* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter 5 Notes
Survey
Document related concepts
Transcript
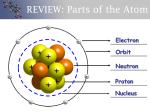
Chapter 5 Notes The periodic table has not always looked as it does today. The first periodic table was arranged by Dmitri Mendelev according to atomic mass. He noticed that physical and chemical appeared to be in a repeating or PERIODIC pattern. He continued to arrange his table in groups according to similar properties, but found that he had several empty spaces between the elements. He proposed that undiscovered elements would fill the empty spaces. Working with Ernest Rutherford, Henry Mosley rearranged Mendelev’s periodic table according to the number of protons (atomic number). This arrangement was consistent with Mendelev’s groupings and lead to the modern PERIODIC LAW, the physical and chemical properties of elements are a function of the atomic number. The PERIODIC TABLE can be defined as the arrangement of elements in order of atomic number so that elements with similar properties are grouped in columns. The periodicity of the periodic table is as follows. Group 1 elements Li Na K Rb Cs Fr atomic number 3 11 19 37 55 87 difference between periods 11-3=8 19-11=8 37-19=18 55-37=18 87-55=32 Group 18 elements He Ne Ar Kr Xe Rn atomic number 2 10 18 36 54 86 difference between periods 10-2=8 18-10=8 36-18=18 54-36=18 86-54=32 Notice how the difference in atomic numbers following the same repeating pattern between groups 1 and 18. The periodicity on the periodic table also explains how electrons fill orbitals. The orbitals are filled in the following manner. Period 1 2 3 4 5 6 7 # of elements 2 8 8 18 18 32 32 order of fill 1s 2s, 2p 3s, 3p 4s, 3d, 4p 5s, 4d, 5p 6s, 4f, 5d, 6p 7s, 5f, 6d, 7p The groups on the periodic table can be broken down into blocks to help with filling electron orbitals. S- Block The s-block elements consist of the elements in groups 1 and 2 of the periodic table. Electrons from these elements fill the s orbital of each period. Group 1 (ALKALI METALS) fill the s orbital with 1 electron. These elements are considered to be reactive metals because they are not readily found in pure form in nature and react violently with water. They have a silvery appearance and are soft. Group 2 (ALKLINE EARTH METALS) fill the s orbital with the second electron. The s-orbital is filled with group 2 elements. These elements are harder and more dense than the alkali metals. They are also less reactive, but their reactivity is enough that they are not readily found in nature in pure form. HYDROGEN and HELIUM are part of the s-block elements, but their properties differ from other s-block elements. P-block The p-block elements consist of the elements in groups 13-18. Electrons from these elements fill the 3 orbitals of the p shell. There are 6 elements providing the 6 electrons needed to fill the orbitals. Metals, metalloids, and non-metals make up the p-block elements. Group 17 contain the HALOGENS, the most reactive non-metals. Halogens will readily react with metals to form salts. Group 18 contain the NOBLE GASES. Noble gases are unreactive because electrons from the gases fill the p orbitals. The s-block and p-block elements make up the MAIN GROUP elements. D-block The metals in groups 3-12 make up the d-block. These metals are called TRANSITION METALS. These metals have the properties associated with typical metals. They are not especially reactive so they can be found in nature in pure form. They are good conductors of energy. Many of these metals do not follow the general rules for electron configuration. F-block The elements of the f-block can be found between the groups 3 and 4 in periods 6 and 7. These elements are found in the insert at the bottom of the periodic table. The elements in period 6 are called the LANTHANIDES. The lanthanides are similar to the alkaline earth metals. The elements in period 7 are called the ACTINIDES. The actinides with the exception of the first 4 elements are synthetic. All actinides are radioactive. Many chemical properties will trend on the periodic table. These properties are atomic radii, ionization energies, and electronegativity. ATOMIC RADII is the distance between the nuclei of identical atoms that are bonded together. Across a period the atomic radii of elements tend to decrease from group 1 to group 18. This decrease in atomic radii is due to the increasing positive charge created by the addition of protons as you move across the period. The more protons the greater the positive charge pulling the negatively charged electrons closer to the nucleus. The atomic radii do not follow the same trend down a group. The atomic radii of elements moving down a group will increase in size. The increase in size is due to the addition of electron orbitals as the energy levels are added. Electrons can be added or removed to atoms creating charged atoms called IONS. Ions can carry either a positive or negative charge. The charge is created by the balance between protons and electrons. An atom with more protons than electrons will carry a positive charge. An atom with more electrons than protons will carry a negative charge. IONIZATION is the process in which electrons are removed from an atom. The energy required to remove the electron is called IONIZATION ENERGY. The greater the ionization energy the more difficult it is to remove an electron from the atom. Ionization energy increases across a period making it more difficult to remove electrons from non-metals. Electrons are more easily removed from metals in groups 1 and 2. The ionization energy will decrease down a group of main group elements due the electrons being located a further distance from the nucleus. The removal of an electron creates the situation where the protons exist in greater numbers than electrons. The charge will be positive. Ions with a positive charge are called CATIONS. ELECTRONEGATIVITY is a measure of the ability of an atom to attract electrons. The energy associated with gaining electrons is called ELECTRON AFFINITY. The greater the electronegativity the more likely an atom is to accept an electron. Electronegativity increases across a period due to the filling of the electron orbitals. Elements in group 17, halogens, are consider to be the most reactive of all non-metals because they have 7 of the 8 electrons needed to fill the s and p orbitals associated with the period. The acceptance of electrons by an atom creates a situation where the number of electrons is greater than the number of protons. These atoms will carry a negative charge and are called ANIONS. The electrons that are lost, gained, or shared are called VALENCE ELECTRONS. These electrons are involved in the formation of ions. The maximum number of valence electrons is 8 regardless to where the element is on the periodic table. You can find the number of valence electrons by counting the number of electrons in the s and p orbitals of the highest energy level.