* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download PrP sc
Neglected tropical diseases wikipedia , lookup
Human cytomegalovirus wikipedia , lookup
Eradication of infectious diseases wikipedia , lookup
Hospital-acquired infection wikipedia , lookup
African trypanosomiasis wikipedia , lookup
Hepatitis B wikipedia , lookup
Schistosomiasis wikipedia , lookup
Coccidioidomycosis wikipedia , lookup
Oesophagostomum wikipedia , lookup
Sarcocystis wikipedia , lookup
Creutzfeldt–Jakob disease wikipedia , lookup
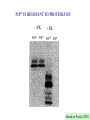
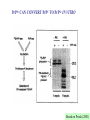
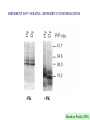
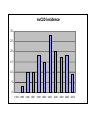
CLINICAL ASPECTS OF BIOCHEMISTRY NEURODEGENERATIVE DISEASES Prion diseases Alzheimer's disease SOME PRION DISEASES TRANSMISSIBLE SPONGIFORM ENCEPHALOPATHIES Species Transmission Scrapie Sheep Infection Bovine spongiform encephalopathy (BSE) Cow Infection (contaminated feed) Kuru Human Infection (cannibalism) Creutzfeldt-Jakob disease Iatrogenic Familial Sporadic Human Infection (growth hormone etc.) Germline mutations of PrP gene Somatic mutations of PrP gene or spontaneous conversion Infection (eating beef?) New variant Gertmann-StrausslerScheinker disease (GSS) human Germline mutations of PrP gene Fatal familial insomnia (FFI) Human Germline mutations of PrP gene SPONGIFORM BRAIN TISSUE From Prusiner (1998) HOW CAN PROTEINACEOUS PARTICLE BE INFECTIOUS? (a) May contain "shielded" nucleic acid? (b) Proteins specify own aa sequence? (c) Normal cells carry a gene that encodes PrP, and the product is changed to a different conformation by the infectious particle? THE PRION HYPOTHESIS TSEs occur when the normal ‘cellular’ form of the prion protein (PrPc) is converted to the abnormal form (PrPsc). PrPc and PrPsc differ in conformation. The conversion is ‘autocatalytic’ - PrPsc facilitates the conversion of more PrPc to PrPsc. TSEs - MAIN TOPICS 1. 2. 3. 4. 5. 6. 7. 8. 9. The nature of PrPc and PrPsc Conversion of PrPc to PrPsc Inherited TSEs Species specificity and species barriers Strains Normal function; role in disease Doppel BSE and nvCJD Therapies? Normal (PrPc) and abnormal (PrPs) forms of prion protein PrPc •Precursor ~ 250 amino acids. Mature PrPc ~ 210 aas •Hydrophobic glycoprotein •GPI anchor (glucosyl phosphatidyl inositol) •NMR structure - C-terminal end a-helical, N-terminal end unordered PrPsc •Same sequence and postranslational modifications as PrPc •Different conformation - more b-sheet •Tends to form insoluble aggregates •More resistant to proteolysis than PrPc •Insoluble PrPsc (in amyloid plaques) not infectious? PrPSc IS RESISTANT TO PROTEOLYSIS -PK +PK PrPSc PrPC PrPSc PrPC Based on Priola (2001) STRUCTURE OF THE HUMAN PRION PROTEIN Globular domain Based on Rivera-Milla et al (2003) HYPOTHETICAL MODELS FOR PrPc AND PrPsc Based on Prusiner (1998) alpha helix beta sheet PrPSc CAN CONVERT PrPC TO PrPSc IN VITRO Based on Priola (2001) TWO MODELS FOR CONVERSION OF PrPC TO PrPSc PrPSc PrPC (a) (b) ~6 Etc Etc Very slow fast fast fast INHERITED FORMS OF CJD, GSS, FFI etc Mutations may stabilise PrPsc conformation e.g. P102L in GSS [when this was engineered into mice they developed 'scrapie') Met/Val129 polymorphism in man - Val homozygotes more susceptible to CJD? Met homozygotes more susceptible to nvCJD? SOME POINT MUTATIONS IN THE PrP GENE THAT CAUSE HUMAN PRION DISEASE Position Normal Mutant 102 Pro Leu 105 Pro Leu 145 Ala Stop 178 Asp Asn 180 Val Ile 200 Glu Lys Based on Priola (2001) SPECIES BARRIERS TO TRANSFER OF PrPSc Sequence differences between PrP from different species may provide (and explain?) some barrier to infection - but incomplete. E.g. Mouse mouse transfer gives more rapid infection than mouse hamster etc. But, mouse hamster hamster gives faster infection, Homologous PrPSc is better at converting PrPC than heterologous STRAINS OF PRION DISEASES Scrapie occurs as about 20 different strains (differentiated by time taken to infect mice and different behavioural effects). CJD occurs as 2-4 different strains. BSE only one. May be explicable in terms of different conformations, but the more strains the more far-fetched this explanation. The biggest problem with the Prusiner model? For 2 CJD strains - evidence for different conformations (pattern of proteolysis) DIFFERENT PrPSc STRAINS - DIFFERENT CONFORMATIONS -PK +PK Based on Priola (2001) NORMAL FUNCTION OF PRPC Not clear; knockout mice lacking PrP are not seriously abnormal Possible roles in cell signalling and in processing copper ions have been suggested WHY DOES PrPSc CAUSE DISEASE? Possible explanations include: • Neurotoxic • Deposits disrupt cells • Deposits disrupt intercellular contacts (synapses etc) • Loss of PrPC DOPPEL (Dpl) A PrP-like protein (~25% sequence identity but shorter). Gene close to PrP gene - could explain variable effect of PrP knockouts Involvement in prion diseases? Based on Behrens & Aguzzi (2002) ANNUAL INCIDENCE OF BSE IN THE UK Number of BSE cases 40,000 30,000 20,000 10,000 16 83 663 85 86 87 88 89 90 91 92 93 94 Year of epidemic 95 96 nvCJD incidence 30 25 20 15 10 5 0 1994 1995 1996 1997 1998 1999 2000 2001 2002 2003 2004 Number of BSE cases 40,000 30,000 20,000 10,000 85 86 87 88 89 90 91 92 93 94 95 96 Year of epidemic nvCJD incidence 30 25 20 15 10 5 0 1994 1995 1996 1997 1998 1999 2000 2001 2002 2003 2004 CJD - POSSIBLE THERAPIES • Drugs that stabilise PrPC (stabilise helical conformation) • Drugs that inhibit aggregation & amyloid (b sheet) formation • Immunization against PrPC or PrPSc