* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download crystalline solids report instructions introduction
Survey
Document related concepts
Transcript
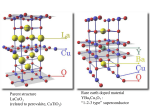
Crystalline Solids Revised 2/8/16 CRYSTALLINE SOLIDS Adapted from Experiments by A.B. Ellis et al, ICE REPORT INSTRUCTIONS All in-lab work for this experiment must be recorded or attached to the ELN. Create a Pre/InLab page in this week’s Experiment folder. Typical lab report sections are NOT required. Because of limitations with the number of models, you will probably need to perform the parts of this experiment out of the order indicated. Many pictures and screenshots should be attached in the observations section. Students will work in pairs. Each pair will turn in a pictorial group report that should consist mostly of annotated pictures. (Make a picture book showing what was learned in lab.) INTRODUCTION Material science, which encompasses the traditional scientific disciplines of chemistry, physics, and engineering, is the study of the synthesis, composition, and properties of solids. In this experiment the composition and properties of one class of material – crystalline solids containing cube-like building blocks called cubic unit cells. Read “Crystal Structures with Cubic Unit Cells” before proceeding further. Both physical and computational models will be used to further understand the structure of solids in this experiment. Also, the properties of nitinol and a superconductor will be investigated. SAFETY PRECAUTIONS Safety goggles and lab aprons must be worn in lab at all times. Liquid nitrogen is extremely cold (–321°F!). Contact with skin may result in severe frostbite. If any liquid nitrogen spills on clothing, remove the clothing immediately, as the trapped liquid will cause severe frostbite to the skin beneath the clothing. Do not touch any metal dipped into liquid nitrogen until it has warmed to room temperature. Do not place liquid nitrogen in a closed container; it can rapidly expand and explosively shatter a container that is not properly vented. Use plastic tweezers to handle superconductors and rare earth magnets; the solids may be toxic. The solid state models 1 Crystalline Solids Revised 2/8/16 contain small spheres and rods - if any are dropped on the floor, pick them up to prevent slips or falls. Wash your hands before leaving lab. Report any spills, accidents, or injuries to your TA. PROCEDURES Before starting the experiment, the TA will ask you to do a quick demonstration or talk-through one of the following: 1) How to pour liquid nitrogen. 2) Assemble the superconductor setup for this experiment (without the liquid nitrogen). Make sure you watch the videos on the course website and read the documents to prepare. These demonstrations will be done every week. Everyone will have presented at least one topic by the end of the quarter. The demonstrations should be short (>1 min) and will be graded. Part A: Models and Odyssey Before coming to lab look through the following Odyssey modules: 99 – The Structure of Elemental Solids, 102 – Crystal Cubic Lattices, 103 – Stacking Orders, 104 – Interstitial Holes, 105 – Ionic Solid Structures. You will need to take screenshots from these modules to match up with the pictures you take of the marble and stick models in lab. These screen shots can be taken during or after lab, but you must look at the Odyssey modules while looking at the marble and stick models in lab so that your pictures will be comparable. In lab, you will find the material to build one of four generic (all colorless) models with cubic unit cells: simple (primitive) cubic, body centered cubic, cubic close packed, and face centered cubic. Your TA will assign you to one of the models and provide you with the instructions for building it. Make sure you compare your model with like models built by other groups. You are responsible for photographing all 4 generic models, not just the one your group built. And, five (colorless and colored) models of specific substances with cubic unit cells are also available: CsCl, NaTl, NaCl, and CaTiO3 (perovskite & fcc). These models will only be 2 Crystalline Solids Revised 2/8/16 identified by the letters A through E. It is up to you to correctly determine the identity of the solid represented by the model. Take pictures of models demonstrating the various properties of solids with cubic unit cells. Annotate the pictures with lines, labels, and descriptions that demonstrate your knowledge of the concepts listed below. Compliment these pictures with screenshots from the Odyssey modules showing the same orientations and/or concepts. Annotate the screenshots in the same way. You will be graded by how thoroughly and concisely you illustrate your knowledge of these concepts. (Note: Concisely means you do in with the least amount of pictures, writing, etc. needed.) • lattice points • types and locations of holes • type of packing & packing order • coordination numbers • type of unit cell • empirical formula • differences between generic model • density of solid • number of atoms per unit cell • which models are different • appearance a single unit cell representations of the same solid? If a specific substance cannot be found in Odyssey, use a combination of related solids in Odyssey (and/or figures found on the web) to compliment the pictures of the marble and stick models. You must have a picture from a model in lab and a screenshot from Odyssey for every model and you must annotate (put labels directly on the pictures and screenshots). Part B: Actual Solids & Models Nitinol (NiTi) A nickel-titanium alloy called "nitinol" is a memory metal that can be twisted or bent without causing crystal defects and returns to its original shape when heated. At high temperatures, nitinol atoms arrange in the orderly crystalline austenite form that resists distortion and “pings” when dropped. At low temperatures, nitinol arrange in the more disordered martensite form that can easily be twisted and bent and will “thud” when dropped. The difference in the unit cells of the two forms explains these observations: austenite has a regular body centered cubic structure while martensite has a related, but distorted structure. During the process of cooling, the high temperature austenite exists until the transition temperature (the point of solid-solid transition 3 Crystalline Solids Revised: 1/11/16 from austenite to martensite) is reached. If the alloy is bent while in the lower temperature martensite phase, gentle heating of the metal above the transition temperature (into the austenite phase) restores the original shape. If a new permanent shape is desired, the metal can be annealed (programmed) at very high temperatures (in a flame) to retain the "memory" of a new shape. Slightly altering the 1:1 ratio of Ni to Ti (i.e., Ni0.99Ti1.01) changes the transition temperature. Therefore, at room temperature one sample of nitinol can be in the martensite form, while another can be in the austenite form. Create your own procedure in lab that varies the temperature of nitinol rods to use their acoustic properties to find the transition temperature for the alloy. Also, using the flame of a candle, anneal a new shape into a thinner nitinol wire and devise a procedure that shows that nitinol has the memory of that shape. Inspect the models for the two forms of nitinol labeled 1 & 2. Which is austenite and which is martensite? Follow the same procedures in Part A to demonstrate your understanding of nitinol’s crystal structure. Explain your results with the rod and wire with respect to the crystal structures. The “1-2-3” Superconductor (YBa2Cu3O7) The "1-2-3" refers to the yttrium, barium, copper atom ratio. In this experiment the interaction between the cooled “1-2-3” superconductor and a samarium magnet will be observed. Understanding this requires the knowledge of band theory. Band theory is an electronic model for solids that explains conductivity by assuming a higher energy level exists above valence electrons called a "conduction band". (Figure 1). Figure 1. Band Theory for solids. 4 Crystalline Solids Revised: 1/11/16 The electrons in this band are not attached to (localized on) individual atoms, but are free to move (delocalized) throughout the entire solid. Electrons can always cross the small band gap of conductors (metals) with the input of ordinary thermal energy (room temp) and create a current. Increasing the metal’s temperature excites atoms causing electron scattering in the conduction band and increases resistance. Conversely, decreasing temperature in ordinary metals decreases resistance. The resistance, however, never does go to zero because electrons are scattered by defects in crystalline structure. However, at very low temperatures, some metals (or metal oxides) undergo a solid-solid transition and the resistance drops to zero, allowing electrical current to flow without hindrance. This remarkable phenomenon of superconductivity allows current to flow indefinitely because of a unique pairing of electrons – Cooper’s pairs. The combined momentum of Cooper pairs prevents electron scattering. Resistance drops to zero below some critical phase transition temperature (Tc). Above Tc, the Cooper pairs dissociate, superconductivity ceases, and the solid becomes normal conducting material. The Tc for YBa2Cu3O7 is 95 K. When the magnet is brought near the YBa2Cu3O7 superconductor cooled in N2(l) (77 K), the magnet’s magnetic field lines penetrate the superconductor inducing a current. This current creates an opposing magnetic field in the superconductor repelling the magnet and leading to its levitation. (Figure 2). The height at which the magnet is levitated reflects the tendency to minimize the total energy of the system: The internal energy of the superconductor increases as the magnet moves closer to the surface and the gravitational potential energy increases as the magnet moves further away from the surface. magnet perpendicular current induced in superconductor super conductor magnetic field induced in superconductor (perpendicular to current & antiparallel to magnet's field Figure 2. Induced current and magnetic field of the superconductor. 5 Crystalline Solids Revised 1/11/16 Superconductor 1. Check out a magnet from your TA. (Loss of magnet will result in a 5 point deduction.) Using plastic tweezers, place the larger superconductor pellet on a stack of pennies in the center of a cutoff Styrofoam cup. The pellet should be level with the top edge of the cup. (Scrape off loose material from the pellet with a spatula. If the pellet is broken, use the largest piece, flat side up.) Use plastic tweezers to place the smaller magnet on the superconductor. 2. Carefully pour N2(l) into the cup, covering the pennies and the bottom of the pellet. Touch the magnet gently with tweezers - it should spin. Take a video and picture. Allow the N2(l) to evaporate so the pellet and magnet warm back to room temperature. (To avoid frostbite, do not touch pellet or magnet until warmed.) 3. The unit cell of YBa2Cu3O7 is created by stacking 3 pervoskite unit cells. (Figure 3). Inspect the two models for the superconductor labeled 3 & 4. Which model is face centered cubic? Which is perovskite? Follow the same procedures in Part A to demonstrate your understanding of the superconductor’s crystal structure. Compare these models to the CaTiO3 models. Cu2+/Cu3+ Ba2+ O2Y3+ Figure 3. The unit cell of YBa2Cu3O7. 6