* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Referral Criteria for consideration of treatment with exanatide (Byetta
Survey
Document related concepts
Transcript
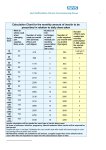
Assessment and treatment pathway for GLP-1 mimetics Inclusion criteria For the treatment of adults with Type 2 Diabetes Mellitus (T2DM) as follows: Triple therapy regimes In combination with metformin and either a sulphonylurea (SU) or a thiazolidinedione when these treatments alone have failed to provide adequate glycaemic control defined as HbA1c < 7.5% (<58 mmol/mol) or other higher level agreed with the individual, AND BMI ≥ 35 kg/m2 in those of European descent (adjusted to BMI 32 kg/m2 for other ethnic groups) AND specific health/psychological problems associated with obesity, OR BMI < 35 kg/m2 (BMI 32 kg/m2 in ethnic groups) but insulin treatment is unacceptable because of occupational implications, or weight loss would benefit other obesity-related comorbidities. Dual therapy regimes This is only an option for people unable to take multiple oral therapy options and whose only current alternative treatment is the early initiation of insulin. GLP-1s can be combined with metformin or an SU only if The person is intolerant of either metformin or a sulphonylurea, or treatment with metformin or a sulphonylurea is contraindicated, AND The person is intolerant of thiazolidinediones (TZD) AND DPP-4 inhibitors (‘gliptins’), OR treatment with thiazolidinediones AND DPP-4 inhibitors is contraindicated. Exclusion criteria Type 1 diabetes or history of diabetic ketoacidosis Children & adolescents below 18 years of age Patients with severe osmotic symptoms and weight loss secondary to hyperglycaemia Refer to SPC & BNF for specific details on use in heart failure or hepatic & renal impairment Clinic schedule GLP-1s can be initiated by prescribers with expertise in diabetes in primary, intermediary or secondary care. First clinic visit Prescriber to assess & prescribe GLP-1 and supply needles & yellow bin (sharps container) Prescriber to give education & info pack + book next review HbA1c & weight to be recorded Month 1 Record weight Give script for 2 months supply and book appointment for 2 months Diabetes clinic only - if treatment is to be continued: Write to GP to request they take over prescribing Ensure patient has sufficient supply until GP has agreed Month 3 Record HbA1c & weight Arrange follow-up appointment in further 3 months. Previous history of pancreatitis Severe gastrointestinal disease Inflammatory bowel disease Diabetic gastroparesis Pregnancy or breastfeeding Older patients (75+) - therapeutic experience limited Metformin and/or TZD dose GLP-1 dose All patients All patients Group 1 (Car/ Motorcycle) Group 2 (LGV/PCV drivers) Dose as per BNF Half dose Consider stopping Telephone review after 1-2 weeks & titrate dose up as appropriate Consider maintaining at half dose If necessary, reintroduce at low dose Continue as before Sulphonylurea dose REVIEW REVIEW Advice to patients Administer via subcutaneous injection only & importance of rotating injection sites What to do if a dose is missed: administer as soon as practical and then continue with the next scheduled dose (except twice daily exenatide - proceed to next dose). Do not exceed prescribed dose in 24hr period Educate on dose timing, test doses, storage and how to use kit as per manufacturer’s instructions Prolonged release version: administer immediately after suspension of powder into solvent Safe sharps disposal (does the patient have a yellow bin for sharps?) Nurse specialist/prescriber to observe patient giving 1st injection and to wait 30 minutes post injection Advise patient regarding nausea as per manufacturer’s instructions and instruct patient to seek prompt medical attention if they develop any symptoms suggestive of pancreatitis Developed by Medicines & Prescribing Team, NHS Solihull CCG & Solihull Community Diabetes Service, Heart of England NHS Trust, May 2015 Approved for use in the NHS by Medicines & Prescribing Subcommittee, NHS Solihull CCG, July 2015. Not for commercial use. Assessment and treatment pathway for GLP-1 mimetics Check driver status and advise patient regarding the DVLA guidance as appropriate. If the patient is to continue taking a sulphonylurea: LGV PCV Drivers - advise to inform DVLA if continuing sulphonylurea Give additional advice related to risks specific to job where appropriate Re-visit hypoglycaemia education – will need to test blood glucose levels Prescriber checklist Concomitant Drug Recommended action More frequent monitoring will be required until GLP-1 dose is stabilised. Check INR within 7 days of GLP-1 initiation and after any dose adjustment Metformin &/or Thiazolidinedione (TZD) The current dose of metformin and/or TZD can be continued Reduce the dose of the sulphonylurea to prevent hypoglycaemia. Sulphonylurea The prescriber should write specific instructions in the medical notes to this effect. If no specific details are given, advise the patient to reduce the dose as in the clinic schedule above DPP-4 inhibitor, repaglinide or SGLT2 Advise patient to stop these medications when GLP-1 is commenced Insulin Only lixisenatide should be prescribed in combination with insulin (only NPH insulin). Refer to hospital diabetes clinic for advice – see local RiCAD NOTE this is a summary only: please refer to SPC data sheet &/or BNF for full prescribing information Warfarin Home blood glucose monitoring Therapy GLP-1 + sulphonylurea (+/- metformin or TZD) GLP-1 + metformin or TZD Recommendations Test once daily, at different times of day, for first 2 weeks. Adjust dose of SU as necessary Increase frequency of monitoring if unwell or symptoms. Once on stable dose, monitoring not required. Monitoring not required Ongoing review Prescriber to review after 6 months from start of therapy & assess response to GLP-1 therapy Community Diabetes Service: If treatment is to be continued, write to GP with on-going management plan and review requirements. Discharge from the service. Dual therapy including GLP-1 Continuation criteria after 6 months Triple therapy including GLP-1 1% (11 mmol/mol) decrease in HbA1c 1% (11 mmol/mol) decrease in HbA1c AND AND tolerability 3% reduction in weight (STOP if unmet) LGV/PCV drivers taking GLP-1 with Metformin or TZD +/- sulphonulyurea 1% (11 mmol/mol) decrease in HbA1c OR maintenance of HbA1c (in exceptional cases only) AND 3% reduction in weight (if GLP-1 prescribed as part of triple therapy regime) AND tolerability AND tolerability Discontinuation criteria Failure to achieve beneficial metabolic response after 6 months (as defined in the criteria above) Treatment not tolerated Compelling indication for insulin ONLY therapy (weight loss with worsening symptoms/increased HbA1c) Symptoms suggestive of pancreatitis such as unexplained persistent abdominal pain with or without vomiting. If pancreatitis is confirmed, do not restart GLP-1 unless an alternative aetiology for the pancreatitis is identified. If a different anti-diabetic treatment is started after the discontinuation of a prolonged release GLP-1, consideration should be given to the prolonged effect of GLP-1 therapy. Caution: Rapid weight loss at a rate of 1.5 kg per week has been reported in patients treated with exenatide. Weight loss of this rate may have harmful consequences e.g. cholelithiasis References NICE TA248, NICE TA203, NICE CG87 Developed by Medicines & Prescribing Team, NHS Solihull CCG & Solihull Community Diabetes Service, Heart of England NHS Trust, May 2015 Approved for use in the NHS by Medicines & Prescribing Subcommittee, NHS Solihull CCG, July 2015. Not for commercial use. Hepatic impairment Elderly patients > 65 years Annual Cost NICE criteria for clinical use Lixisenatide 10mcg once daily for 14 days then increase to 20mcg daily on day 15 At the same time each day within the hour prior to a meal. Do not use in ESRD or if GFR is < 30ml/min. No dose adjustment No dose adjustment but experience in >75 years is limited £649 Follow existing NICE criteria. 5mcg BD increased to 10mcg BD after 1 month. Within the hour prior to a meal, with at least 6 hrs between injections. Do not use in ESRD or if GFR is < 30ml/min Increase from 5mcg to 10mcg with caution in those > 70 years £818 (Lyxumia®) Exenatide (Byetta®) Exenatide prolonged release 2mg once weekly (Bydureon®) Liraglutide (Victoza®) 0.6mg once daily. Increase after at least one week to 1.2mg daily. Avoid 1.8mg (NICE) No dose adjustment Do not use in ESRD or if GFR is < 50ml/min No dose adjustment At the same time each day independent of meals. Do not use in ESRD or if GFR is < 30ml/min Do not use Licensed indication Triple Insulin therapy Yes – when added to basal insulin +/-oral agents (except SU) Refer to local RICaD* Experience in > 75 is very limited On the same day each week at any time, independent of meals. Dual therapy + met + pio Renal impairment + met + SU Timing + pio Dose + SU Drug + met GLP-1 mimetics: Doses, licensed indications, NICE criteria & cost comparison Use only as part of triple therapy regime in combination with met + SU Yes – when added to basal insulin with met or pio, or met plus pio. NICE CG87 No dose adjustment but experience in >75 years is limited £953 No dose adjustment but experience in >75 years is limited £954 Use as part of dual therapy regime added to met or SU, or triple therapy added to met+SU or met+pio NICE TA 248 Dual therapy added to met or SU, or triple therapy added to met+SU or met+pio. The 1.8mg dose is not recommended NICE TA 203 Not licensed for use with insulin Yes – when added to basal insulin only. NOTE: information correct as at May 2015. Please refer to the individual SPCs for full details and up to date information. KEY: Met = metformin, Pio = pioglitazone, SU = sulphonylurea. Boxes shaded in grey are not covered by NICE &/or local guidance. * Lixisenatide is the only GLP-1 approved for use with insulin by the Area Prescribing Committee Formulary & needs a RICaD Developed by Medicines & Prescribing Team, NHS Solihull CCG & Solihull Community Diabetes Service, Heart of England NHS Trust, May 2015 Approved for use in the NHS by Medicines & Prescribing Subcommittee, NHS Solihull CCG, July 2015. Not for commercial use.