* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Diurnal variations of the dominant cycle length of chronic atrial
Remote ischemic conditioning wikipedia , lookup
Management of acute coronary syndrome wikipedia , lookup
Mitral insufficiency wikipedia , lookup
Cardiac surgery wikipedia , lookup
Cardiac contractility modulation wikipedia , lookup
Arrhythmogenic right ventricular dysplasia wikipedia , lookup
Lutembacher's syndrome wikipedia , lookup
Electrocardiography wikipedia , lookup
Quantium Medical Cardiac Output wikipedia , lookup
Atrial septal defect wikipedia , lookup
Dextro-Transposition of the great arteries wikipedia , lookup
Ventricular fibrillation wikipedia , lookup
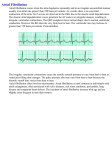
Am J Physiol Heart Circ Physiol 280: H401–H406, 2001. Diurnal variations of the dominant cycle length of chronic atrial fibrillation CARL J. MEURLING,1 JOHAN E. P. WAKTARE,2 FREDRIK HOLMQVIST,1 ANTTI HEDMAN,3 A. JOHN CAMM,2 S. BERTIL OLSSON,1 AND MAREK MALIK2 1 Department of Cardiology, Lund University Hospital, Lund, Sweden; 2Department of Cardiological Sciences, St. George’s Hospital Medical School, London, United Kingdom; and 3Department of Medicine, Kuopio University Hospital, Kuopio, Finland Received 11 February 2000; accepted in final form 2 August 2000 normal sinus rhythm (6, 13). In atrial fibrillation (AF), atrial refractoriness is shortened compared with sinus rhythm, but whether it exhibits diurnal variability is unknown. Heart rate during AF is determined by the atrioventricular (AV) nodal conduction (4), but a modulating effect of atrial inputs has been suggested (22, 26). The aim of this study was to noninvasively detect the presence and to characterize the nature of diurnal variations in atrial electrophysiology during chronic AF. We sought to determine the relationship between fluctuations in AF cycle length and heart rate. The study utilized a noninvasive method, frequency analysis of fibrillatory electrocardiogram (FAF-ECG), which has been validated as a technology for the measurement of atrial cycle length during AF (11). METHODS in humans ranges from shortterm modulations at frequencies corresponding to respiration to significant low-frequency diurnal fluctuations (1, 10, 28). Fluctuations in autonomic nervous system tone acting on the sinus node are an important underlying mechanism (1), but humoral efferent systems are also active (31, 34). However, the whole organism acts as a complex biological system, with diurnal fluctuations in many other parameters such as temperature, blood pressure, metabolism, and activity levels. These may also directly or indirectly alter heart rate. Diurnal fluctuations in atrial refractoriness have been little studied, but it is well recognized that autonomic tone has a profound effect on refractoriness in Study population and data acquisition. Recruited patients had chronic (persistent or permanent) AF of ⬎1 mo duration. Exclusion criteria were pharmacological treatment with -blockers, antiarrhythmic drugs (Ia, Ib, Ic, III), or sympathomimetic agents, diabetes mellitus, overt heart failure, previous cerebrovascular events, or conditions that would possibly cause interference with the autonomic nervous system, such as current alcohol abuse. The study was approved by the local Ethics Committee, and all patients gave written informed consent. The study investigated 21 outpatients (15 men, mean age 64 ⫾ 12 yr, range 34–78 yr). The median duration of AF was 12 mo (range 1–120 mo), and subjects had undergone a mean of one previous unsuccessful cardioversion (none in 6 patients, 1 in 11 patients, 2 in 2 patients, and 3 in 2 patients). Drug therapy included digoxin in 14 patients, an angiotensin-converting enzyme inhibitor in 8, diltiazem in 3, and verapamil in 1. At echocardiography, left ventricular function was normal in 19 patients and slightly impaired in 2. The mean left atrial diameter was 47 ⫾ 7 mm (range 36–66 mm). Long-term ECG was recorded in each patient using a digital three-channel Holter recorder (6632 Altair-DISC recorder, SpaceLabs Burdick, East Syracuse, NY) with a Address for reprint requests and other correspondence: J. E. P. Waktare, Cardiological Sciences, St. George’s Hospital Medical School, Cramner Terrace, London SW17 0RE, UK (E-mail: jwaktare @sghms.ac.uk). The costs of publication of this article were defrayed in part by the payment of page charges. The article must therefore be hereby marked ‘‘advertisement’’ in accordance with 18 U.S.C. Section 1734 solely to indicate this fact. autonomic nervous system; circadian rhythms VARIABILITY OF HEART RATE http://www.ajpheart.org 0363-6135/01 $5.00 Copyright © 2001 the American Physiological Society H401 Downloaded from http://ajpheart.physiology.org/ by 10.220.33.3 on May 5, 2017 Meurling, Carl J., Johan E. P. Waktare, Fredrik Holmqvist, Antti Hedman, A. John Camm, S. Bertil Olsson, and Marek Malik. Diurnal variations of the dominant cycle length of chronic atrial fibrillation. Am J Physiol Heart Circ Physiol 280: H401–H406, 2001.—High-resolution digital Holter recording was carried out in 21 patients (15 men, 64 ⫾ 12 yr) with chronic atrial fibrillation. Dominating atrial cycle length (DACL) was derived by frequency domain analysis of QRST-reduced electrocardiograms. Daytime mean DACL was 150 ⫾ 17 ms, and nighttime mean was 157 ⫾ 22 ms (P ⫽ 0.0002). Diurnal fluctuation in DACL differed among patients: it tended to be virtually absent in those with a short mean DACL, but in those with longer DACL the night-day difference was as much as 23 ms (R ⫽ 0.72, P ⬍ 0.001, correlation of mean DACL to night-day difference). Mean DACL also correlated with ventricular cycle length (R ⫽ 0.40, P ⬍ 0.001), particularly at night (r ⫽ 0.49). The shorter cycle lengths found in this study during the day are consistent with sympathetic and/or other physiological modulation, but since increased vagal tone shortens atrial refractoriness in most models, parasympathetic influences are not likely to play a major role. Alternatively, atrial effective refractory period may not be the sole determinant of atrial cycle length during atrial fibrillation. H402 DIURNAL VARIABILITY OF ATRIAL FIBRILLATION Fig. 1. Graphic summary of the signal processing method. A: within the raw ECG signal, QRS complexes and T waves are identified and optimally aligned. B: once all QRS complexes are identified, an appropriate inverted QRST template is subtracted to leave a residual signal comprising pure atrial fibrillatory activity (C). This signal is subjected to high- and low-pass filtering and is downsampled before the estimation of a frequency power spectrum using fast Fourier transform (D). (See Ref. 11 for full description of the method, including filter and Fourier transform parameters.) RESULTS Data availability. One patient was excluded because the recording contained only 2 h that could be analyzed. The calculations were made on the basis of the remaining 20 patients. The mean duration of analyzable recording was 15.5 h (range 12–16 h). The median starting hour was 1500 (range 0700–1700), and the finishing hour was 0600 (range 0100–2100). Of the potential 310 sequences of 5 min each, a segment suitable for analysis was found in 305 (98%) cases. The median offset from the hour to the beginning of the segments was 5 min (range 0–30 min), with the daytime median offset (7 min) being larger than the nighttime offset (2 min). Downloaded from http://ajpheart.physiology.org/ by 10.220.33.3 on May 5, 2017 sampling rate of 1 kHz and an amplitude resolution of 0.16 V over a ⫾5-mV range for 17 h. Leads were positioned using a novel quasi-orthogonal three-lead configuration (35), which optimizes acquisition of atrial signal in bipolar recordings. For analyses of day-night differences, night was defined as 2330–0630 and day as 0730–2130. Signal processing. The Holter recordings were transferred to a personal computer for standard automated analysis, visual inspection, overreading, and manual correction using commercial software (Vision Premier, SpaceLabs Burdick). A 5-min segment was further chosen in each hour of each recording. The search for such a segment and, subsequently, in the analysis of atrial cycle length was driven by the quality of the original. The same procedure was repeated for each hour of each recording. Initially, the segment of the first 5 min of the hour was taken. If there was low signal-to-noise ratio (i.e., noise amplitude similar to fibrillatory wave amplitude), more than 2% high-amplitude noise (noise ⱖ R-wave amplitude), or baseline wander, the segment was rejected. In such a case, the preceding and following 5-min regions were inspected to identify the closest segment of acceptable quality. If none could be found, the hour was rejected. The selected 5-min segments were analyzed using the FAF-ECG method, which has been described in detail elsewhere (11). Briefly, QRST complexes were subtracted, and frequency domain analysis was performed on the residual ECG signal within the 3- to 12-Hz range using a fast Fourier transform. The peak frequency in the generated spectrum, expressed as a cycle length, constituted the dominant atrial cycle length (DACL). The method is illustrated in Fig. 1. For this analysis the DACL was obtained from a bipolar lead with the positive electrode at a low C1 position (6th intercostal space, right sternal border) and with the indifferent electrode in a left subclavicular position (35). This lead has a superior atrial signal resolution and corresponds approximately to the conventional unipolar V1 lead, in which the DACL has been shown to correlate closely with invasively measured right atrial free wall fibrillatory cycle length (11). All frequency spectra were visually inspected to confirm sufficient quality allowing an accurate DACL determination. Optimum-quality spectra exhibit a single sharp spectral peak in the expected range for DACL (5–11 Hz) with minimum low-frequency components in the 3- to 5-Hz range (⬍50% of DACL power). Spectra were of acceptable quality in the presence of a single spectral peak with low-frequency components with power between 50 and 150% of the DACL power. Where two spectral peaks were present within the 3to 12-Hz band, the component with the highest amplitude was automatically chosen by the FAF-ECG software, but all such cases were manually confirmed. Heart rate was expressed as mean R-R interval, calculated by the commercial Holter software for each 5-min segment. Statistics. Unless otherwise specified, values are means ⫾ SD. Wilcoxon test was used for comparison between paired samples. Spearman ranked correlation coefficients and the U test were used elsewhere. Since there was significant variation between subjects in mean DACL, the hourly DACL values were normalized for selected analyses. This was performed by dividing the individual DACL values by the mean of all valid DACL values for the given subject. P ⬍ 0.05 was considered statistically significant. All statistical analyses were performed using STATISTICA for Windows (version 5.1, StatSoft, Tulsa, OK). DIURNAL VARIABILITY OF ATRIAL FIBRILLATION Manual correction of the DACL was needed on one analyzed segment (the automated analysis identified an outlier peak of a bimodal distribution, while the other peak corresponded to adjacent hours). Diurnal fluctuations in AF cycle length. Mean DACL was 154 ⫾ 20 ms but varied significantly between patients, ranging from 125 to 186 ms, and through the day (Fig. 2). The DACL exhibited significant diurnal fluctuations, with a shorter mean DACL during the day (mean 150 ⫾ 17, range 124–179) than at night (mean 157 ⫾ 22, range 125–195, P ⫽ 0.0002). There was significant interpatient variation in the mean DACL for the recording. The normalized DACL values are presented in Fig. 3. There was a significant correlation between mean DACL and the standard deviation of DACL values for patients (r ⫽ 0.78, P ⫽ 0.00004), with longer DACL being associated with a greater standard deviation Fig. 3. Diurnal fluctuation of the DACL. The DACL has been normalized using the mean DACL for each individual patient. For each hour, the mean, SE, and SD values are shown. The curve is a third-order polynomial fit to the means. (i.e., the DACL variability increased with increasing DACL values). Likewise, the magnitude of the difference between daytime and nighttime mean DACL was strongly related to the mean DACL (Fig. 4). Those patients with short DACL exhibited little diurnal variation, while those with longer mean DACL exhibited night-day differences of up to 23 ms (P ⫽ 0.0004; Fig. 4). Of the patient characteristics studied, none showed a significant relationship with the mean DACL or with the magnitude of the night-day difference (Table 1). Relationship between fluctuations in AF cycle length and heart rate. There was a significant correlation between the mean R-R interval and DACL (r ⫽ 0.30, P ⬍ 0.000001), with long DACL corresponding to long R-R intervals. The correlation was higher during the night (r ⫽ 0.44, P ⫽ 0.000001) than during the day (r ⫽ 0.27, P ⫽ 0.001; Fig. 5). DISCUSSION This study has shown that 1) the AF cycle length during chronic AF exhibits diurnal variability, with longer cycle lengths (i.e., slower AF revolution) occurring at night; 2) there is a correlation between the AF cycle length and ventricular cycle length, particularly at night; and 3) there are marked interpatient differences in the diurnal variability of the AF cycle length, with higher variability in patients with a longer mean AF cycle length. Diurnal rhythms and changes in AF cycle length. The diurnal pattern of variation of atrial cycle length found in this study is in keeping with circadian changes in atrial refractoriness during sinus rhythm (6) and atrial pacing (6, 13). Previous work has shown that atrial cycle length closely correlates with atrial refractory period during AF (15, 17, 36), and the former may therefore be used as an index of the latter. However, this has been tested in laboratory conditions and has not been validated under varying autonomic influences or other diurnal physiological variations. Fig. 4. Relationship between mean DACL and the magnitude of the night-day difference in mean DACL. Downloaded from http://ajpheart.physiology.org/ by 10.220.33.3 on May 5, 2017 Fig. 2. Individual diurnal fluctuations in dominating atrial cycle length (DACL) in 20 patients. H403 H404 DIURNAL VARIABILITY OF ATRIAL FIBRILLATION Table 1. Influence of recorded variables on mean DACL and night-day differences in DACL DACL, ms Feature absent Digoxin treatment ACE inhibitor treatment Normal LV function AF duration ⬎ mean Age ⬎ mean LA diameter ⬎ mean Feature present Mean DACL N-D difference Mean DACL N-D difference 152 159 181 155 149 155 8 8 6 7 7 6 156 147 153 154 170 155 7 6 7 9 9 8 A circadian rhythm is seen in a range of physiological variables, including arterial (24) and pulmonary (19) blood pressure, body temperature, blood pH, hormone levels (25, 31, 34), and autonomic tone (10, 12). Furthermore, there are diurnal differences in levels of physical exertion, respiratory rate, and body posture. A number of these factors are likely to be interrelated and may contribute to diurnal changes in atrial electrophysiology. However, a physiologically plausible direct effector mechanism exists probably for only four modulators, i.e., autonomic modulations, including those due to postural changes, core body temperature, atrial stretch, and hormonal changes. Temperature-induced changes in cardiac refractory period are demonstrable (32) but are mainly seen with major body temperature alterations, rather than with small physiological fluctuations. The effect of atrial stretch on atrial effective refractory period has been studied by AV synchronous pacing (3, 16), which has additional hemodynamic effects. Klein et al. (16) found a strong correlation between changes in atrial effective refractory period and both right atrial pressure and left atrial diameter. In contrast, Calkins et al. (3) found no such effect with or without prior autonomic blockade. Data from isolated cell preparations suggest that significant changes in cardiac refractory period occur only at supraphysiological levels of stretch (27). The levels of circulating catecholamines may affect atrial electrophysiology. However, catecholamine levels are intrinsically linked to other parts of the autonomic nervous system, and no data are available on electrophysiological effects of other diurnally varying hormones. Hence, it seems plausible to propose that the observed diurnal changes in atrial cycle length are largely mediated by autonomic tone fluctuations. Increased sympathetic and vagal tone decreases the atrial refractory period (20, 30, 38), but the effect of electrophysiological properties on fibrillating human atrial tissue is less clear. Some recent data suggest that the chronic fibrillating atrium is more under sympathetic than vagal control (26, 33), but other studies have shown that adrenergic compounds do not affect Fig. 5. Relationship between ventricular rate (x-axis) and DACL (y-axis) at night (A) and during the day (B). Relationship is stronger at night, with a steeper slope of the regression line. Downloaded from http://ajpheart.physiology.org/ by 10.220.33.3 on May 5, 2017 DACL, dominant atrial cycle length; ACE, angiotensin-converting enzyme; LV, left ventricular; AF, atrial fibrillation; LA, left atrial; N-D, night-day. All differences are nonsignificant (P ⬎ 0.05, U test for discrete state variables, Spearman R ranked correlation coefficient for other variables). the chronically fibrillating atrium (32). Unless increased vagal tone has the opposite effect during AF to that observed in normal conditions, the present study supports the hypothesis that either the sympathetic tone is the dominant determinant of changes in atrial cycle length during AF or dominant AF cycle frequency is less closely determined by atrial effective refractory period than is presently presumed. Preserved vagal tone is known to offer an antiarrhythmic defense in the ventricle. Hence, it might exhibit similar effects in the fibrillating atria, e.g., by slowing and/or partially blocking excitation propagation. In such a scenario, an excitable gap of a larger-than-negligible size may exist within fibrillating atrium under dominant vagal influence. In the present study, the variation in refractoriness appeared to show a single diurnal peak, but more complex patterns of circadian fluctuations in atrial electrophysiology have been suggested to occur. Yamashita et al. (37) found that the probability of paroxysmal AF onset, maintenance, and termination had diurnal distributions that varied from a single peak to DIURNAL VARIABILITY OF ATRIAL FIBRILLATION period are involved. It is believed that concealed conduction of the AF impulses into the AV node and electrotonic modulation of AV nodal propagation participate in the determination of ventricular rate (22). The present study found that the net result is a direct relationship between atrial and ventricular cycle length in physiological conditions. Limitations of the study. The study used 17-h, rather than 24-h, Holter recordings, a limitation imposed by the technology available. Although no statistical differences between different daytime and different nighttime hours could be found (data not shown), we cannot exclude an effect of the underrepresentation of certain daytime segments. Because of the variable quality of ambulatory recordings, we were unable to select segments precisely on the hour in a large proportion of segments. Segments during any activity beyond mild exertion are likely to have been unsuitable for analysis, leading to an underestimation of true diurnal changes. The study also required some operator selection, and despite the objective criteria applied, this may have introduced bias. The FAF-ECG method was validated in controlled laboratory settings on supine subjects, and thus a confounding effect of posture, activity, or other variables is possible. However, the quality of spectra (amount of low-frequency signal, sharpness of peaks) in this study was comparable to that in previous studies. Moreover, there are no signal modulations likely to be encountered during ambulatory recordings that fall within the examined frequency range. It is therefore unlikely that the findings were due to confounding factors, rather than to alterations in AF cycle length. The precise intracardiac site corresponding to the DACL from the lead used in this study has not been tested but is likely to be the right atrial free wall. Assessment of other parts of the atria was therefore not performed. Finally, the DACL can only be used as an assessment of atrial refractoriness as long as there are no significant changes in atrial conduction velocity. The authors thank Burak Acar (Dept. of Electrical and Electronics Engineering, Bilkent University, Ankara, Turkey) for help with computer programming. This study was supported by grants from the Swedish Society of Cardiology, Swedish Heart Lung Foundation, and Medical Faculty of Lund University. J. E. P. Waktare is supported by a British Heart Foundation Project Grant. REFERENCES 1. Akselrod S, Gordon D, Ubel FA, Shannon DC, Berger AC, and Cohen RJ. Power spectrum analysis of heart rate fluctuation: a quantitative probe of beat-to-beat cardiovascular control. Science 213: 220–222, 1981. 2. Brugada R, Tapscott T, Czernuszewicz GZ, Marian AJ, Iglesias A, Mont L, Brugada J, Girona J, Domingo A, Bachinski LL, and Roberts R. Identification of a genetic locus for familial atrial fibrillation. N Engl J Med 336: 905–911, 1997. 3. Calkins H, el Atassi R, Leon A, Kalbfleisch S, Borganelli M, Langberg J, and Morady F. Effect of the atrioventricular relationship on atrial refractoriness in humans. Pacing Clin Electrophysiol 15: 771–778, 1992. 4. Chandler ST and Trewby PN. Is respiratory sinus arrhythmia present in atrial fibrillation? A study using two quantitative methods. Med Eng Phys 16: 334–337, 1994. Downloaded from http://ajpheart.physiology.org/ by 10.220.33.3 on May 5, 2017 trimodal patterns. Their study included many more subjects than the present one (212 vs. 21 patients), and the more complex diurnal patterns were demonstrable only in the younger subset of their patients. Interpatient differences in variability of AF cycle length. The variability of AF cycle length was less expressed in subjects with shorter mean DACL, suggesting that patients with short atrial refractoriness are less receptive to presumed autonomic modulation. High-frequency atrial depolarization leads to a progressive shortening of the refractory period, a phenomenon named electrical remodeling (9, 36). There was no trend for arrhythmia duration to be longer in those with a short AF cycle length in the present study, possibly because maximum remodeling occurs within 1 or 2 mo (8, 36). There was a trend for older subjects to have longer AF cycle length. This is consistent with previous studies showing that atrial refractory period increases with age in sinus rhythm (23). None of the examined clinical parameters predicted mean DACL or night-day differences. However, the present study was not of sufficient size to fully explore the influence of multiple cofactors influencing AF electrophysiology. There was a relationship between mean DACL and night-day difference (Fig. 4) and a possible clustering of the subjects’ results with respect to these parameters. In Fig. 2, there might be a suggestion of subpopulations, rather than a smooth random distribution of mean DACL and night-day difference in DACL. Presently and without further confirmation, it is difficult to speculate whether this reflects distinct electrophysiological mechanisms for the maintenance of AF in different patient groups, e.g., focally mediated AF (14), AF related to delayed interatrial conduction (7), and inherited AF (2). Although “focal AF” is mainly described in paroxysmal AF (14), there are data suggesting that it also mediates persistent AF in some patients (18). Alternatively, some patients may have a genetically determined ability to develop a higher and more severe degree of electrical remodeling. Relationship of AF and ventricular cycle length. This study found a strong correlation between fluctuations in AF cycle length and ventricular rate. This may reflect a similar response of both fibrillating atria and the AV node to diurnal fluctuations of autonomic nervous system tone and other physiological variables or a direct effect of changes in atrial cycle length on ventricular rate. Other observations suggest an inverse relationship between atrial and ventricular cycle lengths during AF (5, 22), and thus it is the net effect of diurnal variations in physiological parameters that results in a direct, rather than an inverse, relationship. Under normal conditions (39) and during AF (21, 26, 29), sympathetic discharge reduces AV nodal refractory period while increased vagal tone increases it. The role of atrial inputs in the ventricular rhythm during AF are complex and incompletely understood. Since depolarizations of the atrial input to the AV node occur at intervals of ⱕ200 ms, yet there is marked beat-tobeat variation in the ventricular cycle length during AF, factors other than AV nodal effective refractory H405 H406 DIURNAL VARIABILITY OF ATRIAL FIBRILLATION 22. Meijler FL, Jalife J, Beaumont J, and Vaidya D. AV nodal function during atrial fibrillation: the role of electrotonic modulation of propagation. J Cardiovasc Electrophysiol 7: 843–861, 1996. 23. Michelucci A, Padeletti L, Fradella GA, Lova RM, Monizzi D, Giomi A, and Fantini F. Aging and atrial electrophysiologic properties in man. Int J Cardiol 5: 75–81, 1984. 24. Millar-Craig MW, Bishop CN, and Raftery EB. Circadian variation of blood pressure. Lancet 1: 795–797, 1978. 25. Moore-Ede MC, Czeisler CA, and Richardson GS. Circadian timekeeping in health and disease. Part 1. Basic properties of circadian pacemakers. N Engl J Med 309: 469–476, 1983. 26. Nagayoshi H, Janota T, Hnatkova K, Camm AJ, and Malik M. Autonomic modulation of ventricular rate in atrial fibrillation. Am J Physiol Heart Circ Physiol 272: H1643–H1649, 1997. 27. Penefsky ZL and Hoffman BF. Effects of stretch on mechanical and electrical properties of cardiac muscle. Am J Physiol 204: 433–438, 1963. 28. Pomeranz B, Macaulay RJ, Caudill MA, Kutz I, Adam D, Gordon D, Kilborn KM, Barger AC, Shannon DC, Cohen RJ, and Benson H. Assessment of autonomic function in humans by heart rate spectral analysis. Am J Physiol Heart Circ Physiol 248: H151–H153, 1985. 29. Raeder EA. Circadian fluctuations in ventricular response to atrial fibrillation. Am J Cardiol 66: 1013–1016, 1990. 30. Rensma PL, Allessie MA, Lammers WJ, Bonke FI, and Schalij MJ. Length of excitation wave and susceptibility to reentrant atrial arrhythmias in normal conscious dogs. Circ Res 62: 395–410, 1988. 31. Schofl C, Becker C, Prank K, von zur Muhlen A, and Brabant G. Twenty-four-hour rhythms of plasma catecholamines and their relation to cardiovascular parameters in healthy young men. Eur J Endocrinol 137: 675–683, 1997. 32. Smeets JL, Allessie MA, Lammers WJ, Bonke FI, and Hollen J. The wavelength of the cardiac impulse and reentrant arrhythmias in isolated rabbit atrium. The role of heart rate, autonomic transmitters, temperature, and potassium. Circ Res 58: 96–108, 1986. 33. Sopher SM, Hnatkova K, Waktare JE, Murgatroyd FD, Camm AJ, and Malik M. Circadian variation in atrial fibrillation in patients with frequent paroxysms. Pacing Clin Electrophysiol 21: 2445–2449, 1998. 34. Tuck ML, Stern N, and Sowers JR. Enhanced 24-hour norepinephrine and renin secretion in young patients with essential hypertension: relation with the circadian pattern of arterial blood pressure. Am J Cardiol 55: 112–115, 1985. 35. Waktare JEP, Gallagher MM, Murtagh A-M, Camm AJ, and Malik M. Optimum lead positioning for recording bipolar atrial electrograms during sinus rhythm and atrial fibrillation. Clin Cardiol 21: 825–830, 1998. 36. Wijffels MCEF, Kirchhof CJHJ, Dorland R, and Allessie MA. Atrial fibrillation begets atrial fibrillation: a study in awake chronically instrumented goats. Circulation 92: 1954–1968, 1995. 37. Yamashita T, Murakawa Y, Sezaki K, Inoue M, Hayami N, Shuzui Y, and Omata M. Circadian variation of paroxysmal atrial fibrillation. Circulation 96: 1537–1541, 1997. 38. Zipes DP, Mihalick MJ, and Robbins GT. Effects of selective vagal and stellate ganglion stimulation on atrial refractoriness. Cardiovasc Res 8: 647–655, 1974. 39. Zipes DP and Miyazaki T. The autonomic nervous system and the heart: basis for understanding interactions and effects on arrhythmia development. In: Cardiac Electrophysiology From Cell to Bedside, edited by Zipes DP and Jalife J. Philadelphia, PA: Saunders, 1990, p. 312–330. Downloaded from http://ajpheart.physiology.org/ by 10.220.33.3 on May 5, 2017 5. Chorro FJ, Kirchhof CJ, Brugada J, and Allessie MA. Ventricular response during irregular atrial pacing and atrial fibrillation. Am J Physiol Heart Circ Physiol 259: H1015–H1021, 1990. 6. Cinca J, Moya A, Figueras J, Roma F, and Rius J. Circadian variations in the electrical properties of the human heart assessed by sequential bedside electrophysiologic testing. Am Heart J 112: 315–321, 1986. 7. Daubert C, Mabo P, Berder V, Le Breton H, Leclercq C, and Gras D. Permanent dual atrium pacing in major interatrial conduction blocks: a four years experience (Abstract). Pacing Clin Electrophysiol 16: 885, 1993. 8. Elvan A, Wylie K, and Zipes DP. Pacing-induced chronic atrial fibrillation impairs sinus node function in dogs. Electrophysiological remodeling. Circulation 94: 2953–2960, 1996. 9. Goette A, Honeycutt C, and Langberg JJ. Electrical remodeling in atrial fibrillation. Time course and mechanisms. Circulation 94: 2968–2974, 1996. 10. Hayano J, Sakakibara Y, Yamada M, Kamiya T, Fujinami T, Yokoyama K, Watanabe Y, and Takata K. Diurnal variations in vagal and sympathetic cardiac control. Am J Physiol Heart Circ Physiol 258: H642–H646, 1990. 11. Holm M, Pehrson S, Ingemansson M, Smideberg B, Sormo L, and Olsson SB. Non-invasive assessment of the atrial cycle length during atrial fibrillation in man: introducing, validating and illustrating a new ECG method. Cardiovasc Res 38: 69–81, 1998. 12. Huikuri HV, Kessler KM, Terracall E, Castellanos A, Linnaluoto MK, and Myerburg RJ. Reproducibility and circadian rhythm of heart rate variability in healthy subjects. Am J Cardiol 65: 391–393, 1990. 13. Huikuri HV, Yli-Mayry S, Linnaluoto MK, and Ikaheimo MJ. Diurnal fluctuations in human ventricular and atrial refractoriness. Pacing Clin Electrophysiol 18: 1362–1368, 1995. 14. Jais P, Haissaguerre M, Shah DC, Chouairi S, Gencel L, Hocini M, and Clementy J. A focal source of atrial fibrillation treated by discrete radiofrequency ablation. Circulation 95: 572– 576, 1997. 15. Kim KB, Rodefeld MD, Schuessler RB, Cox JL, and Boineau JP. Relationship between local atrial fibrillation interval and refractory period in the isolated canine atrium. Circulation 94: 2961–2967, 1996. 16. Klein LS, Miles WM, and Zipes DP. Effect of atrioventricular interval during pacing or reciprocating tachycardia on atrial size, pressure, and refractory period. Contraction-excitation feedback in human atrium. Circulation 82: 60–68, 1990. 17. Lammers WJEP, Alessi MA, Rensma PL, and Schalij MJ. The use of fibrillation cycle length to determine spatial dispersion in electrophysiological properties and to characterise the underlying mechanism of fibrillation. New Trends Arrhythmias 2: 109–112, 1986. 18. Lau CP, Tse HF, and Ayers GM. Defibrillation-guided radiofrequency ablation of atrial fibrillation secondary to an atrial focus. J Am Coll Cardiol 33: 1217–1226, 1999. 19. Levy RD, Cunningham D, Shapiro LM, Wright C, Mockus L, and Fox KM. Diurnal variation in left ventricular function: a study of patients with myocardial ischaemia, syndrome X, and of normal controls. Br Heart J 57: 148–153, 1987. 20. Liu L and Nattel S. Differing sympathetic and vagal effects on atrial fibrillation in dogs: role of refractoriness heterogeneity. Am J Physiol Heart Circ Physiol 273: H805–H816, 1997. 21. Matsuda M, Matsuda Y, Yamagishi T, Takahashi T, Haraguchi M, Tada T, and Kusukawa R. Effects of digoxin, propranolol, and verapamil on exercise in patients with chronic isolated atrial fibrillation. Cardiovasc Res 25: 453–457, 1991.