* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download MDS in STEMI by Mary Antoniette Tan and Ana
Survey
Document related concepts
Transcript
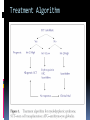
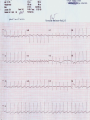
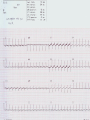
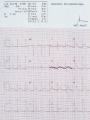
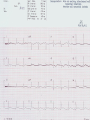
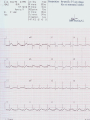
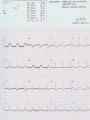
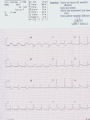
Mortality & morbidity conference Ana Mae Quintal-Quetua, M.D. Mary Antoniette Tan, M.D. Case Objectives To present a case of a patient with Myelodysplastic Syndrome who had multiple medical problems, and developed acute STelevation myocardial infarction To discuss the management and treatment options for a patient with MDS who had STEMI Hematologic- overview of AA and MDS Cardiac Patient Data M.L. 61 years old Female Known HPN, on Diltiazem Known asthmatic Non-diabetic Obese class II Patient Data March 2009, 2nd week - consult at MMC for pancytopenia; BMA done Bone Marrow cytology report (Mar-17-09) 60-70% marrow hypercellularity with erythroid hyperplasia and mild dyserythropoiesis Diagnostic comment: correlation with cytogenetic studies for chromosomal haploinsufficiency is recommended Started on Prednisone; platelet counts improve with increased doses Patient Data March 2009, 4th week- sought 2nd opinion at SLMC; 2nd BMA done Bone Marrow cytology report (Apr-03-09) 5-10% marrow hypocellularity with pancytopenia with relative lymphocytosis; marrow space occupied mainly by fibroadipose tissue Cellular smears with dyserythropoiesis and dysgranulopoiesis Conclusion: Thrombocytopenia secondary to Severe Aplastic Anemia , cannot rule out low-grade Hypoplastic Myelodysplastic Syndrome Patient Data April 5- 9, 2009 - admitted at MMC for ATG administration (with Solumedrol) and was started on Cyclosporine 300mg OD; discharged stable April 2009, 4th week - Prednisone dose was increased up to 100mg OD Steroid-induced hyperglycemia, on Humulin R 80-2 and Humulin N 28-0-10 History of Present Illness 3 days PTA, (+) productive cough, throat irritation and hoarseness but (-) fever, DOB, chest pain, bleeding 12 hours PTA, (+) blood-tinged sputum May 6, 2009 - admitted for blood transfusion due to decreased platelet count (PC 10,000) Physical Examination on Admission General Survey Conscious, coherent, ambulatory, not in respiratory distress Vital Signs BP = 110/70 BMI = 31kg/m2 HEENT Anicteric sclerae, pale palpebral conjunctivae, (+) TPC with whitish plaques on pharyngeal mucosa, (-) CLAD HR = 78 bpm RR = 20 cpm Temp = 37.0C Chest/ Lungs Clear breath sounds, symmetric chest expansion, no rales or wheezes CVS Adynamic precordium, normal rate, regular rhythm, distinct S1 and S2, no murmurs Abdomen Flabby, soft, normoactive bowel sounds, (-) masses or tenderness, no splenomegaly Extremities Full and equal pulses, no pedal edema, no cyanosis, no clubbing Admitting Impression Severe Aplastic Anemia vs Hypoplastic Myelodysplastic Syndrome Upper Respiratory Tract Infection vs Community Acquired Pneumonia Laryngitis, Oral Candidiasis COURSE IN THE WARDS PROBLEM-BASED 1. Pancytopenia BM biopsy (May-29-09, MMC) 10-20% hypocellularity with panhypoplasia, dyserythropoiesis, left- shift granulocytic maturation focal collagen fibrosis, Inc Iron stores CONCLUSION: presence of significant dysplasia favors a diagnosis of Hypocellular Myelodysplatic Syndrome MYELODYSPLASTIC SYNDROME APLASTIC ANEMIA Ineffective Marrow failure secondary hematopoiesis due to abnormal differentiation and maturation, leading to bone marrow failure Peripheral cytopenia, dysfunctional blood elements Possibility of leukemic conversion to inability to produce blood cell components Maybe immune- mediated (T- cells) or due to direct bone marrow injury (drugs, chemicals) Pancytopenia, Bone marrow hypocellularity MYELODYSPLASTIC SYNDROME Bone marrow maybe hypercellular, hypocellular or normal Cytogenetic abnormality involving chromosome 5, 7, 11, 12 and 20 deletions, and/or trisomy 8 Recurrent chromosome deletions Loss of tumor suppressor genes are involved in the pathogenic process APLASTIC ANEMIA Bone marrow consists primarily of fatty space and stromal cells Evolution of AA to MDS/ AML is marked with increased proliferative activity +/- development of cytogenetic abnormalities 9% risk of developing to AML Differentiating AA from Hypoplastic MDS: Diagnosis dictates management and prognosis A hypocellular bone marrow is seen in 20% of MDS , which maybe confused with AA Hypoplastic MDS- cytopenia, marrow dysplasia and marrow hypocellularity Incidence of 7.7% among a large series of MDS patients evaluated Increase in mast cells and lymphoid cells Cytogenetic studies: changes in chromosome 7, especially monosomy 7 3/5 cases transforming from AA to H- MDS showed monosomy 7 Monosomy 7 found in Hypoplastic MDS indicates a poor prognosis, with patients progressing to refractory cytopenia, with 80% risk of evolving into Acute Myeloid Leukemia Trisomy 8- immune pathophysiology; often responds clinically to immunosuppressive Tx International Prognostic Scoring System Treatment Algorithm Treatment MYELODYSPLASTIC Poor response to cytotoxic chemotherapy Stem Cell Transplantation offers cure, 50% survival rate at 3 yrs Demthylating agentsPyrimidine analogues (Azacitidine, Decitabine) Lenalidomide, Cyclosporine, ATG, GCSF APLASTIC ANEMIA Immunosuppression: Combination of Cyclosporine & AntiThymocyte Globulin Stem Cell Transplantation Supportive - transfusion 1. Pancytopenia ISSUES Hemoglobin: 6.3 – 12.4 g/dl WBC: 410 – 5,100 Platelet count: 2,000 – 160,000 Persistent pancytopenia despite multiple blood transfusion and repeated GCSF admin. Risk for severe infection, hemodynamic instability and bleeding 1. Pancytopenia MANAGEMENT Multiple transfusion of blood products ATG Cyclosporine Repeated GCSF administration Steroid administration Erythropoietin, thrombopoietin Vitamin K and tranexamic acid administration Effect of Multiple Blood Transfusion Supportive therapy: PLT transfusion However, repeated PLT transfusion may fail to show the desired increment in PLT counts PLT refractoriness develop in 30-70%; maybe due to the underlying condition (fever, sepsis, drugs) or it may be due to alloimmunization Patients with transfusion failure due to HLAantibodies may be given HLA- matched PLT components 1. Pancytopenia ETIOLOGY Thrombocytopenia secondary to Severe Hypoplastic Myelodysplastic Syndrome Platelet refractoriness secondary to multiple blood transfusion Unresolving Sepsis secondary to Pneumonia in the Immunocompromised 2. Pneumonia and Fungal Laryngitis in the Immunocompromised ISSUES Severe infection; sepsis Unresolving lung infiltrates and persistent pleural effusion Persistent wheezing/bronchospasm sec. to hyperreactive airways 2. Pneumonia and Fungal Laryngitis in the Immunocompromised MANAGEMENT Sputum cultures, blood cultures Chest radiographs, chest UTZ, chest CT scan Attempted thoracentesis/chest pigtail insertion Use of broad-spectrum antibiotics, antifungals, antivirals Inhaled and systemic bronchodilators and mucolytics; steroid administration CT scan of the chest, plain (May 31, 2009) Pneumonia, left lower lobe Soft tissue density in the LUL and pulmonary nodular opacities in the right lung may still be related to the infectious process Subcentimeter granuloma, RLL Minimal pleural effusion, left Calcified lymph nodes, right paratracheal and right hilar regions. These were already noted in the study of April 4, 2009. 3. Respiratory Failure CAUSES Severe infection (laryngeal candidiasis and unesolving pneumonia) and sepsis in the immunocompromised Pulmonary congestion sec.to hypervolemia, hypoalbuminemia Persistent wheezing/bronchospasm sec. to hyperreactive airways Anemia Airway and intrathoracic bleeding Empyema/pyothorax considered 3. Respiratory Failure MANAGEMENT Alternating BIPAP and in-line neb. Endotracheal intubation and subsequent tracheostomy Inhaled and systemic bronchodilators in increased doses Inhaled and systemic mucolytics 4. Hypoalbuminemia CAUSES Nutritional Hepatic failure ISSUES Third spacing: congestion, effusion and edema MANAGEMENT Repeated IV infusions of 25% human albumin 5. Hyperglycemia CAUSES Steroid-induced Stress-induced ISSUES Risk for uncontrolled infection, cardiovascular/coronary events MANAGEMENT CBG monitoring and insulin administration 6. Renal Failure CAUSES Sepsis Decreased renal perfusion sec. to anemia (shunting of blood away from the kidney for protection of O2 delivery to vital organs) Drugs (Cyclosporine, Amphotericin, antibiotics) 6. Renal Failure ISSUES Hypervolemia (increased BP and CVP 1419 cmH2O, edema) due to multiple transfusions, steroid admin. On the 22nd HD, (+) oliguria with rising serum crea 2.0 from 0.9 mg/dl (est. crea clearance 21 ml/min from 47 ml/min) 6. Renal Failure MANAGEMENT Dose adjustments of antibiotics and other medications 12-hour urine collection Diuresis (Furosemide, Spironolactone, Bumetanide) Subsequent hemodialysis 7. Abdominal Distension CAUSES Septic ileus Ileus sec.to metabolic derangement Pseudomembranous colitis considered ISSUES Increasing abdominal girth with intermittent episodes of voluminous watery stools 7. Abdominal Distension MANAGEMENT NPO Plain film of the abdomen CT scan of the abdomen Prokinetics, laxatives, enemas Vancomycin per orem Rectal tube insertion Total parenteral nutrition CT scan of the abdomen, plain (May 31, 2009) Ileus Few descending colon diverticula Distended gall bladder Atrophic pancreas Possible right renal cortical cyst Subcentimeter appendicolith vs. inspissated barium with no associated inflammatory changes The rest is normal 8. Metabolic Encephalopathy CAUSES Sepsis Electrolyte derangements Hypovolemia episodes during hemodialysis Hepatic failure 8. Metabolic Encephalopathy MANAGEMENT Referral to Neurology service on the 27th HD Cranial MRI and MRA of intracranial vessels requested EEG showed encephalopathy probably sec. to hypovolemia (hypotension episodes during dialysis) Correction of electrolyte abnormalities; manage sepsis 9. Elevated Blood Pressure (130-200/70-100 mmHg) CAUSES Volume overload sec. to multiple blood transfusion, steroid use Increased cardiac output sec. to tachycardia Catecholamine release (stress-induced) 9. Elevated Blood Pressure (130-200/70-100 mmHg) MANAGEMENT Ca-channel blockers (Amlodipine, Nicardipine, Verapamil) Clonidine Diuresis 10. Tachycardia with PACs and PVCs (100 – 180 bpm) CAUSES Sepsis/stress-induced Anemia Metabolic derangements (i.e. electrolyte imbalance) Bronchodilators in increased doses 10. Tachycardia with PACs and PVCs (100 – 180 bpm) MANAGEMENT 2D-echocardiogram with CFDS Electrolyte correction Beta-blockers (Metoprolol, Nebivolol) Digoxin stat doses Verapamil Amiodarone Ivabradine Trimetazidine 2D-echocardiogram with CFDS (baseline) May 15, 2009 - Normal LV dimension with NWMC, EF 75%; calcified non- coronary cusp of aortic valve without restriction of motion. Calcified aortic walls; normal MV, TV, PV; minimal pericardial effusion; color flow Doppler= MR, mild; TR, mild; mild pulmonary hypertension as evidenced by pulmonary acceleration time of 100 msec; Doppler evidence of impaired LV relaxation. On the 35th hospital day… Tracheostomy done O2 desaturation noted ~ 90% HR much controlled: 78 bpm BP 104/55 mmHg 12-lead ECG post-trache showed: new Twave inversions at V1 – V3, ST-elevations at II and AVF Cardiac Enzymes May 13: TCPK 60, CKMB 1.5, Trop I 0.08 June 10: TCPK 47, CKMB 7, Trop T 0.51 June 11: TCPK 29, CKMB 2.9, Trop I 0.37 May 13: serum BNP 4,071 2D-echocardiogram with CFDS (repeat) June 11, 2009 - concentric LVH with NWMC, EF 55%; color flow Doppler = MR, mild; Doppler evidence of LV diastolic dysfunction (E/A velocity ratio and isovolumic relaxation time); mild pulmonary hypertension by pulmonary acceleration time 108 msec; compared with previous study done may 15, increase in LV wall thickness; no note of pericardial effusion. On the 41st hospital day… While on hemodialysis, ST-segment elevations were noted on cardiac monitor, and patient had 3 episodes of HR 30s – 40s (5 – 10 secs) BP = 100-130/60-80 mmHg Awake, responsive (-) chest pain 12-lead ECG showed…. 11. STEMI ISSUES Risk factors: age, atherosclerosis, anemia, obesity, HPN, hyperglycemia, dyslipidemia, hemodialysis, multiple blood transfusion (blood viscosity? increased platelet aggregation? endothelial damage?), heparin-induced thrombocytopenia (?) 11. STEMI MANAGEMENT Nitroglycerin drip Nicorandil 5mg/tab 1 tablet BID Coronary angiogram Options: anti-platelet and anti-coagulation (?), PTCA (?) Coronary Angiogram Results: Left ventriculogram – intentionally omitted due to elevated LV EDP 31mmHg Coro angio – L main coronary artery normally patent; LAD normal; L circumflex has slow flow with abrupt cut off distally which indicates an endovascular thrombus; RCA is dominant and normal. Assessment: Coronary thrombus involving L circumflex artery Recommedation: Consider Cilostazol at low dose 50mg BID How Was The Patient Managed? Post- coronary angiography recommendations: Interventional Cardiology - low-dose Cilostazol 50 mg BID Cardiology – Enoxaparin 40mg SC OD Hematology service, however, did not agree Due to persistent pancytopenia, Interventional Cardiology entertained the possibility of HIT, and suggested discontinuation of Heparin and use of Bilvalirudin and Aspirin 325 mg OD Heparin- Induced Thrombocytopenia? HIT is a pro-thrombotic, immune- mediated adverse reaction to heparin therapy; It should be suspected whenever the PLT falls at least 50% and/ or thrombosis occurs between day 4 and 14 following initiation of heparin Based on results reported by Warkentin et al. and a meta- analysis of 15 studies, it appears that HIT type II occurs in 1-3% of all patients receiving UFH & in only 0.3- 0.8% receiving LMWH Heparin-InducedThrombocytopenia? When heparin is administered in the presence of platelet factor 4 (PF4), an immune complex can form and result in the production of anti-PF4/ Heparin antibody (HIT antibody) This complex initially causes PLT aggregation, followed by a decrease in the PLT count (due to consumption and elimination from the circulation) & the development of thrombosis. Paradoxical thrombosis in the presence of thrombocytopenia is a hallmark of HIT Heparin- Induced Thrombocytopenia? Diagnostic tests include C-Serotonin Release Assay, Platelet aggregation test, Heparin- induced platelet aggregation test, ELISA Treatment is prompt discontinuation of heparin and use of an anti-thrombin agent (ie, Argatroban, Bivalirudin) to prevent thrombotic complications Associated thrombotic events frequently noted are Deep Vein Thrombosis, Pulmonary Thromboembolism, Mesenteric Arterial Thrombosis Bivalirudin Newer direct thrombin inhibitor; binds to the substrate- binding site of thrombin, with high affinity Has a rapid, dose- dependent anticoagulation, with half- life 25 minutes Eliminated by the kidneys As with all direct thrombin inhibitors, it has no structural similarities to heparin and has no crossreactivity with HIT antibodies Patient Outcome Hematology service did not agree on the diagnosis of HIT; however agreed on the use of Aspirin 325 mg OD (only 1 dose was given) 3rd day post MI and post angiogram, persistent pancytopenia and hemodynamic instability Ventricular fibrillation After 4o minutes of CPR, patient expired IS IT POSSIBLE TO DO PERCUTANEOUS CORONARY INTERVENTIONS IN PATIENTS WITH MYELODYSPLASTIC SYNDROME? Myelodysplastic Syndrome & Myocardial Infarction The association of CAD and thrombocytopenia due to MDS is rare, and published data on the management of obstructive lesions through PCI is limited This presents a challenge to interventional cardiology, because PCI requires preventing both intracoronary thrombosis with antiplatelet agents, and bleeding after the procedure 61/ M, known case of MDS, hypertensive, presented with pressure- like chest pain, Dx: Unstable Angina; submitted to coronary angiography and stenting Platelet count was 40,000; prophylactic, 10 unit platelet transfusion 2 days prior to coronary angiography 80% stenosis of the RCA and 30% in the LAD After a 6- day Clopidogrel 75 mg OD regimen, and 2 hrs after another 10 unit PLT transfusion, stenting DES of the RCA lesion was done 6F catheter, femoral approach; introducer was removed 4 hrs after the procedure 100 IU/ kg Heparin during the procedure Local compression for 20 minutes; no bleeding nor hematoma was observed after the removal of introducers Discharged with Clopidogrel 75 mg OD to complete 30 days of continuous use. Platelet count remained stable at 40,000 on Clopidogrel 6 months later, TMST showed ST depression in the inferior leads; repeat coronary angiography showed a 70% in-stent stenosis; He refused another PCI 20 months after stenting, patient remained asymptomatic, maintained on the same medication (Clopidogrel 75 OD, propranolol 40 mg BID, Simvastatin 20 mg OD) His last platelet count was 104,000 In spite of the low PLT, this patient evolved with progressively unstable angina, suggesting that regardless of the small number, platelet function remained normal, thereby justifying the use of anti- platelet agents Clopidogrel alone was the treatment of choice in this study, since it is better tolerated than Ticlodipine This case report suggest that PCI in patients with thrombocytopenia is a safe procedure, provided that prophylactic platelet transfusion is done, although restenosis may occur Management Dilemmas Is it possible to do stent placement? •Bare- Metal Stent vs Drug- Eluting Stent? If patient tolerates stent implantation, is it possible to maintain her on an anti- thrombotic regimen? •Single or Dual Anti- PLT therapy? •ASA vs Clopidogrel vs Cilostazol? • Dose and duration? STENTS Stents are used in the management of ACS and its use has improved the outcomes of percutaneous coronary revascularization The metallic surface of the bare-metal stent (BMS) can stimulate thrombosis before it becomes entirely covered with endothelial tissues Disadvantage: subsequent development of an instent restenosis due to significant loss of lumen STENTS Drug – eluting stents (DES) are believed to inhibit restenosis by inhibition of cytokine- mediated and GF- mediated proliferation of lymphocytes and smooth muscle cells DES indicated for high- risk patients (diabetics, long lesions, small vessels, presence of a thrombus) DES and dual anti- PLT therapy with ASA and Clopidogrel reduce stent re-thrombosis rates to <2% However the consequences for patients with potential bleeding conditions are potentially life- threatening Drug- Eluting stents vs Bare metal stents: Off- label indications, A Propensity- score Matched Outcome Study Stent Thrombosis Catastrophic, mortality rate of 20-45% Most commonly, about 1% occurs in the first month after stent implantation ( subacute ) and about 0.19% occur after a few months to a few years ( late ) BMS are associated with higher incidence of stent thrombosis compared to DES Timing of thrombosis appears to be delayed in DES (higher early stent thrombosis with BMS, and late thrombosis with DES) Drug- Eluting stents vs Bare metal stents: Off- label indications, A Propensity- score Matched Outcome Study BMS or DES ? DES is indicated in high- risk patients: diabetic, small vessel, long lesion, presence of a thrombus (off- label) BMS - Shorter duration of anti- platelet therapy (minimum of 2 weeks to 1 month for those at risk of bleeding) thereby minimizing the risk of bleeding • However, BMS are prone to early stent thrombosis Aggressive: PCI BMS has a shorter duration of anti- platelet therapy (minimum of 2 weeks to 1 month for those at risk of bleeding) for post- BMS stent, Clopidogrel 75 mg OD for at least 1 month (ideally up to 1 year if not at risk for bleeding; then it should be given for a minimum of 2 weeks if at risk) a lower dose of 75- 162 mg of ASA is reasonable (during the initial period after stent placement) in patients at risk of bleeding Cilostazol has similar anti- PLT affects with less serious side effects; Use of ANTI- PLATELET AGENTS in Hematologic Patients Literature reports on ITP patients who underwent PTCA/ PCI, with use of anti-PLT: 62/M with PLT 3,000, underwent LAD stenting, tolerated 4wks of ASA and 2wks of Clopidogrel; no bleeding (Caputo, et al.) 68/F with PLT 22,000, underwent LAD stenting, maintained on Ticlodipine, no bleeding (Kikuchi et al.) 54/M with PLT 8,000, underwent BMS stenting, given Enoxaparin; discontinued due to epistaxis (Marques et al.) 77/M with PLT 70,000, underwent PTCA, on ASA and Clopidogrel; discontinued due to petechiae Use of Anti- Platelet Agents ASPIRIN Irreversible inhibition of COX-1, which prevents formation of PGH2, the precursor of thromboxane A2 Patients with thrombocytopenia from whatever cause (PLT 50,000) are at extremely high- risk of excessive bleeding. Therefore, Aspirin is harmful in these patients and should not be administered (Class III RECOMMENDATION) In those who underwent stent placement, a lower dose of 75- 162 mg of ASA is reasonable (during the initial period after stent placement) in patients at risk of bleeding The Society of Thoracic Surgeons Practice Guideline Series : ASA and other Anti- Platelet agents during operative coronary revascularization, 2003 2007 Focused Update on STEMI Guideline CLOPIDOGREL Inhibits binding of PLT to ADP receptors 2007 STEMI guideline • For STEMI patients not undergoing PCI (medical or PTCA without stenting), treatment with Clopidogrel 75mg OD x 14 days • Recommends all post- PCI stent patients receiving a DES, Clopidogrel 75 mg OD for at least 12 months if patient is not at high risk of bleeding; for post- BMS stent, Clopidogrel 75 mg OD for at least 1 month (ideally up to 1 year if not at risk for bleeding; then it should be given for a minimum of 2 weeks if at risk)– Level of evidence, B • Premature discontinuation of Clopidogrel is associated with a marked increase in risk of stent thrombosis, & is the leading independent predictor in multiple studies • For every 100 patients treated with Clopidogrel + ASA, 2 events of CV death, non- fatal MI, or stroke will be prevented but 1 patient will suffer a major bleed (ie, ICH) 1 Prevention of Premature Discontinuation of Dual anti- Platelet Therapy 2 AHA 2007 Focused Guideline on STEMI 3 CURE (Clopidogrel in Unstable angina to prevent Recurrent Event )Trial Predictors of Thrombosis CLINICAL Advanced Age Acute coronary Syndromes Diabetes Renal Failure Low ejection Fraction Premature Discontinuation of Dual anti- PLT Therapy in Patients with Stents (Journal of AHA, 2007) CILOSTAZOLPhosphodiesterase III inhibitor; anti- proliferative effect Cilostazol for RESTenosis (CREST) clinical trial in post BMS Preliminary results suggested that Cilostazol (phosphodiesterase III inhibitor) reduces restenosis by nearly 40% over standard therapy alone It has similar anti- PLT affects with less serious side effects (esp bleeding) ; common SE include thrombocytopenia and leukopenia/ agranulocytosis CILOSTAZOL In the study “Triple vs Dual Anti- platelet Therapy After Coronary Stenting”, addition of Cilostazol resulted in further reduction of PLT activation due to additional suppression of P- selectin (a marker of PLT activity) The rate of major bleeding was similar in both groups However, patients with known bleeding disorders and PLT< 150T were excluded in the study Recommended dose 100 mg BID, but may be decreased to 50 BID if with concomittant use of CYP450 inhibitors (ie, Diltiazem) SINGLE ANTIPLATELET THERAPY OR DUAL ANTI- PLATELET THERAPY Current ACC/ AHA/ Society of Cardiovascular Angiography & Interventions for prevention of stent thrombosis post PCI _________________________________________ At minimum, patients should be treated with : Clopidogrel 75 mg OD & ASA 325 mg OD for 1 month after BMS, 3-6 months after DES, and ideally up to 1 year if they are not at risk for bleeding Dual Anti- PLT Therapy for Prevention of Ischemic Cardiovascular Events & Stent Thrombosis Use of a single antiplatelet regimen may possibly reduce the risk of bleeding •Aspirin - 75- 162 mg is reasonable (during the initial period after stent placement) in patients at risk of bleeding •Cilostazol has similar anti- PLT affects with less serious side effects; Outcome Hematology service agreed on Aspirin 325 mg OD 3rd day post angiogram, persistent pancytopenia and hemodynamic instability ventricular fibrillation After 45 minutes of CPR, patient expired Final Diagnoses Cardiopulmonary Arrest sec.to Ventricular Fibrillation post STEMI, anterolateral wall Myelodysplastic Syndrome Sepsis sec.to Pneumonia and Laryngeal Candidiasis in the Immunocompromised Acute Renal Failure, multifactorial Steroid-induced Hyperglycemia Hypertensive Atherosclerotic Cardiovascular Disease Arrhythmia is a serious complication of STEMI Possibility of another infarction may have precipitated the arrhythmia In retrospect, in an immunocompromised patient with severe hematologic disorder with a coronary artery thrombus, who was septic, undergoing dialysis for acute renal failure, with impending liver failure, would an aggressive approach like PCI do more harm than good? CONCLUSION It is apparent that any medical or surgical intervention has both systematic and random effects, some of which are beneficial and some are unintended negative consequences Therefore , the practice of medicine often necessitates a probabilistic balancing of these conflicting effects, and management should be individualized, depending on the risk: benefit ratio Thank You “….. The science of medicine becomes a structured and organized way of using probability, uncertainty, and facts in preventive medicine and clinical care to best benefit the patient and the community…” -Jenicek, Foundations of Evidence- Based Medicine