* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download NNIN Nanotechnology Open Textbook, Chapter 2
Survey
Document related concepts
Transcript
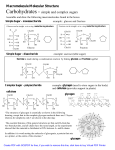
2.2: Sugars and Polysaccharides François Baneyx Department of Chemical Engineering, University of Washington [email protected] Carbohydrates or saccharides are abundant compounds that play regulatory and structural roles and serve as cellular fuel and energy storage. Carbohydrates have for general formula (CH2O)n, where n 3. They are synthesized from atmospheric CO2 and H2O via photosynthesis and polymerized into more complex polysaccharides such as starch, glycogen and cellulose. The simplest carbohydrates are monosaccharides. These simple sugars contain three to nine carbon atoms and are classified as aldoses or ketoses depending on whether their carbonyl group is an aldehyde or a ketone. The smallest monosaccharides are trioses that only contain three carbon atoms. Those sugars containing four, five, six, etc, carbon atoms are called tetroses, pentoses, hexoses, etc, respectively. Table 1 shows the chemical structure of common monosaccharides. A particularly important monosaccharide is glucose which is used to power many cellular processes and serves as a precursor for the synthesis of more complex carbohydrates. Fig. 1 shows that carbons 2to-5 of D-glucose are chiral, leading to 24 = 16 possible stereoisomers that correspond to all possible aldohexoses (four of which are shown in Table 1). While glucose can be found in both L and D forms, the L form is less abundant in Nature and L-glucose only plays a minor role in biological systems. In solution, D-glucose adopts a ring structure (known as pyranose for a 6-membered ring) that results from the reaction of its carbonyl group and alcohol groups (Fig. 1). The denomination refers to the location of the OH substituent on carbon 1 of the ring. If the hydroxyl were located on the same side of the ring as the CH2OH group, the resulting molecule would be called -D-glucose. H O CH2OH 1C H C 2 OH 3 C OH H OH H C OH 4 6 C C OH HO H C 5 H 6 CH2OH OH H H C C H HO C H O 4 5 C HO 3 C O OH H C 2 C H HO H 1 C OH -D-Glucose (-D-Glucopyranose) CH2OH D-Glucose Fig. 1 Formation of cyclic -D-Glucose from its linear form. (linear) NNIN Nanotechnology Open Textbook, Chapter 2 – Biochemistry Fundamentals François Baneyx– Copyright 2006 Page 1 of 7 Table 1 Structure of some common monosaccharides Aldoses D-Triose CHO H C OH CH2OH D-Glyceraldehyde D-Tetroses CHO CHO H C OH H C OH H C OH CH2OH CH2OH D-Erythrose D-Threose OH D-Pentoses CHO H C OH CHO CHO OH C H H C OH H C OH H C OH H C OH H C OH H C OH CH2OH CH2OH CH2OH OH C H D-Ribose D-Arabinose D-Xylose CHO CHO CHO H C OH D-Hexoses C H H C OH H C OH OH C H H C OH H C OH H C OH H C OH CH2OH CH2OH D-Allose D-Glucose H C CHO OH OH C H OH C H OH C H OH C H H C OH OH H C OH CH2OH CH2OH D-Galactose D-Mannose H C NNIN Nanotechnology Open Textbook, Chapter 2 – Biochemistry Fundamentals François Baneyx– Copyright 2006 Page 2 of 7 Table 1 (Continued) Ketotriose Ketoses CH2OH C O CH2OH Dihydroxyacetone Ketotetrose CH2OH C O H C OH CH2OH D-Erythrulose D-Hexoses Ketopentoses CH2OH C CH2OH O H C OH H C OH C OH O C H H C OH CH2OH CH2OH D-Ribulose D-Xylulose CH2OH CH2OH CH2OH CH2OH C O C O C O C O OH C H H C OH OH C H OH C H H C OH H C OH H C OH H C OH H C OH H C OH H C OH CH2OH CH2OH CH2OH CH2OH D-Sorbose D-Togatose D-Psicose D-Fructose OH C H NNIN Nanotechnology Open Textbook, Chapter 2 – Biochemistry Fundamentals François Baneyx– Copyright 2006 Page 3 of 7 D-ribose and D-deoxyribose are two other important monosaccharides that are structural components of RNA and DNA, respectively. The structure of these five carbon ring sugars is shown in Fig. 2. 5 CH2OH 5 CH2OH OH O 1 4 Fig. 2 Structures of D-ribose (left) and D-deoxyribose (right). The two molecules differ by the presence or absence of a hydroxyl group on carbon 2. Not all hydrogen atoms are shown. OH O 1 4 3 2 3 2 OH OH OH H Monosaccharides can polymerize via condensation reactions to yield polysaccharides. The simplest case involves two simple sugars and the generation of a disaccharide. Depending on whether the reaction proceeds with the or form of the sugar, an -1,4 or -1,4 glycosidic bond is generated, giving rise to different disaccharides as illustrated in the case of D-glucose in Fig. 3. 6 CH2OH 6 CH2OH 5 5 O 4 1 OH HO + OH 3 O 4 1 HO 3 -D-Glucose 6 CH2OH 6 CH2OH 5 HO + 4 2 H2O OH OH OH -D-Glucose -D-Glucose 3 2 OH 6 CH2OH 6 CH2OH 5 5 O 4 1 OH 2 OH 2 Maltose (-1,4 glycosidic bond) 1 3 1 OH OH OH HO 3 O 4 O 3 O OH 1 1 HO 2 -D-Glucose OH 5 O 4 OH OH 4 5 OH OH O 6 CH2OH OH 2 5 H2O 6 CH2OH O O 4 1 OH HO OH 3 2 3 OH 2 OH Cellobiose (-1,4 glycosidic bond) Fig. 3 Maltose and cellobiose both result from the condensation of two D-glucose molecules. However, whereas maltose contains an -1,4 glycosidic bond, cellobiose contains a -1,4 bond. NNIN Nanotechnology Open Textbook, Chapter 2 – Biochemistry Fundamentals François Baneyx– Copyright 2006 Page 4 of 7 In addition to reacting with carbon 4, the hydroxyl group of carbon 1 in one sugar may also react with carbon 6 of the second sugar, leading to the formation of an -1,6 glycosidic bond (Fig. 4). Such bonds act as branch points in more complex polysaccharides. 6 CH2OH 6 CH2OH 5 5 O 4 1 OH HO OH 3 2 OH -D-Glucose + 6 CH2OH O 4 5 H2O 1 5 O 4 4 1 OH HO OH 2 OH HO HO 3 OH -D-Glucose O O 1 OH 3 6 CH2 2 OH 3 OH 2 OH Isomaltose (-1,6 glycosidic bond) Fig. 4 Formation of an -1,6 glycosidic bond between two -D-Glucose molecules. Cellulose, the major structural constituent of plants cell wall is one of the most abundant molecules on earth, accounting for more than half the carbon in the biosphere. Cellulose is a linear polymer of up to 15,000 D-glucose monomers linked through -1,4 glycosidic bonds (Fig. 5a). The length of the polymer is variable and cellulose molecular mass typically varies between 50-kDa and 1a 6 CH2OH 6 CH2OH MDa. In wood, cellulose fibers are embedded in a matrix consisting of 5 5 O O other polysaccharides and lignin (a phenolic polymer), yielding a 4 1 O 4 1 OH OH composite material that exhibits remarkable mechanical properties. 3 2 3 2 Because it is tightly packed and n OH OH hydrogen-bonded, cellulose is difficult to degrade into its constituent sugars. 6 CH2OH 6 CH2OH In herbivores, this slow process is b 5 5 catalyzed by cellulases produced by O O symbiotic organisms living in the 4 1 O 4 1 animal intestinal tract. A related OH OH compound, chitin, is the main structural component of invertebrates n 3 2 3 2 exoskeleton although it is also found in NHCCH3 NHCCH3 the cell wall of fungi and certain algae. Chitin is a linear polymer of N-acetylO O D-glucosamine residues linked by - Fig. 5 Structures of cellulose (a) and chitin (b). 1,4 glycosidic bonds (Fig. 5b). NNIN Nanotechnology Open Textbook, Chapter 2 – Biochemistry Fundamentals François Baneyx– Copyright 2006 Page 5 of 7 In addition to playing a structural role, polysaccharides may also serve as nutritional reservoirs. Starch, which is synthesized as insoluble granules by plants, is composed of a mixture of -amylose (a linear polymer of thousands of -D-glucose molecules linked to each other through -1,4 glycosidic bonds) and amylopectin (which consists of -1,4linked glucose chains branching every 25-to-30 residues via -1,6 glycosidic bonds). a 6 CH2OH 6 CH2OH 6 CH2OH 6 CH2OH 5 5 5 5 O 4 1 4 OH 2 b 1 2 OH 6 CH2OH 6 CH2OH 5 5 4 1 OH 2 O 3 2 OH O 1 OH 2 O OH 3 2 OH 6 CH2OH 6 CH2 5 5 O 4 1 OH 3 1 OH OH 4 O 3 O 4 O 3 OH O 1 OH O 3 O 4 OH O 3 O 1 OH 3 OH 5 O 4 O 2 6 CH2OH O 4 1 OH O 2 OH 3 O 2 OH Fig. 6 Structures of amylose (a) and amylopectin (b). Although both are polymers of glucose joined through -1,4 glycosidic bonds, amylopectin branches through -1,6 glycosidic bonds. The molecular mass of amylose, which makes up about 20% of starch, varies between a few thousands Da to 500-kDa (Fig. 6a). Amylopectin, one of the largest molecules in Nature, has a molecular mass that ranges from 1- to 2-MDa (Fig. 6b). In animals, the corresponding storage polysaccharide is glycogen which is chemically similar to amylopecin but much more highly branched (every 8-to-12 glucose residues on the average; Fig. 7b). In contrast to cellulose, amylose, amylopectin and glycogen adopt a NNIN Nanotechnology Open Textbook, Chapter 2 – Biochemistry Fundamentals François Baneyx– Copyright 2006 Page 6 of 7 more amorphous structure because the -1,4 glycosidic bonds do not cause successive glucose residues to flip 180º with respect to each other as -1,4 glycosidic bonds do. Thus, all these compounds can be easily digested allowing for a rapid infusion of glucose in times of metabolic need (Fig. 7). a b Fig. 7. Schematic representation of the structural organization of amylopectin (a) and glycogen (b). In amylopectin, side branches are clustered while glycogen adopts a tree-like structure. NNIN Nanotechnology Open Textbook, Chapter 2 – Biochemistry Fundamentals François Baneyx– Copyright 2006 Page 7 of 7