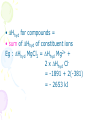* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chemical Energetics
Biochemistry wikipedia , lookup
Click chemistry wikipedia , lookup
Chemical reaction wikipedia , lookup
Atomic theory wikipedia , lookup
Physical organic chemistry wikipedia , lookup
History of molecular theory wikipedia , lookup
Double layer forces wikipedia , lookup
Acid dissociation constant wikipedia , lookup
Hydrogen-bond catalysis wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
Marcus theory wikipedia , lookup
Acid–base reaction wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Lewis acid catalysis wikipedia , lookup
Chemical equilibrium wikipedia , lookup
Spin crossover wikipedia , lookup
Resonance (chemistry) wikipedia , lookup
Bioorthogonal chemistry wikipedia , lookup
Electrochemistry wikipedia , lookup
Thermometric titration wikipedia , lookup
Chemical bond wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
George S. Hammond wikipedia , lookup
Stability constants of complexes wikipedia , lookup
Chemical thermodynamics wikipedia , lookup
Hypervalent molecule wikipedia , lookup
Stoichiometry wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
ENERGETICS /THERMOCHEMISTRY (AS) • 1.Often chemical changes are accompanied by changes in heat content / enthalpy of the materials reacting (H) • 2. This change is shown by a change in temperature. • a. heat is lost to the surroundings • Temperature of reaction mixture rises / increases • H is negative • Exothermic reaction • Enthalpy diagram / energy level diagram OR Reaction pathway/reaction coordinate/extent of reaction • b. heat is absorbed from the surroundings , temperature of reaction mixture decreases / falls • H is positive • Endothermic reaction • 3. a. The value of H depends on temp , pressure and concentrations of reactants. • b. H are measured under standard conditions : • Temperature = 298K ( 250 C ) • Pressure = 1 atm / 1.01 x 105 Pa • Concentrations = 1 mol dm-3 • c. Any H measured under standard conditions is described as standard enthalpy change. • Symbol : H STANDARD ENTHALPIES Definitions State symbols must be included in equations STANDARD ENTHALPHY OF FORMATION ( Hf ) Enthalpy change when 1 mole of a compound is formed from its elements in their standard states , under standard conditions temperature 298 K and pressure of gases at 1 atm • Steps in writing equations: • 1. write formula of compound formed • 2. identify elements required to form the compound • 3. balance equation to form 1 mole of the compound Examples • Mg (s) + ½O2 (g) → 1MgO (s) -602 kJ mol-1 602 kJ evolved for every 1 mole of MgO formed • ½ H2 (g) + ½ Cl2 (g) → 1HCl (g) - 92.3 kJ mol-1 Examples Notes : 1) Hf of elements is zero Eg : Cu(s) Cu(s) , Hf = 0 2) Hf are often theoretical only • Exercise : Write the equation for enthalpy of formation for : KMnO4 (s) and H2O (l) • K(s) + Mn(s) + 2O2(g) 1KMnO4(s) • H2(g) + ½O2(g) 1H2O (l) STANDARD ENTHALPY CHANGE OF COMBUSTION ( Hc ) Enthalpy evolved when 1 mole of the element or compound is completely burned in excess oxygen , under standard conditions • Steps in writing equations : • 1. identify products formed from burning of compound in excess oxygen • 2. balance equation for 1 mole of compound burnt Examples • 1 C (s) + O2 (g) → CO2 (g) Hc = - 394 kJ mol-1 394 kJ evolved for every 1 mole of carbon burnt • 1 H2 (g) + ½O2 (g) → H2O (l) •Exercise : Write an equation for the enthalpy of combustion for C3H6 and CH3OH • 1C3H6 (g) + 9/2 O2 (g) 3CO2 (g) + 3H2O (l) • 1CH3OH (l) + 3/2 O2 (g) CO2 (g) + 2H2O (l) STANDARD ENTHALPY OF ATOMISATION ( Hat ) • Enthalpy required/absorbed • 1 mole of gaseous atoms formed • From the element in its standard state under standard conditions • Examples : • Fe (s) 1 Fe (g) • Example : • ½Cl2(g) 1 Cl (g) + 122 kJ mol-1 • molecules atoms • Note : from Data Booklet • Bond energy Cl-Cl = + 244 kJ mol-1 • Cl-Cl 2 Cl (g) 2 moles atoms • Hat of Cl = ½ x bond energy Cl-Cl STANDARD BOND DISSOCIATION ENTHALPY • Also called bond energy • Energy absorbed • Separate the 2 atoms in a covalent bond in gaseous state under standard conditions , per mole of bond. Examples • 1) HCl (g) → H (g) + Cl (g) , +431 kJmol-1 • 2) CH4 contains 4 x C-H bond • Total energy required to break all bonds in CH4 = 1640 kJ • ¼ CH4 = ¼ ( 4 C-H bond ) = one C-H bond • Average bond energy of one C-H bond • = ¼ ( 1640 ) = +410 kJ • ¼ CH4 (g)→¼ C (g) + H (g),+ 410 kJmol-1 STANDARD ENTHALPY OF NEUTRALIZATION • Energy evolved • Acid reacts with base to form 1 mole of water , under standard conditions • • • • Examples : NaOH + HCl → NaCl + 1 H2O KOH + HCl → KCl + 1 H2O Ionic equation(strong acid + strong base: H+(aq) + OH-(aq) 1 H2O (l), H neutralisation = -57 kJ mol-1 • Generally, • 1) Strong acid / strong alkali : • H = - 57 kJ mol-1 • 2) Weak acid / base : • H = -(<57) kJ mol-1 (eg. -54 kJmol-1) • Reason : Certain amount of energy required to ionise the weak acid or base first STANDARD ENTHALPY OF HYDRATION ( Hhyd ) • Energy evolved • 1 mole of separate gaseous ions dissolved in water under standard conditions • Exothermic as attraction/bond forms between the ions and polar water molecules • Called ion dipole attraction • Examples : • 1 Na+ (g) → Na+ (aq) • 1 Cl- (g) → Cl- (aq) -406 kJmol-1 - 381 kJmol-1 • Hhyd for compounds = • sum of Hhyd of constituent ions Eg : Hhyd MgCl2 = Hhyd Mg2+ + 2 x Hhyd Cl= -1891 + 2(-381) = - 2653 kJ • H (hyd) charge density of ions • Charge density = charge/size • Higher charge density , stronger ion dipole attraction , more exothermic H(hyd) • Eg : • H(hyd) Cl- > H(hyd) Br – • -381 -351 STANDARD ENTHALPY OF SOLUTION ( Hsolution ) • Enthalpy change when 1 mole of a substance dissolved in a stated amount of solvent under standard conditions • Example : • 1 KOH (s) K+ (aq) + OH- (aq) or KOH (aq) - 57 kJ mol-1 STANDARD ENTHALPY CHANGE OF REACTION ( Hr ) • Enthalpy change in a chemical reaction , for the number of moles of reactants as shown in a balanced chemical equation under standard conditions • Example : 4 H2O + 3 Fe Fe3O4 + 4 H2 Hr = x kJ when 4 moles H2O reacts with 3 moles Fe