* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Stoichiometry Week 2
Survey
Document related concepts
Transcript
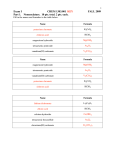
SHS Chemistry 2015-2016 Learning Target I will convert between moles, particles, and grams Warm-up How many atoms are in 0.25 moles of silver (Ag)? What is the mass of 0.25 moles of silver (Ag)? IP#3 DUE WEDNESDAY QUIZ #2 is THURSDAY On a clean sheet of paper perform the following calculations: Percent of water lost Molar mass of water Moles of water lost Molecules of water lost Write the conclusion. Bullet 3: actual mass of water is 14% Check data with another Bullet 4: Add how you would group if you feel like your change or improve the procedure data is not accurate BUBBLE GUM LAB Finish all calculations. Be sure you show your work and circle all final answers. POPCORN LAB On a clean sheet of paper perform the following calculations: Percent of water lost Molar mass of water Moles of water lost Molecules of water lost Check data with another group if you feel like your data is not accurate Write the conclusion. Bullet 1: field corn= corn sitting in a field Bullet 3: actual mass of water is 14% Bullet 4: Add how you would change or improve the procedure Using the below compounds, create your own conversion problems for another table to solve. Remember, you can convert between moles, particles, and grams, but be careful with your units! NaBr (NH4)2S PbSO4 Fe3(PO4)2 Ca(OH)2 Na3PO4 (NH4)2CO3 C6H12O6 Zn(C2H3O2)2 AgF Round 1: Clockwise Seat A: Molar Mass / Seat B: Molar Mass Conversion / Seat C: Percent Composition Seat D: Percent Composition Round 2: Counterclockwise Round 3: (7 5) (5 3) (31) (16) (24) (47) (62) Seat A: Percent Composition / Seat B: Molar Mass / Seat C: Molar Mass Conversion Seat D: Percent Composition Seat A: Percent Composition/ Seat B: Percent Composition / Seat C: Molar Mass Seat D: Molar Mass Conversion Round 4: Backwards from previous Seat A: Molar Mass Conversion/ Seat B: Percent Composition / Seat C: Percent Composition Seat D: Molar Mass Learning Target I will use multiple unit factors to convert between grams and particles Warm Up Calculate the number of molecules present in 353 grams of carbon tetrachloride. Show your work including all units. You will be weighing substances and subtracting the mass of the container to determine the mass of the substances themselves. You will need a calculator, something to write with, your moles islands, and a periodic table. Mass of Plastic Bottle: 9.4 grams Mass of Slender Glass Bottle: 39.1 grams Mass of Squarish Glass Bottle: 46.2 grams Learning Target Continue Mass / Mole/ Particle Conversions Warm Up Independent Practice #2 is due today! Quiz #2 Friday! Combustion Reaction 1CH4 + 2O2 1CO2 + 2H2O Write a problem to trade with your partner that involves converting between mass, moles, or particles (or all three). (2 minutes) Write the solution to your problem, including all the steps for solving it. (2 minutes) Trade and solve your partner’s problem. (3 minutes) Compare your methods for problem solving (1 minute) Learning Target Converting between mass and moles and particles Warm Up Check the independent practice KEY to make sure your answers are correct You have today to prepare for tomorrow’s quiz! Review your independent practice Work on the Mass-Mole-Particle handout. Check your answers with the key to make sure you are on the right track Learning Target I can determine the proper conversion factors to use and can convert between mass and particles of a single substance. Warm Up Prepare for quiz! You will need a periodic table and a calculator Balance this equation: __Na + __CuCl2 -> __Cu + __NaCl 2Na + CuCl2 -> Cu + 2NaCl If 256 grams of Copper (II) Chloride were reacted, how many particles were reacted? If 3.5 x 1024 particles of NaCl were produced, how many grams of NaCl were produced? If 5.3 x 1014 particles of Na were reacted, how many moles of Na were reacted? Take your time and show ALL YOUR WORK Take your time! Did you… Show all your work? Label every number with units and chemical formula? Cancel units? Provide a final answer with units and chemical formula? Due Tuesday 3/28 Quiz 3 Wednesday 3/29