1, BCAT1 and phenylalanine hydroxylase, PAH, genes that catabolize BCAA and AA respectively, have been previously associated with diabetes16 and Downloaded from http://circgenetics.ahajournals.org/ by guest on May 6, 2017 insulin resistance,17 there are no data regarding the associations of single gle nucleotide polymorphisms (SNPs) in these genes with drug induced metabolic changes. hanges. Therefore, Thereffore, in a Th nested cohort ort from from the thee Pharmacogenomic Pha harmacogenomic Evaluation ha Evaluatio io on of Antihypertens Antihypertensive nsiv ns i e Responses (PEA (PEAR) study, we used used us d a targeted d ““pharmacometabolomics phaarmaccomet etaabolom miccs informed infform rmed ed d pharmacogenomics” phaarmaccogen enomic en icss” approach ic approoachh 18 to whet ethe et herr bbaseline he aseeli line ne llevels evel ev e s of the th previously prev evio ev iouusly io ly identified ly id den enti tif ti ifi fied fied d BCAA BC CAA A and an AAA AAA and and SNP SN NPs in investigate whether SNPs genes in their e catabolic pathways, eir pathhway ys,, increase increase the th he odds oddds off development d velo de l pm p ent off IFG, IFG IF G, a phenotype p enotyp ph ype yp associated with iith thh significant ignifi ifi nt increased i ed d risk iskk off ddiabetes iabbete and d cardiovascular cardio rdi dio asc llar di disease ffollo following oll ll iing ng sshort term exposure to atenolol.4, 19 Methods PEAR is a prospective, randomized, parallel group, titration study undertaken to evaluate the pharmacogenomic determinants of the antihypertensive and adverse metabolic responses to atenolol and hydrochlorothiazide in hypertensive participants without a history of heart disease or diabetes. Details regarding study design and enrollment criteria have been previously published.20 The study is registered at clinicaltrials.gov, NCT00246519, http://clinicaltrials.gov/ct2/show/NCT00246519. 4 DOI: 10.1161/CIRCGENETICS.113.000421 Study Population PEAR participants were enrolled from primary care clinics at the University of Florida (Gainesville, FL), Emory University (Atlanta, GA), and the Mayo Clinic (Rochester, MN). Because we are seeking to extend findings generated in a primarily White population, this analysis includes a subset of European American men and women with mild-to-moderate essential hypertension, between the ages of 17-65, selected for metabolomics profiling and with available genotype information within the atenolol arm of PEAR (Supplement Figure 1). For Downloaded from http://circgenetics.ahajournals.org/ by guest on May 6, 2017 metabolomics profiling, patients were divided into quartiles based on the DBP response to atenolol monotherapy. Equal numbers of patients were selected randomly mly within wit ithi it hin each hi each h response res espo quartile using PROC SURVEYSELECT random n tthe ng h P he RO ROC O SU S RVEYSELECT procedure procedu durre (simple rando du om sampling without replacement). n ). P nt) Prior rior to bas baseline seline measurements, meas a urem as men nts, tthose hose rreceiving eceeiv ivin ingg tr in treatment reaatmen en nt for foor hypertension hyypertension p rtten pe nsion on aat enrollment hadd aall lll antihypertensive antih hyp yper erte t ns te nsiv i e drugs druugs discontinued dr diisc scon onti on tinu ti nued d for forr a m median edia ed ian ia an 277 ((interquartile inte terq te rquart rq rttil ilee range rang ra nge [IQR] 19-GD\V3DUWLFLSDQWVZLWK,)*JOXFRVHPJGODWEDVHOLQHZHUHH[FOXGHGIURP GD\V3DUWLFLSDQWVZLWK,)*JOXFRVHPJGODWEDVHOLQHZHUHH[FOXGHG GD\V \ 3DUWLFLS LSDQWVZLLWK K ,) ,)* * JJOX O FRVH PJ JGO GOO DW EDVHOOLQHZHUHH[FOXGHG this analysis. s PEAR Protocol Written informed consent was obtained voluntarily from all participants, and institutional review board approval was obtained from all study sites, which included University of Florida, Mayo Clinic and Emory University. Atenolol was initiated at 50mg, and titrated to 100mg, based on BP > 120/70 mmHg and tolerability. After at least 6 weeks on the final dose, response was determined, including BP measurements and laboratory-based assessments, and results described in this analysis come from this response assessment time point. PEAR is registered at ClinicalTrials.gov, #NCT00246519. 5 DOI: 10.1161/CIRCGENETICS.113.000421 Laboratory Measurements At baseline and after completion of atenolol monotherapy, fasting blood samples were collected for glucose and insulin. Insulin sensitivity status was calculated using the homeostatic model assessment – insulin resistance (HOMA-IR).21 Biochemical Assays Glucose was measured in plasma, on an Hitachi 911 Chemistry Analyzer (Roche Diagnostics) at the central laboratory at the Mayo Clinic, Rochester, MN, using spectrophotometry by an Downloaded from http://circgenetics.ahajournals.org/ by guest on May 6, 2017 automated enzymatic assay. Plasma insulin was measured using the Access ccess Ultrasensitive ve Insulin I immunoassay system (Beckman Instruments). All samples were tested d in du dduplicate, pllic i ate, t andd ddata at eans ea ns of of th tthee duplicate d plicate samples. du reported aree m means Quantificatio io on Amino Acidd Quantification mples were wer ere tran ansf sfer ferre redd from m the the PEAR PEA EAR AR laboratory labo labo b ra rato tory to ry to to the th he metabolomics mettabo me tab lo lomi mics core mi cor oree labor l bor la bo Plasma samples transferred laboratory, o California,, Davis, D viis,, where Da whhere samples sampl ples l were extracted, d dderivatized d, erivatiized d and analy yzed ass University of analyzed 22, 23 e iio sll iin n great at detail ddetail. etail il 22 Briefly, Briefl B riiefl fl mass spectrometry spectrometr tr et used sed daL Leco e P Pegasus Pegas s IV IV time reported previously of flight (TOF) mass spectrometer with 280qC transfer line temperature, electron ionization at 70V and an ion source temperature of 250qC. Mass spectra were acquired from m/z 85-500 at 20 spectra/sec and 1750 V detector voltage. Quantitative data were normalized to the sum intensities of all known metabolites and used for statistical investigation. Genotyping and Quality Control To explore a potential functional mechanism underlying our observation that the 5 AA signature was associated with development of IFG following exposure to atenolol, we sought to investigate genes in common catabolic pathways. Because we had a limited population (n=184) with available genetic data and that did not have IFG at baseline, we used a candidate gene approach 6 DOI: 10.1161/CIRCGENETICS.113.000421 for this analysis. For the BCAAs, we chose to focus on BCAT1, which encodes the cytosolic form of the enzyme branched-chain amino acid transaminase. This enzyme catalyzes the reversible transamination of branched-chain alpha-keto acids to branched-chain L-amino acids essential for cell growth, and importantly, the aminotransferase step is the first step in the catabolic process for Ile, Leu and Val. For the AAs, we chose to focus on PAH, which encodes the enzyme phenylalanine hydroxylase. Phenylalanine hydroxylase catalyzes the hydroxylation of the aromatic side-chain of phenylalanine to generate tyrosine, and is the rate-limiting enzyme Downloaded from http://circgenetics.ahajournals.org/ by guest on May 6, 2017 of the metabolic pathway that degrades excess phenylalanine. Genotypes for SNPs in the BCAT1 and PAH genes were obtained Omni1M ned fr ffrom om tthe h O he mn ni1 i1M M quad GWAS Beadchip. The Omni1M a chi adc h p. p. T hee O mni1M quad is a GWAS chip chi h p that used the Infinium Infi In f nium II assay and genotypes w GenTrain2 calling algorithm. Procedures were ere called using usin ing BeadStudio in Beead dSt S udio o software sof ofttwaaree and an nd Ge GenT nTraain nT n2 call lin ng al algori rith ri hm. Pro Proced oced for quality Cont Control principal component population C trool and and pr an pri inciipa inci pall comp mp ponentt (P (PC) PC) C analysis ana nally lysi siss for for ddetermination fo ete teerm rmiina inationn off pop opul ulat lattion ion substructuree are described iin minor n ddetail etail il iinn the supplementary supp pplementaryy materi pp materials. ials. l After Aft f er QC QC procedures, p ocedures,, m pr allele frequency >0.05, disequilibrium pruning, enc ffilter ilte off >0 il 0 05 and d li linkage nkk diseq di ilibri ilib il ibrii m and ndd monogenic ic SNP SNP pr ning nii a total of 96 SNPs, 69 SNPs in BCAT1 and 27 SNPs in PAH, were analyzed in the genetic association study. Primary Outcome We examined the association between baseline plasma metabolite level of Ile, Leu, Val, Tyr and Phe, as well as SNP genotypes and development of IFG, defined as a new occurrence of fasting JOXFRVHPJGO24, following treatment with atenolol monotherapy. After a 12 hour fast, glucose was measured in all participants at baseline, prior to initiation of atenolol, and again at the end of atenolol therapy. Mean duration of treatment with atenolol was 9 weeks. Participants ZLWKDIDVWLQJJOXFRVHPJGODWEDVHOLQHZHUHH[FOXGHGIURPWKLVVWXG\3DUWLFLSDQWVZLWKD 7 DOI: 10.1161/CIRCGENETICS.113.000421 baseline fasting glucose < 100 mg/dl DQGDIDVWLQJJOXFRVHPJGODWWKHHQGRIDWHQRORO treatment were considered to have developed IFG. Participants with a baseline fasting glucose < 100 mg/dl and a fasting glucose that remained < 100 mg/dl at the end of atenolol treatment were considered to have not developed IFG. Statistical Analyses Frequencies and percentages were calculated for categorical baseline characteristics and were compared by using Chi-square tests between those who did and did not develop IFG. Means and Downloaded from http://circgenetics.ahajournals.org/ by guest on May 6, 2017 standard deviations or medians and interquartile ranges were calculated d for continuous baseline bas ae characteristics and were compared using t-tests or Wilcoxon rank sum test tests, respectively, ts, respe cti tivelly, between those did and glucose, o wh ose whoo di id an nd did not develop IFG. Change Chan Ch nge and percentt cchange hange in fasting gluc c baseline glucose, log-transformed ucose uc cos , baselinee iinsulin nsul ullin and and n baseline bas asellin ne AA AA levels lev evelss were we e log og-traansfo forrmed fo d ddue uee to no nnonon-normal distribution across case/control model t uttio tribut ion on and and compared com ompa pare red ac cross roo cas se/ e/co / ont ntro troll ggroups. rou ouups ps. We We uutilized t li ti lize zed a re rregression gresssi sion ion m od to od assess the association between baseline with the change adjusted a baseli line AA A llevel evell wi ith h th he pe ppercent rcent ch hange g iin n gl gglucose, ucose,, adj justt for age, gender, BMI, glucose r BMI B MI baseline ba li gl l cose and ndd ins iinsulin, lin lin andd HOMA HOMA IR IIR, R in i the th h entire tii cohort ohh t with iith th h metabolomics data available (n=150). Whether a participant developed IFG, as a binary outcome variable, was fitted by multiple logistic regression models. In separate analyses, each baseline plasma AA level was entered in the logistic model individually as a primary risk predictor. Because the distribution of those who did and did not develop IFG was significantly different by gender and previous research has shown that plasma amino acid levels differ significantly by gender,25 we conducted a subgroup analysis where each baseline plasma AA level as a primary risk predictor of IFG development was tested separately by gender. The values of AA level were standardized (mean=0 and standard deviation=1) so that the regression coefficients are interpreted as odds ratio per standard deviation.12 Odds ratios for AA level predicting IFG were 8 DOI: 10.1161/CIRCGENETICS.113.000421 estimated. For logistic regression analyses, we tested four separate models: 1) unadjusted, 2) adjusted for baseline age, sex and BMI, 3) model two plus fasting glucose and fasting insulin and 4) model three plus HOMA-IR, to test the effect of insulin resistance. We evaluated the composite effect of the AAs by combining the individual p values from the model that included HOMA-IR. Since the BCAA and AAA were all strongly correlated (r = 0.56-0.91, p<0.0001 for all comparisons), an extension to Fisher’s combination for correlated tests was performed, for testing significance of the composite p value.26 For amino acid analyses, P values less than 0.05 Downloaded from http://circgenetics.ahajournals.org/ by guest on May 6, 2017 were considered statistically significant. For pharmacogenomics analyses, deviations from Hardy Weinberg Equilibrium berg E quil ilib il ibrium i ((HWE) HW were assessed s us sed usin using in ng Fisher’s Fishher Fi er’s Exact Test with alpha=0.05.The alphaa=0 =0.05.The associat associations atio tio ions of the BCAT1 an and n PAH SNPs and baseline and IFG weree evaluated evalu lu uated d using ng logistic loggistticc regression, reegrresssi sion on, adjusting on addju justin sting ng for foor age,, gender, gen nder,, bbas as glucose, insulin sulin and and HOMA HOM OMA A (model ( od (m o ell 4 above), abovee), and n the nd the h first firrstt 2 PCs PCs Cs for for ancestry. anc ncestr tryy. To tr To account acco ac coun untt for f multiple comparisons, mparisons, mpa p risons,, alpha alph phha levels levels l for for the h SNP SNP associations associiatiions were set based based d on the total num number of SNPs included cll dded ed d in i the th h analysis, anal all sis is which hhich ichh was as 96: 96 0.05/96 0 05 05/9 05/96 /966 = 00.0005. 0005 0005 SNP SNP genot genotype t pe QC QC andd genetic association analyses were conducted in PLINK.27 All other analyses were performed by using SAS v9.3 (SAS institute, Cary, NC). Results Pharmacometabolomic Analyses Among the 234 participants randomized to the atenolol group in the PEAR study, metabolomic profiling was conducted on 150 (Supplement Figure 1). Of those, 122 did not have IFG at baseline and are the focus of the metabolomics analysis. A total of 24 participants developed IFG during an average 9 weeks of treatment with atenolol. Baseline characteristics of the participants included in the metabolomic analysis are summarized in Table 1 and do not differ from the entire 9 DOI: 10.1161/CIRCGENETICS.113.000421 atenolol cohort (data not shown). While those who developed IFG were more likely to be men, the cohort was similar in age, body mass index and systolic and diastolic BP, in those who did and did not develop IFG. Baseline fasting glucose was higher (Table 1), and change in glucose following treatment with atenolol was significantly greater among those who developed IFG than those who did not (mean 13.5 mg/dl vs. 2.1 mg/dl, p<0.0001) (Supplement Figure 2A). Median baseline plasma levels of Ile, Leu, Val and Phe were significantly higher in those who developed IFG than those who did not, while there was no difference in baseline levels of Tyr (Supplement Downloaded from http://circgenetics.ahajournals.org/ by guest on May 6, 2017 Figure 2B). Baseline levels of all five AAs were significantly associated with ith percentt change ch hange gee iin n glucose, even en after afte af terr ad te adjustment dju justtment for age, sex, BMI, baseline baseeline glucose and ba ndd insulin and HOMA-IR. HOMAThe beta coefficients baseline AAs shown oefffic oe i ients of the he bas asselin in ne fivee A As aare ree sho hownn iinn Table Tablle 2. Ta Association with n off amino ami min no acids aciids wit ith it h impaired impa paair ired fasting fas asti ting ti n glucose glucose see Associationn between baseline odds baseli liine AA AA level levell (per ( er SD (p S increment)) andd od dds fo fforr de ddevelopment velopm p ent of IFG IF F for models 1-4 in Results Model i the th h ooverall erall all ll cohort hort iiss summarized s mmari rii edd in in Supplement S pplement pll t Table Tabl bl 11. R Res llts ts from f Mode M odde 4 showed the following odds ratio and 95% confidence interval for IFG: Ile 2.29 (1.31-4.01), Leu 1.80 (1.10-2.96), Val 1.77 (1.07-2.92), Tyr 2.13 (1.20-3.78) and Phe 2.04 (1.16-3.59). (Figure 1). When the p values from the individual AAs were combined, the composite p value was 2x10-5 for Model 4, indicating a very strong association between this five AA metabolite profile and odds for IFG following atenolol treatment. In subgroup analysis of odds for IFG according to gender, with the covariates from Model 4, in women (n=6 developed IFG and n=62 did not develop IFG), the ORs ranged from 1.18-3.67, however none of the associations reach statistical significance likely due to the small number of women that developed IFG. In men (n=18 developed IFG and n=36 did not develop IFG), the ORs ranged from 1.75-2.47, and baseline 10 DOI: 10.1161/CIRCGENETICS.113.000421 levels of Ile, Tyr and Phe were associated with significantly increased adjusted odds for developing IFG. Data summarizing adjusted odds for IFG according to gender are presented in Supplement Table 2. Pharmacogenetic Analysis Patients with the PAH rs2245360 AA genotype had the highest incidence of IFG (41.7%), compared with patients with AG genotype (15.2%) and GG genotype (7.4%), p for trend=0.0003, which achieved statistical significance (Figure 2). In analyses of odds per allele Downloaded from http://circgenetics.ahajournals.org/ by guest on May 6, 2017 for developing IFG after atenolol monotherapy, PAH rs2245360 had adjusted ORs of 3.51 3.51, 51, 95% 51 confidence interval 1.62-7.63, p = 0.0015. None of the BCAT1 SNPs tested estedd achieved achi hieved hi d Bonferroni Bon onffe corrected statistical significance t issti tatis t ca call si sign g iffic icance for development of IIFG. F . FG Conclusions n ns We have shown, first h hown, for the fi irs rstt time, time ti m , that me t at baseline th b se ba seli line li ne plasma plaasm smaa levels leveels l of of Ile, Ile, e Leu, Leu e , Va Val, l Tyr and Phe, Phee as well as a gene catabolic strongly with developing ene ne iin n a ca cata tabo boli licc pathway, li p th path pa t wa wayyy,, are are stro st trong ngly gly y aassociated ssoc ss ocia iate tedd wi ith increased incr in crea ease sedd od odds dds ds of of de deve develo velo lo IFG in hypertensive participants, treated relatively short-term (9 weeks) with atenolol, a commonly prescribed ȕ blocker. Importantly, IFG is an independent predictor of diabetes,4 and adding measures of anthropometrics and/or insulin resistance status into prediction models is often more informative for assessing diabetes risk.28 Here, we show that baseline levels of 4 of the 5 AAs investigated are significantly different in those who do and do not develop IFG, and the AAs can further inform a prediction model for drug induced IFG, an early metabolic risk phenotype in the diabetes continuum. Our data extend recent findings from the Framingham Offspring study,12 an observational study primarily in Whites, that showed a strong association between these AAs and incident diabetes. Overweight, obesity, glucose intolerance and insulin resistance have been closely linked 11 DOI: 10.1161/CIRCGENETICS.113.000421 through many important biochemical and regulatory pathways.29 Knowledge that higher blood concentrations of Ile, Leu, Val, Tyr and Phe are elevated in people with obesity, insulin resistance or diabetes is not new.30 The continued rise in prevalence of risk conditions for diabetes in the last 2 decades has increased the need to better understand all relevant underlying pathways. Recent developments in metabolite profiling or metabolomics, have provided insight into additional biochemical pathways that play an important role in glucose and insulin regulation.31 In a study comparing lean and obese individuals, BCAA and AAA were recently Downloaded from http://circgenetics.ahajournals.org/ by guest on May 6, 2017 recognized as a metabolite cluster strongly associated with insulin sensitivity. sitivity. y 32 Additiona Additionally, nall na BCAA have been associated with coronary artery disease 33 and diabetes. tes.122 W Wee hhave ave furt further rthhe rt he B AA A as a sig gni n ficant predictor for ateno nolo no lol-induced IFG,, iin n a population of middlemii confirmed BCAA significant atenolol-induced aged, otherwise wisse healthy, hy hype hypertensive erttensi siive individuals, ind n iv nd vid duaalss, pr providing rovid din ingg aadditional dddittional al evi evidence viidencce fo for or its uutility till as nt riskk biomarker. bioomar bi arke k r. ke r In In an a analysis ana naaly lysis st stra rati ra t fi ti fied ed d by by gender, geend nder er, Hu er uff f ma mann, eett al l, sh how owed ed d tthat tha h a ha an important stratified Huffman, al, showed cluster of large arge g neutral AA’s AA’ A’s (Leu/Ile A’ ( eu/I (L / le /I l rati ratio, io, V Val, al, l, P Phe, he, T Try, ry, pproline roliine andd hhistidine) istiidi dine)) alongg with uric acid was a significant ignifi ifi nt independent indd dent predictor dict off insulin di iins n li lin sensitivity sensiti itii iitt in in both bothh men andd women. omenn 225 In our subgroup analysis evaluating the 5 AA signature stratified by gender, we confirmed an association with increased odds for IFG in men, and saw similar, though non-significant trends in women. Our inability to confirm an association in women is likely due to the small number of women who developed IFG in our study, and warrants further investigation in a larger cohort of women treated with atenolol. Importantly, evidence also exists suggesting that these AAs may play a causal role in metabolic dysregulation. A study evaluating the effect of a high fat diet with or without BCAA supplementation in rats demonstrated that the high fat plus BCAA diet resulted in a higher rate of insulin resistance than the high fat diet alone.32 Similarly, a small study of healthy men who were 12 DOI: 10.1161/CIRCGENETICS.113.000421 infused a solution containing 18 AAs showed that AAs impair both insulin-mediated suppression of glucose production and insulin-stimulated glucose disposal in skeletal muscle.34 Together, these data suggest that these metabolites contribute to the development of metabolic dysregulation and are not just innocent bystanders in the metabolic disease continuum.35 Our data from a relatively overweight/obese hypertensive population suggest that treatment with atenolol may be another environmental exposure that has interactions between BCAA, AAA and glucose regulation, as we observed significantly increased odds for IFG, an important early Downloaded from http://circgenetics.ahajournals.org/ by guest on May 6, 2017 predictor for diabetes, associated with all amino acids tested. PAH, on chromosome 12 (12q22-q24.2) has been associated with alt altered lttered d met metabolic tab bol oliic ic h African he Afr f iccan American Ame meri r can cohort from the Insulin Insu suli su lin Resistance At the h rosclerosis Family Study, status. In the Atherosclerosis age between the ag he 12q22-24.2 12q 2qq22 22-224.2 region regiion (PAH) re ((PA PA AH) andd ac acut u e in ut iinsulin nsuliin response reespponsse se too glucose gluccose was strong linkage acute Linkage kage ka age ttoo fasting f st fa stin tin i g insulin in in has insuli ha previously prrev evio ious io usly ly been ly bee eenn reported repo re portted po d nnear earr 12 ea 12q23 2q23 q23 in tthe q2 he A he Amish mis mi is as observed.17 Linka well.36 Our observation of a significant siign g ifficant difference diff di fference iin ff n de ddevelopment velopm p ent off IFG FG accordingg to PAH H rs2245360 genotype genott pe ssuggests ggests t thi this his gene ma may play pla pll an iimportant m rt t ffunctional nctional tii all role oll iinn metabolic etab boli lic risk. ȕ-adrenergic blockers have long been recommended as first-line therapy for the treatment of hypertension, especially in patients with coronary artery disease.37 HoZHYHUȕ-blockers have been implicated in altering glucose homeostasis, primarily through inhibition of pancreatic insulin secretion and promoting insulin resistance. ȕ-receptor selectivity appears to play a role in the degree of downstream metabolic effects, which include not only glucose increases but also weight gain and dyslipidemia. Nonselective and higher-dose selective agents result in the largest adverse metabolic changes.38 1HZHUȕ-blockers including nebivolol and carvedilol appear to minimally affect glucose homeostasis and improve insulin sensitivity.39, 40 While the mechanisms XQGHUO\LQJWKLVGLIIHUHQWLDOHIIHFWRQJOXFRVHUHPDLQXQFOHDUWKH\OLNHO\H[WHQGEH\RQGȕ 13 DOI: 10.1161/CIRCGENETICS.113.000421 blockade. $WHQROROWKHȕEORFNHUXVHGWRWUHDWparticipants in PEAR is cardioselective, and the 100mg dose used is the usual dose used to treat hypertension. We observe that AAs and PAH, a gene previously associated with insulin sensitivity, are also associated with atenolol-associated IFG. These data suggest the underlying mechanisms of drug-induced and primary dysmetabolism may be the same. Drugs may be an environmental trigger in patients with metabolic risk factors. There are a few limitations of our study that are worthy of mention. First, while we targeted our analysis on a metabolite cluster previously identified for risk of diabetes in a Downloaded from http://circgenetics.ahajournals.org/ by guest on May 6, 2017 population of Whites, we conducted our analyses only in a cohort of European American Americans. an ns. Because our cohorts is relatively small, particularly for analyses by gender, results should nder, th these result ts sh be replicated WUHDWHGZLWKȕEORFNHUVand e in oother ed ther th er ppopulations oppul u ations with hypertension, hypertension onn, untreated untreated and WU UHD HDWHGZLWKȕEORFNHUV other drugs tthat metabolic risks, while did haat possess adverse adverrsee m e abol et o ic ri ol iskss, forr cconfirmation. onf nfir nf irma ir m tion ma on. Second, Seco ondd, whi hii e we hile we di id observe obb a significant with atenolol, cannot n association nt asso sooci ciat a ionn with at wiith t IFG IFG FG in our ou u cohort coho h rt treated ho treat atted dw ith it th at tenol ollol ol, l we can anno an not exclude no excl clud ludde th tthat h other metabolites findings European b bolites are also pplaying layiingg a role. rolle. Third, Thi h rdd, our fi nddings g were iidentified d ntif de ifie if i d among g Europe pe American hhypertensive individuals, only generalizable pertensi rt sii e iindi ndi di id als ls treated tr tedd with iith th h atenolol te loll andd as ssuch ch h are onl l generali ali li abl able ble among similar patient populations. +RZHYHUXVHRIȕEORFNHUVLQJHQHUal, and atenolol specifically is highly prevalent. In 2010, more than 36 million prescriptions were filled for atenolol or atenolol combinations in the US.41 Lastly, further investigation is warranted to 1) determine whether this 5 amino acid signature is causal in the development of IFG or is simply a marker of insulin resistance and impaired beta cell function and 2) confirm our pharmacogenetic association and to extend this finding to other antihypertensive agents associated with hyperglycemia, including thiazide diuretics and other race and ethnic groups at high risk for metabolic dysfunction. In conclusion, we have extended the previous findings associating BCAA and AAAs 14 DOI: 10.1161/CIRCGENETICS.113.000421 with incident diabetes, to atenolol-induced IFG development. Our findings are important as they suggest novel biomarkers for the identification of those individuals at risk of developing antihypertensive treatment-induced diabetes. They may also provide insights to help better understand the mechanisms of E-blocker-induced dysglycemia. While pharmacogenomics has shown to be informative with regard to drug:gene interactions for ȕEORFNHU%3UHVSRQVH42 this study represents one of the first to employ a targeted pharmacometabolomic investigation combined with a pharmacogenomics investigation informed by the pharmacometabolomic Downloaded from http://circgenetics.ahajournals.org/ by guest on May 6, 2017 findings for the antihypertensive drug-induced dysglycemia phenotype. e. With the prevalence p evalen pr enc of en overweight, obesity, insulin resistance, hypertension and diabetes on the he ri rise ise not onl only nlyy in n tthe he US, but worldwide, wide, e, understanding und nder erst stan st nding as many aspects of the th he mechanisticc unde underpinnings deerpinnings of drug in induced adverse metabolic taaboolic effectss aass ppossible ossibl b e is iimportant. bl mp portaantt. Pharmacometabolomics Phaarmac acom ac o ettabolom om om miccs is a new, new w, bu but ut rap rapidly p e that eld th hatt has has promi mise iinn defining mi defi finiing pat thways implicated impllicatted d in mechanisms mech chanisms off variation ch variiatiion of growing field promise pathways response to th ther therapies erap er apie ap iess an andd co comp compliments mpli mp lime ment me ntss information nt info in form rmat rm atio at ionn gained gain ga ined ed from fro rom m a ph phar pharmacogenomics arma ar maco ma coge co geno ge nomi no mics cs appr aapproach. pprr pp h may bbe off immense i l as the h focus f f drug d h d a Together, they value for therapy moves towards personalized approach. Acknowledgments: This study was presented at the American Heart Association 2012 Scientific Sessions meeting, Los Angeles, California, in abstract form. Funding Sources: This work was funded by the Pharmacometabolomics Research Network (RC2 GM092729) and the NIH Pharmacogenomics Research Network (U01 GM074492). Additional funding includes: K23 HL086558 (RMC-D), K23 HL091120 (ALB), and grants from the NIH National Center for Research Resources to the University of Florida (UL1 TR000064), Emory University (UL1 TR000454), and Mayo Clinic (UL1 TR000135). Conflict of Interest Disclosures: RK-D holds patents in the metabolomics field. All others have none. 15 DOI: 10.1161/CIRCGENETICS.113.000421 References: 1. Roger VL, Go AS, Lloyd-Jones DM, Benjamin EJ, Berry JD, Borden WB, et al. Subcommittee AHASCaSS. Heart disease and stroke statistics--2012 update: A report from the american heart association. Circulation. 2012;125:e2-e220. 2. Bombelli M, Facchetti R, Sega R, Carugo S, Fodri D, Brambilla G, et al. Impact of body mass index and waist circumference on the long-term risk of diabetes mellitus, hypertension, and cardiac organ damage. Hypertension. 2011;58:1029-1035. 3. Hayden MR, Sowers JR. Treating hypertension while protecting the vulnerable islet in the cardiometabolic syndrome. J Am Soc Hypertens. 2008;2:239-266. Downloaded from http://circgenetics.ahajournals.org/ by guest on May 6, 2017 4. Nichols GA, Hillier TA, Brown JB. Normal fasting plasma glucose and risk of type 2 diabetes diagnosis. Am J Med. 2008;121:519-524. 5. Tabák AG, Jokela M, Akbaraly TN, Brunner EJ, Kivimäki M, Wittee DR. DR R. Trajectories Traject j toriess ooff glycaemia, insulin sensitivity, and insulin secretion before diagnosis off typee 2 diabetes: dia iabe bete be tes: te s: An An analysis from 2009;373:2215-2221. o the om the whitehall w itteh wh e al alll ii study. Lancet. 2009;3 373 73:2215-2221. 6. Cooper-DeHoff Gums Impact DeH Hoff RM, We Wen S,, Beitelshees Bei eittelshe ei h ess AL, he AL,, Zineh Zin neh h I, Gu Gum ms JG, JG, Turner Turrneer ST, ett aal. l.. Imp mpacc of abdominal obesity adverse antihypertensive obbessity on incidence inc n id dencee off ad advers rse m metabolic etaabolic ef effects ffe fectss aassociated ssocciated ed dw with itth an antihypertensi nti tihyypertten nsi medications. Hypertension. 2010;55:61-68. s Hy s. H peert r ensiion. 20 2010 1 ;55: 10 5:61 61-68. 61 1 7. Messerli FH,, Bangalore diuretics Banggalore S, S JJulius uliu i s S. S. Risk/benefit Risk/ k be b nefi fiit assessment off beta-blockers beta-bblock kers and diureti precludes their Circulation. 2008;117:2706-2715; heir use fo forr firs ffirst-line fi irs rstt-li tline li ne therapy the hera rapy iin rapy n hypertension. hype hy hype pert rten rt ensi en s on si on. Ci Circ rcul rcul ulat la io ion on. 20 2008 088;1 08;1 ;117 177:2 :270 7 6-27155 discussion 2715. 2715 2 7155 71 8. Elliott WJ, Meyer PM. Incident diabetes in clinical trials of antihypertensive drugs: A network meta-analysis. Lancet. 2007;369:201-207. 9. Bakris G, Stockert J, Molitch M, Zhou Q, Champion A, Bacher P, et al. Risk factor assessment for new onset diabetes: Literature review. Diabetes Obes Metab. 2009;11:177-187. 10. Kaddurah-Daouk R, Kristal BS, Weinshilboum RM. Metabolomics: A global biochemical approach to drug response and disease. Annu Rev Pharmacol Toxicol. 2008;48:653-683. 11. Trupp M, Zhu H, Wikoff WR, Baillie RA, Zeng ZB, Karp PD, et al. Metabolomics reveals amino acids contribute to variation in response to simvastatin treatment. PLoS One. 2012;7:e38386. 12. Wang TJ, Larson MG, Vasan RS, Cheng S, Rhee EP, McCabe E, et al. Metabolite profiles and the risk of developing diabetes. Nat Med. 2011;17:448-453. 13. Würtz P, Soininen P, Kangas AJ, Rönnemaa T, Lehtimäki T, Kähönen M, et al. Branchedchain and aromatic amino acids are predictors of insulin resistance in young adults. Diabetes 16 DOI: 10.1161/CIRCGENETICS.113.000421 Care. 2013;36:648-655. 14. Cooper-Dehoff R, Cohen JD, Bakris GL, Messerli FH, Erdine S, Hewkin AC, et al. Predictors of development of diabetes mellitus in patients with coronary artery disease taking antihypertensive medications (findings from the International Verapamil sr-Trandolapril Study [INVEST]). Am J Cardiol. 2006;98:890-894. 15. Lindholm LH, Ibsen H, Borch-Johnsen K, Olsen MH, Wachtell K, Dahlöf B, et al. Risk of new-onset diabetes in the losartan intervention for endpoint reduction in hypertension study. J Hypertens. 2002;20:1879-1886. Downloaded from http://circgenetics.ahajournals.org/ by guest on May 6, 2017 16. Rampersaud E, Damcott CM, Fu M, Shen H, McArdle P, Shi X, et al. Identification of novel candidate genes for type 2 diabetes from a genome-wide association scan in the old order amish: Evidence for replication from diabetes-related quantitative traits and from independent populations. Diabetes. 2007;56:3053-3062. 17. Rich SS, Bowden DW, Haffner SM, Norris JM, Saad MF, Mitchell al. Identification ll BD, D ett al l. Id Identi tifi ti ficca fi of quantitative trait loci for glucose homeostasis: The insulin resistancee atherosclerosis ather eros er oscl os cleero cl rosi siss study si stud st u (iras) family study. Diabetes. y st tud u y. Diab Di ia ettes e . 2004;53:1866-1875. 18. Ji Y, Hebbring Glycine ebb ebb bbring S, Zhuu H, H, Jenkins Jenki kiins n GD, GD, Biernacka Biernnackaa J, J, Snyder Snyd Sn ydeer K, yd K, et aal. l.. G ly ycinee aand nd a gglycine ly ycii dehydrogenase n se (g nas ((gldc) ldc)) ssnp npp aass ccitalopram/escitalopram ittalo opr pram a /e /esccittalop opram op m response resp sponnsee bbiomarkers sp io omaarkkerrs in i ddepression: epreession: ep n: Pharmacometabolomics-informed pharmacogenomics. Ther. m bol metab o om omiicss-iinf nfor forme med ph phar armaco ar oge geno n miics no cs. Cl Clin in Pharmacol Pharm rmacoll The rm h r. 22011;89:97-104. he 0111;8 89: 9 97 97-11 19. Levitzky MJ, ky YS,, Pencina M ky J, D'Agostino D'A Ago g stino RB, RB,, Meigs RB M ig Me igs JB, JB, Murabito M rabi Mu b to JM, bi JM, Vasan Vasan RS,, et al. Impact Im m of impaired disease: The study. Coll d fasting glucose glluc gluc ucos osee onn ccardiovascular os arrdi diov ovvas ovas ascu cu cula ula larr di dise seeas seas ase: e: T hee fframingham rami ra mingha ming gham ham he hheart artt stud ar st tud udy. J Am C Cardiol. 2008;51:264-270. 008;51:264 008 08;51 51:26 2644 27 2700 20. Johnson JA, Boerwinkle E, Zineh I, Chapman AB, Bailey K, Cooper-DeHoff RM, et al. Pharmacogenomics of antihypertensive drugs: Rationale and design of the pharmacogenomic evaluation of antihypertensive responses (pear) study. Am Heart J. 2009;157:442-449. 21. Wallace TM, Levy JC, Matthews DR. Use and abuse of homa modeling. Diabetes Care. 2004;27:1487-1495. 22. Fiehn O, Wohlgemuth G, Scholz M, Kind T, Lee do Y, Lu Y, et al. Quality control for plant metabolomics: Reporting msi-compliant studies. Plant J. 2008;53:691-704. 23. Scholz M, Fiehn O. Setupx--a public study design database for metabolomic projects. Pac Symp Biocomput. 2007:169-180. 24. American Diabetes Association. Standards of medical care in diabetes--2014. Diabetes Care. 2014;37 Suppl 1:S14-80. 25. Huffman KM, Shah SH, Stevens RD, Bain JR, Muehlbauer M, Slentz CA, et al. Relationships between circulating metabolic intermediates and insulin action in overweight to 17 DOI: 10.1161/CIRCGENETICS.113.000421 obese, inactive men and women. Diabetes Care. 2009;32:1678-1683. 26. Galwey NW. A new measure of the effective number of tests, a practical tool for comparing families of non-independent significance tests. Genet Epidemiol. 2009;33:559-568. 27. Purcell S, Neale B, Todd-Brown K, Thomas L, Ferreira MA, Bender D, et al. Plink: A tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet. 2007;81:559-575. 28. Lyssenko V, Almgren P, Anevski D, Perfekt R, Lahti K, Nissén M, et al. Predictors of and longitudinal changes in insulin sensitivity and secretion preceding onset of type 2 diabetes. Diabetes. 2005;54:166-174. Downloaded from http://circgenetics.ahajournals.org/ by guest on May 6, 2017 29. Kim JA, Wei Y, Sowers JR. Role of mitochondrial dysfunction in insulin resistance. Circ Res. 2008;102:401-414. 30. Felig P, Marliss E, Cahill GF. Plasma amino acid levels and insulin secretion n secret tion in i obesity. obeesi sitty N Engl J Med. 1969;281:811-816. 31. Newgard CB. Interplay between lipids rd C rd B. Inte errpplay laay be etw twee eeen lipi li ipi pids dss aand ndd bbranched-chain r ncched-ch ra ch chai hai a n am aamino inoo ac in aacids idss in in ddevelopment ev vel elop opme op m ntt of insulin resistance. Metab. stan st nce. Cell M etabb. 22012;15:606-614. 0122;15::6066-6144. 32. Newgard r CB rd CB, An A J, Bain B in Ba in JR, JR, Muehlbauer Mue uehl ue hlbbaueer MJ, hl MJ Stevens MJ Stev St even enss RD, en RD Lien Lien en LF, LF, F, et al. al. l A bbranched-chain ranc ra nche h dhe amino acid-related metabolic signature contributes -related metabo oli licc si sign gn nat a urre th tthat at ddifferentiates ifffe fere reent ntia iaate tess obese obbese esse an aand d le lean a humans an hum umans and contr r to insulin resistance. Cell Metab. e esistance. Metabb. 20 22009;9:311-326. 099;9 9:3 311 1 -326 2 . 26 33. Shah SH, MJ, Stevens RD, DR, Haynes C, et al. Association H Bain Baii JR, JR Muehlbauer M ehlba ehlb hlb er MJ M J Ste S te ens RD R D Crosslin C li DR Ha Ha nes C all A Associa s cia i of a peripheral blood metabolic profile with coronary artery disease and risk of subsequent cardiovascular events. Circ Cardiovasc Genet. 2010;3:207-214. 34. Tremblay F, Marette A. Amino acid and insulin signaling via the mtor/p70 s6 kinase pathway. A negative feedback mechanism leading to insulin resistance in skeletal muscle cells. J Biol Chem. 2001;276:38052-38060. 35. Shah SH, Svetkey LP, Newgard CB. Branching out for detection of type 2 diabetes. Cell Metab. 2011;13:491-492. 36. Hsueh WC, Mitchell BD, Aburomia R, Pollin T, Sakul H, Gelder Ehm M, et al. Diabetes in the old order amish: Characterization and heritability analysis of the amish family diabetes study. Diabetes Care. 2000;23:595-601. 37. Chobanian AV, Bakris GL, Black HR, Cushman WC, Green LA, Izzo JL, et al. Seventh report of the joint national committee on prevention, detection, evaluation, and treatment of high blood pressure. Hypertension. 2003;42:1206-1252. 38. Jacob S, Rett K, Henriksen EJ. Antihypertensive therapy and insulin sensitivity: Do we have 18 DOI: 10.1161/CIRCGENETICS.113.000421 to redefine the role of beta-blocking agents? Am J Hypertens. 1998;11:1258-1265. 39. Bakris GL, Fonseca V, Katholi RE, McGill JB, Messerli FH, Phillips RA, et al. Metabolic effects of carvedilol vs metoprolol in patients with type 2 diabetes mellitus and hypertension: A randomized controlled trial. JAMA. 2004;292:2227-2236. 40. Celik T, Iyisoy A, Kursaklioglu H, Kardesoglu E, Kilic S, Turhan H, et al. Comparative effects of nebivolol and metoprolol on oxidative stress, insulin resistance, plasma adiponectin and soluble p-selectin levels in hypertensive patients. J Hypertens. 2006;24:591-596. 41. 2010 top 200 generic drugs by total prescription. 2011;2012:http://drugtopics.modernmedicine.com/drugtopics/data/articlestandard//drugtopics/25 2011/727243/article.pdf. Downloaded from http://circgenetics.ahajournals.org/ by guest on May 6, 2017 42. Shin J, Johnson JA. Pharmacogenetics of beta-blockers. Pharmacotherapy. otherappy. 2007;27: 2007;27:874-887. :87 8 Table 1 Baseline a eliine character ase characteristics erristiics off the th co ccohort ohoort w with itth me metabolomics metabo bolo bo lom lo miccs data, dataa, according acc ccordiing cc n too IFG G sta status a Characteristic i istic European American A i ((n, %)) Women (n,%) Current smoker (n,%) Mean age, years Mean BMI, kg/m2 Mean waist circ., cm Mean SBP, mmHg Mean DBP, mmHg Median Fasting glucose, mg/dL Median Fasting insulin, μIU/mL Mean Potassium, meq/L Median HOMA IR Develope ped IFG Developed (n=2 (n =24) =2 4)) (n=24) Diid Not Develop IFG Did ((n=98) (n n=98) 24, 10 24 1000 98, 10 98 1000 6, 25* 3, 13 52.5 (9) 30.5 (4.5) 98.5 (11) 143 (9) 92 (5) 93 (86.0-98.0)* 8.3 (4.8-11.2) 4.5 (0.40) 1.86 (1.06-2.56) 62, 63* 13, 13 49.6 (10) 30.3 (6.2) 96.6 (13) 145(10) 92 (6) 88.3 (83.5-91.5)* 6.6 (4.9-9.6) 4.3 (0.34) 1.42 (1.05-1.97) Continuous variables that are normally distributed (age, BMI, waist circumference, SBP and DBP) are presented as mean (standard deviation). Continuous variables such as glucose, insulin and HOMA are not normally distributed and are presented as median (interquartile range). Categorical variables were presented as number and percentages. BMI=body mass index, SBP=systolic blood pressure, DBP=diastolic blood pressure, HOMA-IR=homeostatic model assessment-insulin resistance. * denotes a p value < 0.05 by using Chi-square or Wilcoxon rank sum test to compare frequency between those who did and did not develop IFG. 19 DOI: 10.1161/CIRCGENETICS.113.000421 Baseline AA Table 2: Association between baseline and percent change in glucose following treatment with atenolol. Beta coefficients were from multiple regression adjusted for baseline glucose, baseline insulin, HOMA-IR, gender, age and body mass index (BMI). Downloaded from http://circgenetics.ahajournals.org/ by guest on May 6, 2017 Beta Coefficient (SE) P value Ile 0.31 (0.08) 0.0003 Leu 0.28 (0.08) 0.001 Val 0.24 (0.08) 0.004 Tyr 0.21 (0.068) 0.01 Phe 0.18 (0.08) 0.03 03 SE= standard error, Ile=isoleucine, Leu=leucine, Val=valine, Tyr=tyrosine, Phe=p Phe=phenylalanine phe heny nyla ny lala la lani la nine ni ne Figure Legends: gends: Figure 1. Odds ratio and 95% Confidence Intervals (per standard deviation) for baseline amino acid level and development of impaired fasting glucose (IFG) from logistic regressions adjusted for age, gender, BMI, baseline fasting glucose and insulin and HOMA-IR (model 4), n=122. BMI = Body Mass Index, HOMA-IR=Homeostatic Model Assessment – Insulin Resistance, Ile=isoleucine, Leu=leucine, Val=valine, Tyr=tyrosine, Phe=phenylalanine Figure 2. From the genotyped population, incidence of impaired fasting glucose according to the top PAH SNP, n=184. IFG=impaired fasting glucose. 20 Downloaded from http://circgenetics.ahajournals.org/ by guest on May 6, 2017 Downloaded from http://circgenetics.ahajournals.org/ by guest on May 6, 2017 Downloaded from http://circgenetics.ahajournals.org/ by guest on May 6, 2017 Is a Diabetes-Linked Amino Acid Signature associated with Beta Blocker-Induced Impaired Fasting Glucose? Rhonda M. Cooper-DeHoff, Wei Hou, Liming Weng, Rebecca A. Baillie, Amber L. Beitelshees, Yan Gong, Mohamed H.A. Shahin, Stephen T. Turner, Arlene Chapman, John G. Gums, Stephen H. Boyle, Hongjie Zhu, William R. Wikoff, Eric Boerwinkle, Oliver Fiehn, Reginald F. Frye, Rima Kaddurah-Daouk and Julie A. Johnson Circ Cardiovasc Genet. published online March 13, 2014; Circulation: Cardiovascular Genetics is published by the American Heart Association, 7272 Greenville Avenue, Dallas, TX 75231 Copyright © 2014 American Heart Association, Inc. All rights reserved. Print ISSN: 1942-325X. Online ISSN: 1942-3268 The online version of this article, along with updated information and services, is located on the World Wide Web at: http://circgenetics.ahajournals.org/content/early/2014/03/13/CIRCGENETICS.113.000421 Data Supplement (unedited) at: http://circgenetics.ahajournals.org/content/suppl/2014/03/13/CIRCGENETICS.113.000421.DC1 Permissions: Requests for permissions to reproduce figures, tables, or portions of articles originally published in Circulation: Cardiovascular Genetics can be obtained via RightsLink, a service of the Copyright Clearance Center, not the Editorial Office. Once the online version of the published article for which permission is being requested is located, click Request Permissions in the middle column of the Web page under Services. Further information about this process is available in the Permissions and Rights Question and Answer document. Reprints: Information about reprints can be found online at: http://www.lww.com/reprints Subscriptions: Information about subscribing to Circulation: Cardiovascular Genetics is online at: http://circgenetics.ahajournals.org//subscriptions/ SUPPLMEMENTAL MATERIAL Supplement Methods Genotyping and Quality Control Participants were excluded if sample genotype call rates were below 95% and SNPs were excluded if genotype call rates were below 90%. The genotype data were not reclustered after QC filters, but the genotype and sample call rate was recalculated after QC. Sample contamination was detected by checking gender mismatches using X chromosome genotype data and cryptic relatedness was estimated by pairwise identityby-descent (IBD) analysis implemented using PLINK (http://pngu.mgh.harvard.edu/purcell/plink/).1 Heterozygosity was also assessed using PLINK, by estimating the inbreeding coefficient, F. After the QC procedures, the total SNP call rate in the remaining individuals was 99.519%. Hardy-Weinberg equilibrium was assessed using with chi-square tests with one degree of freedom. To address the issue of population substructure and admixture in our racially and ethnically diverse population, a Principal Component Analysis (PCA) was performed in all subjects on a linkage disequilibrium (LD) pruned dataset using the EIGENSTRAT method.2 Race/ethnic groups were confirmed with PCA clustering results. If race/ethnic category disagreed strongly, patients were re-categorized to reflect the PCA result, considered to better reflect genetic ancestry. The top principal components (PCs 1-2) that provided the best separation of ancestry clusters were selected to be included as covariates for analysis. Supplement References 1. 2. Purcell S, Neale B, Todd-Brown K, Thomas L, Ferreira MA, Bender D, et al. Plink: A tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet. 2007;81:559-575. Price AL, Patterson NJ, Plenge RM, Weinblatt ME, Shadick NA, Reich D. Principal components analysis corrects for stratification in genome-wide association studies. Nat Genet. 2006;38:904-909. Supplement Table 1 Association between baseline amino acid level (per standard deviation) and odds for developing impaired fasting glucose following treatment with atenolol Ile Leu Val Tyr Phe Model 1 OR (95% CI), p value Model 2 OR (95% CI), p value Model 3 OR (95% CI), p value 1.91 (1.23-2.96), 0.004 1.69 (1.10-2.58), 0.017 1.68 (1.09-2.59), 0.020 1.64 (1.06-2.54), 0.028 1.62 (1.03-2.54), 0.037 1.64 (1.03-2.62), 0.037 1.46 (0.93-2.28), 0.101 1.54 (0.97-2.45), 0.068 1.68 (1.05-2.71), 0.032 1.67 (1.03-2.71), 0.039 2.28 (1.29-4.01), 0.004 1.76 (1.07-2.88), 0.025 1.76 (1.06-2.91), 0.027 2.03 (1.16-3.56), 0.014 2.01 (1.14-3.54), 0.016 Model 4 OR (95% CI), p value 2.29 (1.31-4.01), 0.0034 1.80 (1.10-2.96), 0.019 1.77 (1.07-2.92), 0.025 2.13 (1.20-3.78), 0.010 2.04 (1.16-3.59), 0.014 Model 1 = unadjusted, model 2 = model 1+ age, sex, BMI, model 3 = model 2 + baseline glucose and baseline insulin, model 4 = model 3 + HOMA IR. Each amino acid level is included in the models as a continuous variable. Values are odds ratios per standard deviation (95% confidence intervals) and p values for impaired fasting glucose from an unadjusted logistic regression model and conditional logistic regression models adjusted for baseline fasting glucose and baseline fasting insulin or baseline fasting glucose and baseline fasting insulin. Ile=isoleucine, Leu=leucine, Val=valine, Tyr=tyrosine, Phe=phenylalanine Supplement Table 2 Association between baseline amino acid level (per standard deviation) and odds for developing impaired fasting glucose following treatment with atenolol according to gender Women Men n=68 (6 developed IFG and 62 n=54 (18 developed IFG and 36 did did not develop IFG) not develop IFG) OR (95% CI), p value OR (95% CI), p value Ile 3.67 (0.84-16.35), 0.084 2.06 (1.09-3.89), 0.025 Leu 1.90 (0.51-7.05), 0.339 1.75 (0.97-3.16), 0.063 Val 1.36 (0.52-3.58), 0.531 1.85 (0.97-3.54), 0.062 Tyr 1.18 (0.38-3.66), 0.780 2.47 (1.15-5.32), 0.020 Phe 1.36 (0.46-4.05), 0.576 2.25 (1.07-4.72), 0.032 Values are odds ratios (OR) per standard deviation (95% confidence intervals [CI]) and p values for impaired fasting glucose from logistic regression models adjusted for age, body mass index, baseline fasting glucose, baseline fasting insulin and HOMA IR. Each amino acid level is included in the models as a continuous variable. Ile=isoleucine, Leu=leucine, Val=valine, Tyr=tyrosine, Phe=phenylalanine SUPPLEMENT FIGURE LEGENDS Figure 1. Flow diagram of participants included in the analyses. Figure 2. In the 122 participants with metabolomics data and without impaired fasting glucose at baseline, A. Change in glucose following treatment with atenolol according to those who did and did not develop IFG, B. Baseline amino acid levels according to those who did and did not develop IFG for Isoleucine, Leucine, Valine, Tyrosine and Phenylalanine. The data in B are presented as ion counts (measurement unit). Horizontal bars are median and 25th and 75% percentile. Whiskers are 5th and 95th percentile. Supplement Figure 1