* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Germ cells are the only cells which are transmitted from one
Survey
Document related concepts
Transcript
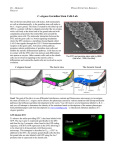
Germ Cell Development U934/UMR3215 – Genetics and Developmental Biology Jean-René Huynh Chef d'équipe Tel: +33 (0)1 56 24 69 47 Germ cells are the only cells which are transmitted from one generation to the next and can be considered immortal. Germ cells produce highly specialized cells, called gametes, which carry the genetic and cytoplasmic information defining a given species and which can initiate the formation of an entire organism. Understanding how germ cells develop is not only of paramount medical interest for reproductive medicine, but is also crucial to comprehend how animal shapes and forms evolve through generations. Drosophila adult females present several key advantages as a model system to study germ cell development. In each ovary, there are germline stem cells (GSCs), which produce eggs (the female gamete) throughout the female life. It is thus possible to follow the entire development of germ cells from stem cell to fertilized egg in a single fly. Drosophila geneticists also keep on generating ever-more refined tools to control the function of every gene in the genome. It is for example possible to switch off one specific gene in germ cells at one precise time. In females, GSCs are located at the anterior apex of a specialized structure called the germarium. GSCs divide asymmetrically leading to the formation of a self-renewing GSC and a differentiating cystoblast. The cystoblast then undergoes four rounds of asymmetric divisions with incomplete cytokinesis, leading to the formation of a cyst of 16 germline cells interconnected by cytoplasmic bridges called ring canals. Only one cell becomes the egg and goes through meiosis, while the 15 remaining cells become polyploidy nurse cells. Despite representing an excellent model system to study stem cell biology, cell cycle control, meiosis, cell fate determination or cell polarity, the germarium remains poorly explored. Our lab has taken a forward genetic strategy to find the genes regulating these different processes. We have mutagenized randomly the genome and generated mutant lines, which are defective in the formation of the germline. Based on this collection of mutants, we address the following questions: 1) How is germline stem cell growth regulated? GSCs divide actively and need to recover their initial volume/mass quickly after each division. We found that specialized mechanisms are INSTITUT CURIE, 20 rue d’Ulm, 75248 Paris Cedex 05, France | 1 Germ Cell Development U934/UMR3215 – Genetics and Developmental Biology involved. 2) How is the duration of cytokinesis regulated in germ cells? We identified mutations in Drosophila Aurora-B and Cyclin-B genes, which regulate complete abscission in germline stem cells and incomplete abscission in differentiating germ cells. 3) How do homologue chromosomes find each other during meiosis? We are studying the potential role of the cytoplasmic cytoskeleton in regulating chromosome organization during the early steps of meiosis in the germarium. 4) How is the germline genetic material protected from DNA damages? The genetic information contained in the female gamete needs to be safely transmitted to the next generation. We are studying how mobile DNA elements such as transposons are silenced in germline cells. Key publications Year of publication 2015 Anahi Molla-Herman, Ana Maria Vallés, Carine Ganem-Elbaz, Christophe Antoniewski, Jean-René Huynh (2015 Oct 17) tRNA processing defects induce replication stress and Chk2-dependent disruption of piRNA transcription. The EMBO journal : 3009-27 : DOI : 10.15252/embj.201591006 Nicolas Christophorou, Thomas Rubin, Isabelle Bonnet, Tristan Piolot, Marion Arnaud, Jean-René Huynh (2015 Oct 13) Microtubule-driven nuclear rotations promote meiotic chromosome dynamics Nature cell biology : 1388-400 : DOI : 10.1038/ncb3249 Neuza Reis Matias, Juliette Mathieu, Jean-René Huynh (2015 Feb 3) Abscission is regulated by the ESCRT-III protein shrub in Drosophila germline stem cells. PLoS genetics : e1004653 : DOI : 10.1371/journal.pgen.1004653 Year of publication 2013 Nicolas Christophorou, Thomas Rubin, Jean-René Huynh (2013 Dec 19) Synaptonemal complex components promote centromere pairing in pre-meiotic germ cells. PLoS genetics : e1004012 : DOI : 10.1371/journal.pgen.1004012 INSTITUT CURIE, 20 rue d’Ulm, 75248 Paris Cedex 05, France | 2 Germ Cell Development U934/UMR3215 – Genetics and Developmental Biology Juliette Mathieu, Clothilde Cauvin, Clara Moch, Sarah J Radford, Paula Sampaio, Carolina N Perdigoto, François Schweisguth, Allison J Bardin, Claudio E Sunkel, Kim McKim, Arnaud Echard, Jean-René Huynh (2013 Aug 12) Aurora B and cyclin B have opposite effects on the timing of cytokinesis abscission in Drosophila germ cells and in vertebrate somatic cells. Developmental cell : 250-65 : DOI : 10.1016/j.devcel.2013.07.005 Year of publication 2008 Pierre Fichelson, Clara Moch, Kenzo Ivanovitch, Charlotte Martin, Clara M Sidor, Jean-Antoine Lepesant, Yohanns Bellaiche, Jean-René Huynh (2008 Nov 14) Live-imaging of single stem cells within their niche reveals that a U3snoRNP component segregates asymmetrically and is required for self-renewal in Drosophila. Nature cell biology : 685-93 : DOI : 10.1038/ncb1874 INSTITUT CURIE, 20 rue d’Ulm, 75248 Paris Cedex 05, France | 3