* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Environmental Engineering - Pollution: Types, Sources and
Survey
Document related concepts
Transcript
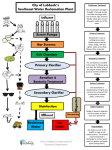
Environmental Engineering Pollution: Types, Sources and Treatments CIEG 125 Introduction to Civil Engineering Fall 2005 Lecture 7 Introduction • Two approaches to pollution: • “end-of-pipe” treatment; environmental remediation and treatment e.g., precipitators and water treatment facilities at power plants • pollution prevention (e.g, using hydropower) Outline • • • • • • • Introduction Municipal Solid Waste Hazardous Waste Municipal Wastewater (i.e., sewage) Drinking Water Air Pollution Summary Municipal Solid Waste in U.S. • 1989: 4 lbs of garbage per person-day • 200 million tons of garbage per year • Composition: • • • • • ~40% (by weight) is paper ~20% is glass and metal ~25% is food/yard waste ~10% is other waste ~5% is plastic • 70% MSW to landfills, 15% is recycled Hazardous Waste • Waste defined as hazardous if: • • • • 10/24/2005 ignitable corrosive reactive toxic (e.g., gasoline) (e.g., acids) (e.g., cleaning solvents) (e.g., cadmium) 1 Hazardous Waste • In US, between 250 and 750 million tons of HW generated each year • Majority of HW generated by: • chemical processing industry • petroleum/coal processing • metals processing • 90% is wastewater • Hazardous constituents only 1-2% Secure Landfill Methods for Treating HazardousWaste • Biological Treatment - novel microorganisms that breakdown material • Chemical treatment that transforms the material into a less toxic form • Carbon Adsorption • Incineration • Deep Well Injection • Secure Landfills Municipal Wastewater (Sewage) • Physical Characteristics: • 1 m3 weighs about 3 kg • 500 g are solids: • 250 g are dissolved solids such as Ca, Na • 125 g are insoluable solids that will settle out in about 30 min in quiescent conditions • 125 g are insoluable solids that will remain in suspension causing high turbidity States of Solution Impurities Dissolved Substances • Substances can exist in water in one of three classifications: • Dissolved substances can be atoms or complex molecular compounds. • Removed from solution by: • dissolved - a substance truly in solution that can’t be removed without a phase change • suspended - suspended solid particles are large enough to settle out or be removed by filtration • colloidal - colloidal particles are very small particles not able to be removed by filtration, but not dissolved in the water. 10/24/2005 • distillation - substance or water is changed to gas • precipitation - substance combines with chemical to form a solid and settle out • adsorption - substance reacts with a solid particle, such as granular activated carbon (GAC) 2 Suspended Solids Colloidal Particles • Suspended solids are removed from water by physical methods: • Small particles that are not removable by physical means. Removed by: • high-force centrifugation • filtration through membranes w/ small pores • addition of coagulant and flocculation • sedimentation • filtration • centrifugation • Cause turbidity - I.e., cloudiness • degree to which a colloidal suspension reflects light at 90 degrees to the entrance beam. Coagulation and Flocculation BOD and COD • To remove colloids from a water: • Wastewater contains materials that oxidize (transform into CO2 and H2O using O2) • These materials thus demand oxygen (O2), but so do fish and other waterborne life • If the materials are oxidized by microorganisms, called biological oxygen demand or BOD • If they are oxidized by direct chemical reaction, called chemical oxygen demand or COD • a coagulant is added to the water • coagulant must be nontoxic, insoluable • two most common: alum and ferric iron • The coagulant • reacts with the water, • forms large complexes, called flocs (process called flocculation) • flocs enmesh the colloid particles • flocs settle out of solution. Municipal Wastewater (Sewage) • Chemical Characteristics: • • • • • • • Alkalinity (CaCO3) Biochemical Oxygen Demand Chemical Oxygen Demand Chloride Nitrogen Carbon Phosphorous Activated Sludge Treatment (medium) 100 mg/L 200 mg/L 500 mg/L 50 mg/L 40 mg/L 150 mg/L 10 mg/L • BOD material removed by a microbiological process. • Bacteria added to wastewater and aerated. • The bacteria grow and form activated sludge • The bacteria consume the BOD material • Sludge is settled out of water and recycled. • Pathogenic organisms 10/24/2005 3 Wastewater Treatment Wastewater Treatment Drinking Water Quality Factors Some Sources of Water Pollution • Physical: color, turbidity, taste, oder • Chemical: hardness, chlorides, flourides, iron, lead, toxic inorganic substances (nitrates, cyanides and heavy metals), toxic organic substances ... • Microbiological: disease producing orgs • Radiological: radioactive substances • • • • • Drinking Water Treatment Coagulant (e.g., alum) added to turbid water in rapid mix tank. Mix is slowly stirred in the flocculation basin, where larger particles of precipitation (called flocs) are formed. Flocs are then settled out in sedimentation basin. 10/24/2005 Acidified water from mine drainage Runoff of soil from agricultural areas Animal residues from feedlots/farms Pesticides, herbicides and fertilizers Effluents from industrial materials processing (chemicals, metals, paper and petroleum) Types of Air Pollution • Chloroflourocarbons (CFCs) that deplete the ozone layer (used for degreasing) • Halons deplete ozone (for fire suspression) • Carbon Monoxide (CO): silent killer • Carbon Dioxide (CO2): green house gas • Methane (CH4): green house gas • Nitrous Oxide (N2O) depletes ozone 4 Types of Gaseous Pollution • VOCs (Volatile Organic Compounds) which cause smog and cancer • NOx (Nitric Oxide, NO, and Nitrogen Dioxide NO2) which result from high-temperature combustion in air, forms smog • SO2 (Sulfur Dioxide) from paper production and fossil fuel combustion • Odorants: amines (fish), sulfides (rotten eggs), and mercaptans (skunk spray) Methods for Treating Air Emissions • • • • • • • Sources of Gaseous Pollution • Combustion of fossil fuels generating SO2, NOx, CO2 • Use and evaporation of solvents • Generation of volatile compounds and odorants by chemical processes Scrubber System Settling chambers for particulate matter Scrubbers (gas through mist) Precipitators (gas through EMF) Absorber systems (pollutant dissolved) Adsorber systems (pollutant retained) Condensors Flares and Incinerators Absorber Tower System 10/24/2005 Adsorber System 5 Summary • There are a variety of types of pollutants • They find their way into land, water, groundwater and air • There are a variety of physical, biological and chemical approaches to removing pollutants from these media 10/24/2005 6