* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Rotational and Vibrational Levels of Molecules
Hydrogen atom wikipedia , lookup
Wave–particle duality wikipedia , lookup
Particle in a box wikipedia , lookup
Rigid rotor wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Atomic theory wikipedia , lookup
Symmetry in quantum mechanics wikipedia , lookup
X-ray fluorescence wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
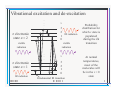
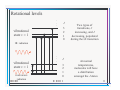
Rotational and Vibrational Levels of Molecules Lecture 23 www.physics.uoguelph.ca/~pgarrett/Teaching.html Review of L-21 • Beer-Lambert law I = I 0e • Transmittance • Absorbance − Cx = I 0e − x I (λ ) T= I 0 (λ ) I 0 (λ ) = 0.4343 x A = log I (λ ) • Extinction coefficient = 0.4343 Rotations and vibrations • We have examined electronic transitions in molecules – But they can also rotate and vibrate – ex. O2 Rotations and vibrations • Become more complicated for more complex molecules – ex. H2O rotational modes Rotations and vibrations • Become more complicated for more complex molecules – ex. H2O vibrational modes Vibrations • Just like the electrons, molecular motion is governed by quantum mechanics – Energies due to rotation and vibration are quantized • Molecular vibrations – Chemical bond acts like a spring and can display SHM – Have an effective spring constant k for the bond involved and effective mass meff – Angular frequency k = meff – Energy of vibration Ev = (v + 1 2 ) = (v + 1 2 )hf – ½ ω comes from quantum mechanics and represents zero-point energy Vibrations • Vibrational energy Ev = (v + 1 2 ) = (v + 1 2 )hf • Vibrational quantum number v = 0,1,2,3,… • The zero point energy ½ ω implies molecule never stops vibrating, even when its in the v = 0 state! – Zero point energy cannot be harvested or extracted – Still exists at absolute zero • All molecules are then in v = 0 state • Energy levels are equally spaced with separation ω • Obey selection rule ∆v = ±1 if no accompanying electronic transition – Otherwise can be anything Molecular vibrations • For diatomic molecule with mass M1 and M2, effective mass meff can take the simple form M 1M 2 meff = M1 + M 2 • Energy scale for molecular vibrations is much less than for electronic excitations • Excitation energies correspond to IR region of the spectrum – Typical wavelengths are 2 – 50 µm = 2000 – 50000 nm for organic molecules • Vibrational levels are built on electronic states – each electronic state will host the whole range of vibrational states Vibrational excitation and de-excitation . . . v 3 2 1 IR radiation 0 π electronic state n = 2 visible radiation v 3 2 1 0 Fundamental IR transition IR radiation . . . π electronic state n = 1 visible radiation Probability distribution for which v state is populated during the ∆n transition At normal temperatures, most of the molecules will be in the v = 0 state Molecular rotations • In quantum mechanics, the rigid rotor has energy levels EJ = 2 J (J + 1) 2ℑ where ℑ is the moment of inertia (PHY1080), J is the angular momentum, J = 0,1,2,3,… 2 • The quantity 2ℑ is called the rotational parameter • Moment of inertia, hence rotational parameter, can be different for each rotation axis • Excitation energies correspond to the microwave region • Energy scale for rotations << vibrations – Each vibrational level has rotational bands built on it • Selection rule ∆J = ±1 Rotational levels . . . J 3 2 1 0 vibrational state v = 1 Two types of transitions, J increasing, and J decreasing, populated during the ∆v transition IR radiation At normal temperatures, molecules will have a distribution amongst the J states 3 2 1 0 microwave radiation vibrational state v = 1 J . . . Vibrational-rotational IR spectrum • HCl 2→ →3 1→ →2 3→ →4 4→ →5 5→ →6 6→ →7 7→ →8 8→ →9 9→ →10 10→ →11 11→ →12 Energy levels • Taking rotations, vibrations, and electronic excitation into account E n ,v , J = E n + E v + E J E n ,v , J n 2h 2 = + (v + 1 2 ) 2 2me + 2 2ℑ J (J + 1) Ring molecule • If the measuring instrument has very good resolution, it is possible to see the discrete transitions • Complex molecules may have many vibrational modes, rotational modes, etc. The combination of these different modes leads to a “smearing” of the discrete spectrum (temp. effects too) so that broad bumps appear rather than discrete lines Water absorption spectrum Radiowave IR