* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download File - Miss Cummings
Survey
Document related concepts
Transcript
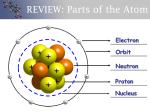
Standards: SCSh1-9 SC3 Students will use the modern atomic theory to explain the characteristics of atoms. b. Use the orbital configuration of neutral atoms to explain its effect on the atom’s chemical properties. SC4. Students will use the organization of the Periodic Table to predict properties of elements. a. Use the Periodic Table to predict periodic trends including atomic radii, ionic radii, ionization energy, and electronegativity of various elements. b. Compare and contrast trends in the chemical and physical properties of elements and their placement on the Periodic Table. Unit Four: The Periodic Table History of the Periodic Table Dmitri Mendeleev is considered the Father of the Periodic Table. He arranged elements by increasing atomic mass and predicted the properties of missing elements. Henry Moseley discovered that each element has a unique atomic number and arranged the elements by increasing atomic number rather than my Mendeleev’s atomic mass. Modern Periodic Law states that properties of elements repeat periodically or in a pattern when elements are arranged by Moseley’s increasing atomic number (rather than my Mendeleev’s atomic mass). Organization of the Periodic Table There are 7 rows/periods, one for each of the 7 energy levels (quantum number = n). The lanthanides and actinides (part of row 6 and 7) were moved and placed down at the bottom to make the table shorter and easier to fit on one page. There are 18 columns/groups/families. Elements in the same column/group/family have similar electron distributions and the same number of valence electrons. Valence Electrons- electrons in the outer energy level that are available for chemical bonding (Note: For columns 1-2 and 13-18, the number in the one’s place represents the number of valence electrons.) Metals are shiny, hard, malleable (can be pounded into sheets), ductile (can be drawn into wires), good conductors of heat and electricity, have high density, and have high melting and boiling points. Nonmetals are brittle, poor conductors of heat and electricity, have low density, and have low melting and boiling points. Semimetals have properties of both metals and nonmetals, are brittle, have high melting and boiling points, have high density, and are semiconductors of electricity. Everything to the right of the semimetal line is a nonmetal, while everything to the left is a metal, except Hydrogen. State of Matter: There are two liquids on the periodic table: Mercury/Hg and Bromine/Br. There are twelve gases on the periodic table: Hydrogen, Nitrogen, Oxygen, Fluorine, Chlorine, and ALL of the Noble Gases. (Think “Heck No Obese Fat NOBLE CLown) Everything else on the periodic table is a solid. Miss Cummings – Chemistry 1 Electron Configurations Simplified All elements want to be like the nearest noble gas, because noble gases are very stable with completely filled valence shells (Octet Rule; 8 Electrons in the Highest Energy Level). Note: The first energy level can hold only 2 electrons, this is why helium is placed with the noble gases. Example: Neon 1s22s22p6 Neon follows the octet rule because 2 is highest energy level given and it has 8 electrons total, 2 in the s sublevel and 6 in the p sublevel. Noble gases can be used to abbreviate electron configurations. Instead of writing the entire electron configuration, you can replace part of the electron configuration with the noble gas found right before the element in brackets. Example: Sulfur Unabbreviated 1s22s22p6 3s23p4 Sulfur Abbreviated [Ne] 3s23p4 Instead of using Aufbau/the “Diagonal Rule” to write electron configurations, you can use the block method shown below. Trends on the Periodic Table Atomic radius is half the distance between the nuclei of two like atoms. It increases down a family/group, as more energy levels are added, making the atom bigger. It decreases across a period, as the number of protons increases and there is a stronger pull on the electrons, making the atom smaller. Electronegativity is a measure of how much an atom pulls electrons away from other atoms. It increases across a period as elements want to gain electrons to be more like the noble gases on the right side. It decreases down the family/group, because adding more energy levels increases the distance between the valence electrons and the positive nucleus. Miss Cummings – Chemistry 2 Ion- any atom that has an overall positive or negative charge Cation- atom that has a positive charge because it has given an electron away (Think “It is positive to give.”//CATions are PAWsitive.) Anion- atom that has a negative charge because it has stolen an electron (Think “It is negative to steal.”) [Note: ISOTOPES vary neutrons and IONS vary electrons.] Note: Ionization energy is the energy necessary to make an ion. It increases across a period and decreases down a family/group, because as the atomic radius decreases, it takes more energy to pull electrons away from the nearby positive nucleus. Ionic radius Cations are smaller because there are fewer electrons extending from the nucleus, while anions are larger because there are more electrons extending from the nucleus. NOTE: All trends are towards Fluorine, except RADIUs, which increases towards RADIUm. Miss Cummings – Chemistry 3