* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Stoichiometry - HCC Learning Web
Survey
Document related concepts
Transcript
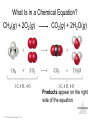
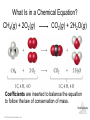
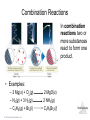
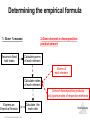
Lecture Presentation Chapter 3 Chemical Reactions and Reaction Stoichiometry © 2015 Pearson Education, Inc. James F. Kirby Quinnipiac University Hamden, CT Stoichiometry • The study of the mass relationships in chemistry • Based on the Law of Conservation of Mass (Antoine Lavoisier, 1789) “We may lay it down as an incontestable axiom that, in all the operations of art and nature, nothing is created; an equal amount of matter exists both before and after the experiment. Upon this principle, the whole art of performing chemical experiments depends.” —Antoine Lavoisier © 2015 Pearson Education, Inc. Chemical Equations Chemical equations are concise representations of chemical reactions. Stoichiometry © 2015 Pearson Education, Inc. What Is in a Chemical Equation? CH4(g) + 2O2(g) CO2(g) + 2H2O(g) Reactants appear on the left side of the equation. Stoichiometry © 2015 Pearson Education, Inc. What Is in a Chemical Equation? CH4(g) + 2O2(g) CO2(g) + 2H2O(g) Products appear on the right side of the equation. Stoichiometry © 2015 Pearson Education, Inc. What Is in a Chemical Equation? CH4(g) + 2O2(g) CO2(g) + 2H2O(g) The states of the reactants and products are written in parentheses to the right of each compound. (g) = gas; (l) = liquid; (s) = solid; (aq) = in aqueous solution © 2015 Pearson Education, Inc. Stoichiometry What Is in a Chemical Equation? CH4(g) + 2O2(g) CO2(g) + 2H2O(g) Coefficients are inserted to balance the equation to follow the law of conservation of mass. Stoichiometry © 2015 Pearson Education, Inc. Why Do We Add Coefficients Instead of Changing Subscripts to Balance? • Hydrogen and oxygen can make water OR hydrogen peroxide: 2 H2(g) + O2(g) → 2 H2O(l) H2(g) + O2(g) → H2O2(l) Stoichiometry © 2015 Pearson Education, Inc. Three Types of Reactions • Combination reactions • Decomposition reactions • Combustion reactions Stoichiometry © 2015 Pearson Education, Inc. Combination Reactions • In combination reactions two or more substances react to form one product. • Examples: – 2 Mg(s) + O2(g) – N2(g) + 3 H2(g) – C3H6(g) + Br2(l) © 2015 Pearson Education, Inc. 2 MgO(s) 2 NH3(g) C3H6Br2(l) Stoichiometry Decomposition Reactions • In a decomposition reaction one substance breaks down into two or more substances. • Examples: – CaCO3(s) – 2 KClO3(s) – 2 NaN3(s) © 2015 Pearson Education, Inc. CaO(s) + CO2(g) 2 KCl(s) + O2(g) 2 Na(s) + 3 N2(g) Stoichiometry Combustion Reactions • Combustion reactions are generally rapid reactions that produce a flame. • Combustion reactions most often involve oxygen in the air as a reactant. • Examples: – CH4(g) + 2 O2(g) – C3H8(g) + 5 O2(g) © 2015 Pearson Education, Inc. CO2(g) + 2 H2O(g) 3 CO2(g) + 4 H2O(g) Stoichiometry Formula Weight (FW) • A formula weight is the sum of the atomic weights for the atoms in a chemical formula. • This is the quantitative significance of a formula. • The formula weight of calcium chloride, CaCl2, would be Ca: 1(40.08 amu) + Cl: 2(35.453 amu) 110.99 amu Stoichiometry © 2015 Pearson Education, Inc. Molecular Weight (MW) • A molecular weight is the sum of the atomic weights of the atoms in a molecule. • For the molecule ethane, C2H6, the molecular weight would be C: 2(12.011 amu) + H: 6(1.00794 amu) 30.070 amu Stoichiometry © 2015 Pearson Education, Inc. Ionic Compounds and Formulas • Remember, ionic compounds exist with a three-dimensional order of ions. There is no simple group of atoms to call a molecule. • As such, ionic compounds use empirical formulas and formula weights (not molecular weights). Stoichiometry © 2015 Pearson Education, Inc. Percent Composition One can find the percentage of the mass of a compound that comes from each of the elements in the compound by using this equation: (number of atoms)(atomic weight) % Element = (FW of the compound) × 100 Stoichiometry © 2015 Pearson Education, Inc. Percent Composition So the percentage of carbon in ethane is (2)(12.011 amu) %C = = (30.070 amu) 24.022 amu 30.070 amu × 100 = 79.887% Stoichiometry © 2015 Pearson Education, Inc. Counting numbers and the mole Stoichiometry © 2015 Pearson Education, Inc. Avogadro’s Number • In a lab, we cannot work with individual molecules. They are too small. • 6.02 × 1023 atoms or molecules is an amount that brings us to lab size. It is ONE MOLE. • One mole of 12C has a mass of 12.000 g. © 2015 Pearson Education, Inc. Stoichiometry Molar Mass • A molar mass is the mass of 1 mol of a substance (i.e., g/mol). • The molar mass of an element is the atomic weight for the element from the periodic table. If it is diatomic, it is twice that atomic weight. • The formula weight (in amu’s) will be the same number as the molar mass (in g/mol). © 2015 Pearson Education, Inc. Stoichiometry Using Moles Moles provide a bridge from the molecular scale to the real-world scale. Stoichiometry © 2015 Pearson Education, Inc. Chemical arithmetic…… ….…….how many conversions do I need to perform? ……....which conversions do I need to perform? Stoichiometry © 2015 Pearson Education, Inc. Mole Relationships • One mole of atoms, ions, or molecules contains Avogadro’s number of those particles. • One mole of molecules or formula units contains Avogadro’s number times the number of atoms or ions of each element in the compound. Stoichiometry © 2015 Pearson Education, Inc. *Which calculations do or do not involve a single conversion? Answer YES or NO (1 step). consider 15.02 g H2O as an example 1. Moles of water Y 2. Moles of oxygen N 3. Number of Atoms of hydrogen N 4. Mass of hydrogen Y 5. Number of atoms of oxygen N Stoichiometry © 2015 Pearson Education, Inc. Conversions (cont’d) Consider 10.02 g Na3PO4 Mass (Na3PO4) Mol (Na3PO4) Mass (PO43-) 1 step Mass (Sodium) Mol (Sodium) Mol (phosphate) multistep Ions (sodium) Stoichiometry Atoms (oxygen) © 2015 Pearson Education, Inc. Calculate the missing quantities in the table below Compound Mass of Compound Moles of… Atoms of… Mg3N2 Mg (1.52 mol) N CuSO4.5H2O H O (3.75 X 1019 atoms) H2S2O7 S (0.125 mol) O Iron(III) chloride 13.75 g Fe Cl Stoichiometry © 2015 Pearson Education, Inc. Empirical Formula (Simplest Formula) The formula of a substance written with the smallest integer subscripts. For example: The empirical formula for N2O4 is NO2. The empirical formula for H2O2 is HO. © 2015 Pearson Education, Inc. Stoichiometry Copyright © Cengage Learning. All rights 3 | 27 Determining Empirical Formulas *Finding the moles is the critical step One can determine the empirical formula from the percent composition by following these three steps. Stoichiometry © 2015 Pearson Education, Inc. Determining the empirical formula 1. Given % masses Assume a fixed total mass 2.Given element or decomposition product amount Calculate grams of each element Grams of each element Calculate moles of each element Convert decomposition products into grams/moles of respective elements Express as Empirical formula © 2015 Pearson Education, Inc. Calculate the mole ratio Stoichiometry Determining the Empirical Formula Beginning with percent composition: • Assume exactly 100 g so percentages convert directly to grams. • Convert grams to moles for each element. • Manipulate the resulting mole ratios to Stoichiometry obtain whole numbers. Copyright © Cengage © 2015 Pearson Education, Inc. Learning. All rights 3 | 30 Manipulating the ratios: Divide each mole amount by the smallest mole amount. If the result is not a whole number: Multiply each mole amount by a factor to make whole numbers. For example: © 2015 Pearson Education, Inc. Stoichiometry Copyright © Cengage Learning. All rights 3 | 31 Benzene is composed of 92.3% carbon and 7.7% hydrogen. What is the empirical formula of benzene? 1 mol C 92.3 g C 7.685 mol C 12.01 g C 1 mol H 7.7 g H 7.64 mol H 1.008 g H 7.685 1 7.64 7.64 1 7.64 Empirical formula: CH © 2015 Pearson Education, Inc. Stoichiometry Copyright © Cengage Learning. All rights 3 | 32 Molecular Formula A formula for a molecule in which the subscripts are whole-number multiples of the subscripts in the empirical formula. © 2015 Pearson Education, Inc. Stoichiometry Copyright © Cengage Learning. All rights 3 | 33 To determine the molecular formula: • Compute the empirical formula weight. • Find the ratio of the molecular weight to the empirical formula weight. molecular weight n empirical formula weight • Multiply each subscript of the empirical formula by n. © 2015 Pearson Education, Inc. Stoichiometry Copyright © Cengage Learning. All rights 3 | 34 Benzene has the empirical formula CH. Its molecular weight is 78.1 amu. What is its molecular formula? Empirical formula weight 13.02 amu 78.1 6 13.02 Molecular formula C6H6 © 2015 Pearson Education, Inc. Stoichiometry Copyright © Cengage Learning. All rights 3 | 35 Sodium pyrophosphate is used in detergent preparations. It is composed of 34.5% Na, 23.3% P, and 42.1% O. What is its empirical formula? 1 mol Na 34.5 g Na 1.501 mol Na 22.99 g Na 1 mol P 23.3 g P 0.7523 mol P 30.97 g P 1 mol O 42.1 g O 2.631 mol O 16.00 g O © 2015 Pearson Education, Inc. Stoichiometry Copyright © Cengage Learning. All rights 3 | 36 1.501 mol Na 1.501 2.00 2 = 4 0.7523 0.7523 0.7523 mol P 1.00 2 = 2 0.7523 2.631 mol O 2.631 3.50 2 = 7 0.7523 Empirical formula Stoichiometry Na4Copyright P2O7 © Cengage © 2015 Pearson Education, Inc. Learning. All rights 3 | 37 Hexamethylene is one of the materials used to produce a type of nylon. It is composed of 62.1% C, 13.8% H, and 24.1% N. Its molecular weight is 116 amu. What is its molecular formula? 1 mol C 62.1 g C 5.171 mol C 12.01 g C 1 mol H 13.8 g H 13.69 mol H 1.008 g H 1 mol N 24.1 g N 1.720 mol H 14.01 g N © 2015 Pearson Education, Inc. Stoichiometry Copyright © Cengage Learning. All rights 3 | 38 5.171 mol C 13.69 mol H 1.720 mol H 5.171 3 1.720 13.69 8 1.720 1.720 1 1.720 Empirical formula C3H8N © 2015 Pearson Education, Inc. Stoichiometry Copyright © Cengage Learning. All rights 3 | 39 The empirical formula is C3H8N. Find the empirical formula weight: 3(12.01) + 8(1.008) + 1(14.01) = 58.104 amu 116 n 58.10 2 Molecular formula: C6H16N2 © 2015 Pearson Education, Inc. Stoichiometry Copyright © Cengage Learning. All rights 3 | 40 Stoichiometry The calculation of the quantities of reactants and products involved in a chemical reaction. Interpreting a Chemical Equation The coefficients of the balanced chemical equation may be interpreted in terms of either (1) numbers of molecules (or ions or formula units) or (2) numbers of moles,Stoichiometry Copyright © Cengage depending on your needs. Learning. All rights 3 | 41 © 2015 Pearson Education, Inc. To find the amount of B (one reactant or product) given the amount of A (another reactant or product): 1. Convert grams of A to moles of A Using the molar mass of A 2. Convert moles of A to moles of B Using the coefficients of the balanced chemical equation 3. Convert moles of B to grams of B Using the molar mass of B © 2015 Pearson Education, Inc. Stoichiometry Copyright © Cengage Learning. All rights 3 | 42 Propane, C3H8, is normally a gas, but it is sold as a fuel compressed as a liquid in steel cylinders. The gas burns according to the following equation: C3H8(g) + 5O2(g) 3CO2(g) + 4H2O(g) How many grams of CO2 are produced when 20.0 g of propane is burned? © 2015 Pearson Education, Inc. Stoichiometry Copyright © Cengage Learning. All rights 3 | 43 Molar masses C3H8: 3(12.01) + 8(1.008) = 44.094 g CO2: 1(12.01) + 2(16.00) = 44.01 g 1 mol C 3H8 3 mol CO2 44.01 g CO2 20.0 g C 3H8 44.094 g C 3H8 1 mol C 3H8 1 mol CO2 59.8856987 3 g CO 2 59.9 g CO2 (3 significant figures) Stoichiometry 3 | 44 © 2015 Pearson Education, Inc. Propane, C3H8, is normally a gas, but it is sold as a fuel compressed as a liquid in steel cylinders. The gas burns according to the following equation: C3H8(g) + 5O2(g) 3CO2(g) + 4H2O(g) How many grams of O2 are required to burn 20.0 g of propane? © 2015 Pearson Education, Inc. Stoichiometry Copyright © Cengage Learning. All rights 3 | 45 Molar masses: O2 2(16.00) = 32.00 g C3H8 3(12.01) + 8(1.008) = 44.094 g 1 mol C 3H8 5 mol O 2 32.00 g O 2 20.0 g C 3H8 44.094 g C 3H8 1 mol C 3H8 1 mol O 2 72.5722320 5 g O 2 72.6 g O2 (3 significant figures) © 2015 Pearson Education, Inc. Stoichiometry Copyright © Cengage Learning. All rights 3 | 46 Limiting Reactant The reactant that is entirely consumed when a reaction goes to completion. Once one reactant has been completely consumed, the reaction stops. Any problem giving the starting amount for more than one reactant is a limiting reactant problem. © 2015 Pearson Education, Inc. Stoichiometry Copyright © Cengage Learning. All rights 3 | 47 All amounts produced and reacted are determined by the limiting reactant. How can we determine the limiting reactant? 1.Use each given amount to calculate the amount of product produced. 2.The limiting reactant will produce the lesser or least amount of product. © 2015 Pearson Education, Inc. Stoichiometry Copyright © Cengage Learning. All rights 3 | 48 Magnesium metal is used to prepare zirconium metal, which is used to make the container for nuclear fuel (the nuclear fuel rods): ZrCl4(g) + 2Mg(s) 2MgCl2(s) + Zr(s) How many moles of zirconium metal can be produced from a reaction mixture containing 0.20 mol ZrCl4 and 0.50 mol Mg? © 2015 Pearson Education, Inc. Stoichiometry Copyright © Cengage Learning. All rights 3 | 49 1 mol Zr 0.20 mol ZrCl 4 0.20 mol Zr 1 mol ZrCl 4 1 mol Zr 0.50 mol Mg 0.25 mol Zr 2 mol Mg Since ZrCl4 gives the lesser amount of Zr, ZrCl4 is the limiting reactant. 0.20 mol Zr will be produced. Stoichiometry 3 | 50 © 2015 Pearson Education, Inc. Urea, CH4N2O, is used as a nitrogen fertilizer. It is manufactured from ammonia and carbon dioxide at high pressure and high temperature: 2NH3 + CO2(g) CH4N2O + H2O In a laboratory experiment, 10.0 g NH3 and 10.0 g CO2 were added to a reaction vessel. What is the maximum quantity (in grams) of urea that can be obtained? How many grams of the excess reactant Stoichiometry are left at the end of the reactions? Copyright © Cengage © 2015 Pearson Education, Inc. Learning. All rights 3 | 51 Molar masses NH3 1(14.01) + 3(1.008) = 17.02 g CO2 1(12.01) + 2(16.00) = 44.01 g CH4N2O 1(12.01) + 4(1.008) + 2(14.01) + 1(16.00) = 60.06 g 1 mol NH 3 1 mol CH4 N 2 O 60.06 g CH4 N 2 O 10.0 g NH 3 17.024 g NH 3 2 mol NH 3 1 mol CH4 N 2 O 17.6 g CH4 N 2 O 1 mol CO 2 1 mol CH4 N2 O 60.06 g CH4 N 2 O 10.0 g CO 2 44.01 g CO 2 1 mol CO 2 1 mol CH4 N 2 O 13.6 g CH4 N 2 O CO2 is the limiting reactant since it gives the lesser amount of CH4N2O. Stoichiometry 3 | 52 13.6 g CH4N2O will be produced. © 2015 Pearson Education, Inc. To find the excess NH3, we need to find how much NH3 reacted. We use the limiting reactant as our starting point. 1 mol CO2 2 mol NH 3 17.02 g NH 3 10.0 g CO2 44.01 g CO2 1 mol CO2 1 mol NH 3 7.73460577 1 g NH 3 7.73 g NH 3 reacted Now subtract the amount reacted from the at start starting amount: 10.0 −7.73 reacted 2.27 g remains 2.3 g NH3 is left unreacted. (1 decimal place) © 2015 Pearson Education, Inc. Stoichiometry 3 | 53 Theoretical/Expected Yield Theorerical yield is the maximum amount of product that can be obtained by a reaction from given amounts of reactants. This is a calculated amount based on a balanced chemical equation. © 2015 Pearson Education, Inc. Stoichiometry Copyright © Cengage Learning. All rights 3 | 54 Actual/Experimental Yield The amount of product that is actually obtained. This is a measured amount. Percentage Yield actual yield percentage yield 100% theoretical yield © 2015 Pearson Education, Inc. Stoichiometry Copyright © Cengage Learning. All rights 3 | 55 2NH3 + CO2(g) CH4N2O + H2O When 10.0 g NH3 and 10.0 g CO2 are added to a reaction vessel, the limiting reactant is CO2. The theoretical yield is 13.6 of urea. When this reaction was carried out, 9.3 g of urea was obtained. What is the percent yield? 3 | 56 © 2015 Pearson Education, Inc. Stoichiometry Copyright © Cengage Learning. All rights Theoretical yield = 13.6 g Actual yield = 9.3 g 9.3 g Percent yield = 100% 13.6 g = 68% yield (2 significant figures) © 2015 Pearson Education, Inc. Stoichiometry Copyright © Cengage Learning. All rights 3 | 57 Silver chloride can be made according to the following chemical equation NaCl(aq) + AgNO3(aq) → NaNO3(aq) + AgCl(s) When solutions containing 0.351 g AgNO3 and 0.250 g NaCl were mixed, the amount of AgCl precipitated was 0.250 g. Calculate the percent yield of AgCl. Stoichiometry © 2015 Pearson Education, Inc.