* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download A Practical Overview of Antiarrhythmic Drugs Commonly Used in
Survey
Document related concepts
Transcript
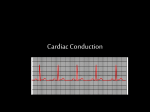
A Practical Overview of Antiarrhythmic Drugs Commonly Used in Atrial Fibrillation RESOURCE SESSION Olavo Fernandes, Pharm.D. Pharmacy Practice Leader, Toronto General Hospital, UHN Assistant Professor, University of Toronto October 2002 Cardiac Conduction System SA node – primary pacemaker (60100 bpm) – autonomic nervous system – vagus nerve (parasym.) – catacholamines (sym) AV node – filter (40-60 bpm) – controls number of impulses reaching the ventricle from atria Bundle of His – conducts impulses to bundle branches Perkinje system – < 40 bpm – bifarcates into several bundle brunches Electrocardiogram (ECG) P wave – repolarization of ventricle – depolarization of atria QRS Complex – depolarization of ventricle – QRS interval normally < 0.12 secs. – widened QRS: prolonged conduction – QRS > 0.12 secs: conduction from ventricle or supraventricular T wave U wave – uncertain PR interval – < 0.2 seconds – conduction velocity – beginning of P wave to onset of QRS QTc interval – < 0.4 seconds – refractory period – beginning of Q to end of T Cardiac Action Potentials Sodium-dependent Fibres/ Fast Fibres – atrial and ventricular tissue – Phase O, 1, 2, 3, 4 Calcium-dependent Fibres – – – – – SA and AV nodes only 3 phases Ca enters instead of Na in Phase O higher resting membrane potential increase in slope of phase 4 Refractory Period Automaticity – intrinsic property of spontaneous impulse generation Classification (Circulation 2001; 104: 2118-2150) Atrial Arrhythmias Goals of Therapy convert to sinus rhythm control ventricular response rate relieve associated symptoms (palpitations, fatigue, dyspnea, syncope, angina, heart failure) prevent recurrence prevent complications: life threatening arrhythmias, stroke, MI, tachycardia Atrial Fibrillation- Rate Control Why use an agent for rate control? » better filling time, better diastolic function » consider risks of conversion, embolic risks, drug side effects OPTIONS – Digoxin » increase vagal tone (decrease AV node conduction), inhibit Na/K Pump – Beta Blockers ( metoprolol, esmolol, atenolol) » slow SA node, slow AV node conduction, effect on refractory period – Calcium Channel Blockers (diltiazem, verapamil) » block slow calcium channels, slow AV node conduction Rate Control Agent Considerations DIGOXIN – vagally mediated – slow onset- 4hrs (large VD) – poor effectiveness during high sympathetic tone (stress) – positive inotrope (benefit with concurrent CHF) – proarrhythmic, side effects Rate Control Agent Considerations BETA BLOCKERS – faster onset (minutes) – directly effect on AV node (effective during stress) – negative inotrope (concern in CHF) – may be useful with concurrent CAD/Post MI patients – bronchospasm (asthma) – effect on blood sugar (DM) – main SE: hypotension CALCIUM CHANNEL BLOCKERS – faster onset (minutes) – directly effect on AV node (effective during stress) – negative inotrope (concern in CHF) verapamil > diltiazem – may be useful with concurrent CAD/Post MI patients – main side effect:hypotension – cost diltiazem > verapamil Conversion of Atrial Fibrillation Considerations – better cardiac function, more times in A Fib harder to convert to NSR, emboli and anticoagulation, may need rate control during conversion Direct Current Conversion – 100J, 200J, 300J, 360J – burns, relapse, sedation, worsened arrhythmias Pharmacological Options – Amiodarone (least proarrhythmic, some AVN block, least negative inotropy, very costly IV) – Procainamide, Sotalol – Quinidine – Ibutilide Management of newly discovered AF (Circulation 2001; 104: 2118-2150) Pharmacologic cardioversion of AF<7 days (Circulation 2001; 104: 2118-2150) Pharmacologic cardioversion of AF>7 days (Circulation 2001; 104: 2118-2150 Pharmacological management of patients with recurrent AF (Circulation 2001;1104: 2118-2150) Recurrent paroxysmal AF Minimal or no symptoms Anticoagulation and rate control as needed Disabling symptoms Anticoagulation and rate control as needed Heart Disease No (or minimal)* Yes Flecainide Propafenone Sotalol HF CAD Hypertension Amiodarone Amiodarone Sotalol LVH>1.4 cm Disopyramide Procainamide Quinidine non pclg Amiodarone Yes No Disopryamide Procainamide Quinidine Amiodarone Flecainide Propafenone Amiodarone Sotalol Disopryamide Procainamide Quinidine Drug Profile: Digoxin – binds and inhibits Na/K ATPase – slows AV node conduction (vagus nerve) – time to steady state: 5-7 days (2-3 wks in RF) – elimination half life: 35-40 hrs (2-5 days in RF) Mechanism – CHF, AF- rate control Kinetics – dist: wide 7-8 L/kg (skeletal muscle, myocardium, kidneys) – ptn binding : 20-40% – elimination: renal 70-80%; hepatic (up to 30%) – time to peak: 1-4 hr (IV); 2-6 hr (po) Indications Dosing – load: 0.250mg IV over 15 min repeat in 6hr; 0.250 mg po q6h x 4 (total 1 mg) – renal dysfunction load 0.125mg q6h (total 0.5-0.75 mg) – initial mx dose: 0.125 - 0.250 mg po Drug Profile: Digoxin Adverse Effects – GI: N, V, A, D – CNS: disorientation, confusion – Ocular: colour vision disturbances (yellow-green), halos, photophobia – CV: bradycardia, heart block, VT – more prone to toxicity with hypokalemia (sensitizes myocardium to digoxin effect) – toxicity: hyperkalemia, ventricular arrhythmias, visual disturbances – DIGIBIND Drug Interactions – physically adsorption (antacids, sucralfate, resins) – antibiotics (digoxin metabolized by gut bacteria) – increased serum level (quinidine, amiodarone, verapamil) – assay interference (spironolactone) Monitoring – ECG, HR, renal function, potassium, digoxin levels Digoxin: serum levels Therapeutic Range – 1.2- 2.5 nmol/L – AFib at higher end of target – not < 8 hrs and preferably trough level before next dose – at least 5 days after starting tx or changing dose – Increased levels Indications – suspected toxicity/ confirmation – initiation or change in therapy – changes in renal function – clinical deterioration – addition of interacting medications – routine monitoring - yearly – subtherapeutic response Sample Collection Time Increased Levels – advanced age, renal disease, hepatic disease, amiodarone, verapamil, CsA, quinidine Decreased levels – hyperthyroidism, binding drug interactions Drug Profile: Digoxin ADVANTAGES Positive inotrope Maybe useful in patients with concurrent AF / CHF LIMITATIONS Limited to atrial arrhythmias Limited efficacy Pro-arrhythmic Narrow therapeutic range Limited efficacy during high sympathetic tone Caution with adverse effects/ drug interactions Drug Profile: Diltiazem Mechanism: – blocks slow calcium channels – slows AV node conduction – vasodilatation Adverse Effects – IV: hypotension, bradycardia – worsening CHF symptoms – heart block Administration/Dosing – bolus and continuous infusion – 0.25 mg/kg over 2 minutes, after 15 minutes can give 0.35 mg/kg over 2 minutes – continuous infusion 10mg/hr (up to 15mg/hr) x 24 hr – D5W, NSS, 2/3-1/3 – refrigerated for storage Drug Interactions – AV node blocking agents (BB, CCB, digoxin) – hypotensive agents – negative inotropes Monitor – ECG, BP, HR – CHF symptoms Drug Profile: Metoprolol Mechanism: – competitive block agonist effect of sympathetic neurotransmitters – block adrenergic stimulation of cardiac action potentials – Beta-1 selective agent – slows AV node conduction – IV: hypotension, bradycardia – worsening CHF symptoms – bronchospasm in asthma – heart block Drug Interactions – AV node blocking agents ( CCB, digoxin) – hypotensive agents – negative inotropes Administration/Dosing – 12.5 - 100 mg po bid – 5mg IV push for acute management ( if necessary 10-15 mg IV q 6h) Adverse Effects Monitor – ECG, BP, HR – CHF symptoms Drug Profile: Amiodarone – complex pcl profile: actions of all classes – conduction slowing (class I) – BB activity (class II) – prolong APD and refractory period (class III) – AVN conduction slowing (IV) – blocks cellular K channels – elimination: primarily hepatic metabolism / biliary excretion – active metabolite: desethylamiodarone Mechanism: Kinetics – bioavailability: 35-65% – half life: mean 52 days – volume of distribution: 5000L Kinetic Implications » loading doses » delayed AA effect » delayed elimination if drug stopped » compliance » role of levels Drug Profile: Amiodarone Indications IV – suppression of recurrent sustained VT, ongoing VT/VF, acute conversion of AF – CAMIAT and EMIAT – showed amio. - low incidence of proarrhythmia; safe in LV dysfunction Primary prevention of SCD – RFs for SCD: LV dysfunction, frequent or complex ectopics – GESICSA and CHF STAT – MADIT (ICD) Atrial Fibrillation/Flutter – slow VRR (AVN block) – convert to NSR – maintain NSR after conversion – dose lower in atrial arrhythmias Post MI Secondary prevention -SCD – CASCADE, AVID – ICD preferred – amio. second line Drug Profile: Amiodarone Administration/Dosing – 150-300 mg IV over 10 minutes followed by 0.5 - 2 mg/kg minute infusion (1000mg / 24 hrs) – See handout – continue oral load over several weeks – Ventricular arrhythmias » 1200-1800 mg/d x 1-2 wks; 800mg/d x 2 wks; 600mg/d x 4 wks; then 200-400mg/d – Atrial arrhythmias » 600-800 mg/d x 4wks; 400 mg/d x 2-4 wks; 200 mg/d thereafter Adverse Effects (IV) – hypotension (related to vehicle) – peripheral vein phlebitis (run at < 2mg/ml) – IV infusions > 2 hrs administer in glass bottles of D5W – proarrhythmia : rare – 100% liver metabolism Monitor – vitals, ECG, BP, HR – vein site for phlebitis Amiodarone :Adverse Effects Long term oral therapy – 80% report side effects, in trials only 10-20% have side effects necessitating withdrawal – SE appear to be dose related – minimize doses/ reduce dose is sx occur – regular monitoring in preventing and managing SEs CV – sinus bradycardia (0-10%), AV conduction disturbances and heart block (2-5%), rare TdP – increase plasma cholesterol Pulmonary – most feared adverse effect – pulmonary fibrosis (2-7%) can be fatal in 10% of cases – appears in pts with > 400mg/ day – CXR: bilateral and diffuse changes/ interstitial infiltrates – Sx: dyspnea, cough, chest pain, pleural rub Amiodarone : Adverse Effects Pulmonary – type 1: hypersensitivity (after first few weeks) with development of fever, SOB, cough; tx : stop amio. – type 2: interstitial / alveolar pneumonitis (7 mos - 2 yrs); insidious onset of nonproductive cough, fatigue, SOB, pleuritic CP, fever; infiltrates and pulmonary fibrosis on CXR; tx: stop amio GI – increase in AST/ALT/ ALP (25%) – hepatitis, hepatic failure – N, V, A common Thyroid – inhibits conversion T4 to T3 – hypo (3%) or hyperthyroidism (2%) – hypothyroidism: » rare after first 18 months » responds to thyroid replacement – hyperthyroidism » occur at any time, difficult to manage (thyroidectomy) Amiodarone: Adverse Effects Dermatolgicial – skin reaction (15%) – photosensitivity (10%)- limit sun exposure and sunscreen – long term blue-gray discoloration (1-7%) – labs (renal function, CBC, LFTs, thyroid function), ECG, CXR » baseline, as sx occur and every 4-6 months – PFTs, eye exam: baseline and as symptoms occur – ECG, HR, BP at regular intervals CNS – 40% have CNS effects (fatigue, tremor, ataxia, peripheral neuropathy) Optho. – corneal deposits – rare: visual disturbances – sx: photophobia, blurred vision, blue-green halos Monitoring Drug Interactions – warfarin, digoxin, BB, CCB, procainamide, quinidine – (see handout) Drug Profile: Amiodarone ADVANTAGES effective in a wide range of arrhythmias neutral effects on inotropy (CHF patients) safety in CAD and LV dysfunction low incidence of proarrhythmia some rate control properties in AF LIMITATIONS many chronic side effects drug interactions IV amiodarone - very expensive Drug Profile: Procainamide – Class 1a AA- blocks Na channels – prolongs refractory period and decreases conduction velocity Kinetic Implications – active metabolite- NAPA Adverse Effects – – – – – Administration/Dosing – IV and PO regimens – depends on indication and renal function – paradoxical initial increase in HR – levels to prevent toxicity (drug and NAPA) Mechanism: hypotension, bradycardia proarrhythmic (TdP) drug-induced lupus (SLE) GI: nausea, vomiting CNS: disorientation Drug Interactions – amiodarone, Septra (TMP/SMX) Monitor – ECG, BP, HR