* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download 5TETRACYCLINES
Survey
Document related concepts
Transcript
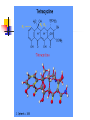
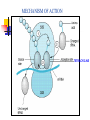
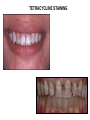
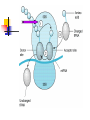
TETRACYCLINES HISTORY Obtained from soil actinomyces. Introduced in 1948- chlortetracycline under the trade name of ‘aureomycin’ from S.aureofaciens Followed by oxytetracycline in 1950 Tetracycline in 1952 CHEMISTRY The basic tetracycline structure consists of four benzene rings with various constituents on each ring. The crystalline bases are faintly yellow, odorless, slightly bitter compounds. They are only slightly soluble in water at pH 7 but they can form soluble sodium salts and hydrochloride. R7 R6 R5 CLASSIFICATION GROUP-I SHORT ACTING 6-8 hr TETRACYCLINE OXYTETRACYCLINE CHOLRTETRACYCLINE GROUP-II INTERMEDIATE ACTING 12 hr DEMECLOCYCLINE METHACYCLINE LYMECYCLINE GROUP- III LONG ACTING 16-18 hr DOXYCYCLINE MINOCYCLINE MECHANISM OF ACTION Primarily bacterostatic Inhibit protein synthesis by binding to 30S ribosome in susceptible organisms It interferes with the attachment of aminoacyl t-RNA to the m- RNA ribosome As a result peptide chain fails to grow MECHANISM OF ACTION TETRACYCLINE ANTIMICROBIAL SPECTRUM All gram +ve and negative cocci except strept.pyogenes ,staphylococci aureus Gram +ve bacilli such as clostridia,corynebacterium but not mycobacterium Sensitive gram –ve bacilli ,H.ducreyi,V.cholerae, Other organisms spirochetes, ricketssiae . E.histolytica actinomyces, mycoplasma RESISTANCE Tetracycline concentrating becomes less efficient Elaboration of tetracycline inactivating enzymes Plasmid mediated synthesis of a “protection proteins” which protects the ribosomal binding site from tetracycline Complete cross resistance is seen among tetracycline groups. Partial cross resistance with chloramphenicol PHARMACOKINETICS incompletely absorbed from GIT Absorbs faster taken on empty stomach Doxycycline and minocycline not interfered by food chelating property -not taken with milk, iron , antacids Higher concentration in liver ,spleen :bind to connective tissue and of bone and teeth CSF – ¼ the plasma conc whether meninges inflammed or not Metabolised in liver, undergoes enterohepatic circulation Excretion mainly by glomerular filteration. Dose has to be decreased in renal failure except doxycycline Administration Tetracyclines to be taken ½ hr before or 2 hr after food Not by IM Slow IV in severe cases Topical preparations – high risk of sensitization But can be given for topical application ADVERSE EFFECTS Irritative effects on GI mucosa, esophageal ulceration due to release of material from capsules Dose related toxicity - liver damage –fatty infiltration of liver, precipitates acute hepatic necrosis in pregnant women - kidney damage - Reversible fancony syndrome-caused by degraded by products – epitetracycline, anhydrotetracycline, epianhydrotetracycline ADVERSE EFFECTS Photo toxicity-sun burn /severe skin reaction , with demeclocycline, doxycycline Teeth and bones- brown discoloration of teeth, susceptible to caries TETRACYCLINE STAINING ADVERSE EFFECTS • • • • Antianabolic effect Increased intracranial pressure in infants Diabetes insipidus –demeclocycline Vestibular toxicity- minocycline : ataxia, vertigo, nystagmus • Hypersensitivity • Superinfection – less with doxycycline and minocycline PRECAUTION Pregnancy, lactation and in children below 8 years patients on diuretics cautiously used in renal /hepatic diseases Do not mix it with penicillin – inactivation occurs CLINICAL USES Drug of choice in Venereal diseases – LGV, G.inguinale Atypical pneumonia- mycoplasma Cholera Brucellosis Rickettsial infection Acne Plague H.pylori CLINICAL USES Amoebiasis- E. histolytica Malaria-adjuvant to quinine/sulfodoxinepyrimethamine for chloroquine resistant malaria Bronchitis Lymes disease Leptospirosis Community acquired pneumonia Meningococcal carrier- minocycline 200mg/d SIADH-demeclocycline CHLORAMPHENICOL Source: Streptomyces venezulae Bacteriostatic Spectrum: aerobic and anaerobic G+ve and G –ve organisms (S.typhi), ricketsiae, H. influenza, N. meningitidis Mechanism of action Binds to 50 s ribosome Inhibits peptidyl transferase Prevents attachment of peptide chain from P site to A site Prevents formation of peptide bond Resistance Production of chloramphenicol acetyltransferase ( plasmid encoded) Decreased permiability Low affinity of ribosome for chloramphenicol Cross resistance with tetracyclines Kinetics Oral- well absorbed Wide distribution to various tissues , body fluids, CNS Conjugated with glucuronic acid Excretion in urine, feces, bile Uses Enteric fever- 500mg 6 hrly Rickettsial infection – rocky mountain spotted fever, typhus fever Meningococcal meningitis Anaerobic infections in wounds, abcesses Topical- eye, ear- endopthalmitis, conjunctivitis Alternative to tetracyclines for brucellosis, cholera, rickettsia Alternative to erythromycin for whooping cough Adverse effects Gastrointestinal: Nausea, vomiting, diarrhoea Aplastic anemia Bone Marrow - Non dose related (idiosyncratic) Dose and duration of therapy related (Due to mammalian mitochondrial protien synthesis Inhibition ) Gray baby syndrome: vomiting, flaccidity, hypothermia, gray color, shock, collapse Due to blockade of electron transport in liver , heart, stomach Superinfection