* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Periodic Table[1]
Survey
Document related concepts
Transcript
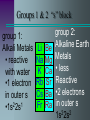
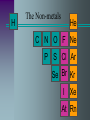
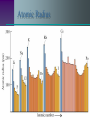
The Periodic Table Objectives Discuss how the periodic table is organized. Use the periodic table to predict atomic behavior. Predict characteristics of an element based on its location. Dmitri Mendeleev 1873 • Was trying to establish a pattern in the 63 known elements. • Used atomic mass, physical and chemical properties. • He arranged and rearranged, until he came to this, the first periodic table. Periodic Law Mendeleev’s table based on increasing atomic mass was useful but didn’t work with all elements. Modern Periodic Table is arranged according to increasing Atomic Number When elements are arranged according to increasing atomic number, there is a periodic repetition of their physical and chemical properties. Modern Periodic Chart Atomic # Symbol Name Atomic Mass 6 C Carbon 12.011 Increases in size, mass, & atomic # Metals Non-metals Metals versus Non-Metals Metals –good conductors –all solids (ex. mercury) Non-metals –poor conductors – solid, liquid, gas Main Group Elements Main-Group Elements The electron configuration of these elements in quite consistent. All elements are filling the s and p orbitals Chemical properties of each vertical group or “family” are similar due to the number of electrons in their outer energy level. Groups 1 & 2 “s” block group 1: Alkali Metals • reactive with water •1 electron in outer s 2 1 •1s 2s Li Na K Rb Cs Fr Be Mg Ca Sr Ba Ra group 2: Alkaline Earth Metals • less Reactive •2 electrons in outer s 1s22s2 Transition Metals “d” block Sc Y La Ac Ti Zr Hf Rf V Nb Ta Db Cr Mo W Sg Mn Tc Re Bh Fe Ru Os Hs Co Rh Ir Mt Ni Cu Zn Pd Ag Cd Pt Au Hg Uun Uuu “The Southern Island” “f” block Lanthanides: reactive metals with irregular electron configurations Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Am Cm Bk Cf Es Fm Md No Lr Actinides: so unstable that they are radioactive. Those Elements In Between B Metalloids Al Si “Other Ga Ge As Metals” In Sn Sb Te Tl Pb Bi Po H The Non-metals He C N O F Ne P S Cl Ar Se Br Kr I Xe At Rn H “Other Non-metals” C N O These include P S some of the most Se common elements on Earth! Halogens •Halogen means “salt-former”. •Most reactive non-metals. •All are missing Electron in p Sub-level F Cl Br I At 1s22s22p5 The Noble Gases •Basically unreactive because their electron configuration is filled. •P sublevel with 6 electrons •For this reason, noble gases don’t usually bond. He Ne Ar Kr Xe Rn Noble Gases are stable and un-reactive All the other elements accept or donate electrons to attain Noble gas electron configurations Although atoms of other elements are neutral, they lack the full outer energy level needed to be STABLE So how do elements achieve stability? •give electrons •take electrons •share electrons 6.3 Periodic Trends Atomic Radiusdefined as the distance between nuclei of two atoms Down a group radius increases due to the addition of higher energy levels Across a period radius actually decreases due to the additional positive charge of the nucleus as protons a re added Atomic Radius Ions The Loss or gain of an electron results in the formation of a charged atom known as an ion. The charge of Oxidation Number, can be determined by comparing the number of positive protons with negative electrons Cat-ions are Positive charged ions An-ions are negatively charged ions Ionization Energy The energy needed to remove an electron from the atom (in a gaseous state) Ionization energy tends to decrease down a group. Electrons in higher energy levels become easier to remove from the atom as they are farther from the attractive force of the protons. Ionization Energy increases across a period as it becomes more difficult to remove electrons as the energy level fills up Ionization Energy Electronegativity Ability of an atom of an element to attract electrons from another element in forming a compound Electronegativity values decrease from top to bottom down a group Tend to increase across a period from left to right. Noble gases do not attract electrons and have no electronegativity Fluorine has the highest value at 4.0, Cesium the lowest at 0.7 Periodic trends