* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Energy
Open energy system models wikipedia , lookup
Kinetic energy wikipedia , lookup
100% renewable energy wikipedia , lookup
Cogeneration wikipedia , lookup
Public schemes for energy efficient refurbishment wikipedia , lookup
Compressed air energy storage wikipedia , lookup
World energy consumption wikipedia , lookup
Energy storage wikipedia , lookup
Energy Charter Treaty wikipedia , lookup
Low-Income Home Energy Assistance Program wikipedia , lookup
Regenerative brake wikipedia , lookup
Zero-energy building wikipedia , lookup
International Energy Agency wikipedia , lookup
Low-carbon economy wikipedia , lookup
Energy returned on energy invested wikipedia , lookup
Energy efficiency in transport wikipedia , lookup
Energy policy of Finland wikipedia , lookup
Alternative energy wikipedia , lookup
Distributed generation wikipedia , lookup
Energy harvesting wikipedia , lookup
Gibbs free energy wikipedia , lookup
Internal energy wikipedia , lookup
Energy policy of the European Union wikipedia , lookup
Negawatt power wikipedia , lookup
Energy in the United Kingdom wikipedia , lookup
Micro combined heat and power wikipedia , lookup
Environmental impact of electricity generation wikipedia , lookup
Energy Independence and Security Act of 2007 wikipedia , lookup
Energy efficiency in British housing wikipedia , lookup
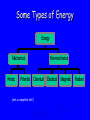
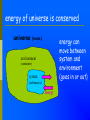
Energy Chapter 16 Energy: Ability to do Work Potential Energy (PE) = Energy of position aka STORED energy Kinetic Energy (KE) = Energy of motion Radiant Energy = Electromagnetic radiation ex: sunlight Some Types of Energy Energy Mechanical Kinetic Potential Non-mechanical Chemical Electrical (not a complete list!) Magnetic Radiant Units of Energy SI 1 system - unit of energy is JOULE (J) Joule ≅ amount of energy required to lift 1 golf ball about 1 meter other energy units: calorie (cal) Calorie (Cal) british thermal units (BTU) 1 calorie = 4.18 Joules 1 Calorie = 1000 cal = 1 kilocalorie = 4180J Kinetic Energy KE so = ½ x mass x velocity2 = ½ mv2 KE of matter depends on: how heavy and how fast Potential Energy stapler rubberband popper anything can have PE = energy of position = stored energy PE can be converted to KE Magnets PE in the system of 2 magnets depends on their relative position when magnets get close together they will pull together due to attraction when magnets are far apart they can’t attract each other Electromagnetic Radiation sunlight – visible radiation ultraviolet radiation infrared radiation gamma rays x-rays microwaves radiowaves Energy in Chemistry chemical energy: energy stored within chemical bonds between atoms heat: form of energy flows from warmer object to cooler object Heat Energy heat: energy associated with motion of atoms/molecules in matter symbol for heat energy = Q or q Heat Energy heat depends on amount of substance present can only measure changes in heat energy not absolute value of heat energy Temperature measure of average KE of particles in sub temperature is NOT energy TEMP does NOT depend on amount ENERGY does ! Law of Conservation of Energy energy is neither created nor destroyed in ordinary chemical or physical change energy before = energy after reminder: nuclear rxn a small amount of mass is converted to energy Energy can be converted from one form to another potential to kinetic golf ball hit off tee radiant to electric solar heat to electricity electric to heat electric stove cooking food chemical to kinetic burning charcoal on grill chemical to electrical batteries creating electricity ALL physical & chemical changes are accompanied by change in energy Thermochemistry: chemistry of energy changes Energy Transfer measure changes in heat (amount energy transferred from one substance to another) measure energy lost somewhere or energy gained somewhere else energy of universe is conserved universe (room) environment Environment (container) system (substances) energy energy can move between system and environment (goes in or out) EXothermic Change system releases heat to environment what happens to temperature of environment? temperature of system EXo - heat is EXiting what happens to temperature of system? environment temperature of environment system what happens to energy level of system? ↓ Exothermic Change energy lost = energy gained (system) (environment) Endothermic Change system absorbs heat from environment what happens to temperature of environment? temperature of environment ENdo - heat ENters system what happens to temperature of system? temperature of system environment system what happens to energy level of system? ↑ Endothermic Change energy lost = energy gained (environment) (system) Heat Flow heat ALWAYS flows from hotter object to cooler object cold pack on leg: heat flows from leg to cold pack! • leg cools down; cold pack warms up Calorimetry changes in heat energy are measured using calorimeter (energy lost = energy gained) difficult to monitor “system” easy to monitor “environment” (water) energy lost/gained by environment = energy gained/lost by system calorimeter: used to measure heat changes “universe” = styrofoam cup “enviroment” = water**** “system” is whatever put in water (reactants) quantity (amount) of heat transferred depends on: amount temperature change mass of substance specific heat of substance Calculating Heat Transferred simple system: •pure substance in single phase •calculate heat gained or lost using: Q = mcT Q = amount of heat transferred m = mass of substance c = specific heat capacity of the substance T = temperature change = Tfinal – Tinitial Specific Heat amount heat energy required to raise temp of 1 gram of substance by 1oC symbol =c specific heat = a physical constant unique see for each pure substance Table B for water (4.18J/g˚C) typical word problem 10 grams of NaOH(s) is dissolved in 100 g of water & the temperature of the water increases from 22C to 30C. dissolving process: exothermic was it endothermic or exothermic? how do you know? temperature ↑ Solid Dissolving in Water What’s happening when NaOH dissolves? Add H2O NaOH molecules close together, not interacting NaOH molecules pulled apart & interact with H2O molecules Calorimetry calculate energy released by NaOH as it dissolves in water energy lost by NaOH = energy gained by water • easier to calculate from H2O perspective Q = mcT Q = energy (Joules) m = mass (grams) c = specific heat capacity (Table B) T = temperature change = Tf - Ti Calorimetry & Q = mCT temperature to 30C what of water increased from 22C 30C -22C = 8C = T mass to use? temp change was for water, so use mass H2O m= 100 g same goes for specific heat capacity; calculate heat absorbed by water C H 0 = 4.18J/gC 2 Q = mcT Q = (100 g)(4.18 _J )(8C) g C Q = 3344 J Stability and Energy if energy is high, stability is low if energy is low, stability is high