* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Covalent Bonding and Molecular Structures
Nuclear physics wikipedia , lookup
History of subatomic physics wikipedia , lookup
Hydrogen atom wikipedia , lookup
Atomic nucleus wikipedia , lookup
Atomic theory wikipedia , lookup
Resonance (chemistry) wikipedia , lookup
Molecular orbital wikipedia , lookup
Bent's rule wikipedia , lookup
Hypervalent molecule wikipedia , lookup
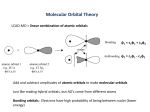
Chapters 8 and 9 Covalent Bonding and Molecular Structures Objectives You will be able to: 1. Write a description of the formation of the covalent bond between two hydrogen atoms to form a hydrogen molecule. Your description should include mention of overlapping atomic orbitals to form molecular orbitals. It should also include rough pictures of the atomic orbitals and the molecular orbitals. 2. Write a description of the assumptions made in the Linear Combination of Atomic Orbitals (LCAO) approximation for the molecular orbital approach to covalent bonding. 3. Write an explanation for why bonding molecular orbitals are more stable and why antibonding molecular orbitals are less stable than the separate atomic orbitals that form them. 4. Write a description of how the σ1s, σ*1s, σ2s, σ*2s, σ2p, σ*2p, π*2p, and π*2p are formed from the 1s, 2s and 2p atomic orbitals. 5. Draw sketches of the σ1s, σ*1s, σ2p, σ*2p, π2p, and π*2p molecular orbitals. 6. Write an explanation for why the σ2p molecular orbital of O2 is lower energy than the π2p molecular orbitals and why the σ*2p is higher energy than the π*2p. 7. Draw molecular orbital diagrams for H2, O2, F2, and Ne2. 8. From a molecular orbital diagram of a molecule, predict its bond order. 9. From a molecular orbital diagram for a molecule, predict whether it is paramagnetic or diamagnetic. 10.Draw the molecular orbital diagrams for CO and NO and use them to predict the bond order of each and to explain why each is relatively stable despite their strange Lewis Structures. 11.Describe the advantages and disadvantages of scientific models. 12.Write a list of the basic assumptions of the valence‑bond model of covalent bonding. 13.Write or identify the number of valence electrons for each of the Representative elements. 14.With reference to the valence bond model, describe how the covalent bond forms between fluorine atoms in F2. 15.Write or identify a description of the information given in a Lewis structure. 16.With reference to the valence bond model, describe how the covalent bond forms between hydrogen atoms in H2. 17.Explain how the carbon atom in CH4 is able to form four equivalent covalent bonds to hydrogen atoms with bond angles of 109.5°. 18.Describe (1) how sp3 hybrid orbitals form, (2) their shapes, and (3) how they are arranged in space. 19.Write or identify a description of the tetrahedral electron group geometry. 157 158 Chapter 8 & 9 Covalent Bonding and Molecular Structures 20.For ethene (ethylene) molecules, C2H4: (1) explain how the carbon atoms are able to form four equivalent covalent bonds to hydrogen atoms, (2) explain why all the bond angles are about 120°, (3) with reference to the valence bond model, explain how the double bond between carbon atoms forms, and (4) explain why one bond in the double bond is weaker than the other. 21.Describe (1) how sp2 hybrid orbitals form, (2) their shapes, and (3) how they are arranged in space. 22.Write or identify a description of the trigonal planar electron group geometry. 23.For ethyne (acetylene) molecules, C2H2: (1) explain how the carbon atoms are able to form two equivalent covalent bonds to hydrogen atoms, (2) explain why the bond angles are 180°, (3) with reference to the valence bond model, explain how the triple bond between carbon atoms forms, and (4) explain why two bonds in the triple bond are weaker than the other one. 24.Describe (1) how sp hybrid orbitals form, (2) their shapes, and (3) how they are arranged in space. 25.Write or identify a description of the linear electron group geometry. 26.Explain how the nitrogen atom in NH3 is able to form three equivalent covalent bonds to hydrogen atoms with bond angles of 107°. 27.Write or identify a description of the trigonal pyramid molecular geometry. 28.Explain how the nitrogen atom in NH4+ is able to form four equivalent covalent bonds to hydrogen atoms with bond angles of 109.5°. 29.Explain how the oxygen atom in H2O is able to form two equivalent covalent bonds to hydrogen atoms with bond angles of 105°. 30.Write or identify a description of the bent molecular geometry. 31.With reference to the valence bond model, explain how the triple bond forms in the carbon monoxide, CO, molecule. 32.Explain how the boron atom in BF3 is able to form three equivalent covalent bonds to fluorine atoms with bond angles of 120°. 33.Explain how the phosphorus atom in PF5 is able to form five equivalent covalent bonds to fluorine atoms with bond angles of 90°, 120°, and 180°. 34.Write or identify a description of the trigonal bipyramid electron group geometry. 35.Explain how the sulfur atom in SF4 is able to form four covalent bonds to fluorine atoms with bond angles of 90°, 120°, and 180°. 36.Write or identify a description of the see-saw molecular geometry. 37.Explain how the iodine atom in IF3 is able to form three covalent bonds to fluorine atoms with bond angles of 90° and 180°. 38.Write or identify a description of the T-shaped molecular geometry. 39.Explain how the xenon atom in XeF2 is able to form two covalent bonds to fluorine atoms with a bond angle of 180°. 40.Write or identify a description of the linear molecular geometry. 41.Explain how the sulfur atom in SF6 is able to form six equivalent covalent bonds to fluorine atoms with bond angles of 90° and 180°. 42.Write or identify a description of the octahedral electron geometry. 43.Explain how the iodine atom in IF5 is able to form five equivalent covalent bonds to fluorine atoms with bond angles of 90° and 180°. 44.Write or identify a description of the square pyramid molecular group geometry. 159 45.Explain how the xenon atom in XeF4 is able to form four covalent bonds to fluorine atoms with bond angles of 90° and 180°. 46.Write or identify a description of the square planar molecular geometry. 47.Write an explanation for why the elements listed on the Table 8,9.3 can have each of the bonding patterns listed. 48.Given a Lewis structure or enough information to get one, do the following: (1) list the hybridization for each atom in the structure, (2) write a description of the formation of each bond in terms of the overlap of atomic orbitals, (3) describe the electron group geometry around each atom that has two or more atoms attached to it, (4) draw the geometric sketch of the molecule, including bond angles, and (5) describe the molecular geometry around each atom that has two or more atoms attached to it. 49.Given a Lewis structure or enough information to get one, determine the formal charge for each atom in the structure. 50.Given a formula for a molecule or polyatomic ion, draw a reasonable Lewis structure that corresponds to the formula. 51.Given a Lewis structure or enough information to get one, do the following: (1) identify whether it is best described as having resonance or not, (2) if can be seen as having resonance, draw the Lewis Structures for all of the reasonable resonance forms, and (3) draw the resonance hybrid for the formula. 52.Draw or identify cis and trans isomers for molecules with carbon-carbon double bonds. 53. Given a structure for a triglyceride, identify it as saturated or unsaturated, and identify whether it is more likely to be solid or liquid at room temperature. 54. Given a formula for a molecular compound, predict whether or not it represents a polar molecule. 55.Write a description of how London forces come to be. 56.Write an explanation for why larger molecules have stronger London forces than smaller molecules. 57.Write a description of how London forces play a part in the attractions between polar molecules. 58.Given a formula for a pure substance, identify which of the following types of substances it represents: ionic compound, metallic substance, polar molecular compound with hydrogen bonds, polar molecular substance without hydrogen bonds, or nonpolar molecular substance. 59.Identify carbon in the diamond form, C(dia), and silicon dioxide, SiO2 as examples of network crystals. 60.Given a formula for a pure substance, identify which of the following attractions is the predominant force holding its particles in the liquid and solid state: ionic bonds, metallic bonds, covalent bonds, dipole-dipole attractions, hydrogen bonds, or London forces. 61.Given chemical formulas for two substances that are about the same size but have different types of attractions between the particles, predict which substance would have the strongest attractions between the particles in the liquid and solid state. 160 Chapter 8 & 9 Covalent Bonding and Molecular Structures 62.Given chemical formulas for two substances with the same type of attraction between the particles but different sizes, predict which substance would have the stronger attractions between the particles in the liquid and solid state. 63. Given descriptions of two compounds that differ in the percentages of their structures that are polar and nonpolar (hydrophilic and hydrophobic), predict their relative solubilities in water and in hexane. 64.Explain why amphetamine can pass through the blood brain barrier more easily than epinephrine. 65.Explain why methamphetamine is often converted to methamphetamine hydrochloride. 66.Convert between the definition and the term for the following words or phrases. Chapters 8 and 9 Glossary Molecular orbital A volume that contains a high percentage of the electron charge for an electron in a molecule or a volume within which an electron in a molecule has a high probability of being found. Bonding molecular orbital Formed from in-phase interaction of two atomic orbitals. This leads to an increase in negative charge between two nuclei where the atomic orbitals overlap and leads to more +/- attraction between the negative charge generated by the electrons and the nuclei. Antibonding molecular orbital Formed from out-of-phase interaction of two atomic orbitals. This leads to a decrease in negative charge between two nuclei where the atomic orbitals overlap and leads to less +/- attraction between the negative charge generated by the electrons and the nuclei. Valence bond model The model for covalent bonding that includes the assumptions that (1) only the highest-energy (valence) electrons participate in bonding, (2) covalent bonds arise due to the overlap of atomic orbitals on adjacent atoms, forming molecular orbitals, and (3) covalent bonds often form to pair unpaired electrons. Valence electrons The highest‑energy s and p electrons for an atom. The electrons that are most important in the formation of chemical bonds. The number of valence electrons for the atoms of an element is equal to the element’s A-group number on the periodic table. Electron-dot symbol A representation of an atom that consists of its elemental symbol surrounded by dots representing its valence electrons. Lone pair Two electrons that are not involved in the covalent bonds between atoms but are important for explaining the arrangement of atoms in molecules. They are represented by pairs of dots in Lewis structures. Lewis structure A representation of a molecule that consists of the elemental symbol for each atom in the molecule, lines to show covalent bonds, and pairs of dots to indicate lone pairs. Hybrid orbital An atomic orbital formed from a blend of atomic orbitals, e.g. four sp3 hybrid orbitals are formed from the blend of one s orbital and three p orbitals. Bond angle The angle formed by straight lines (representing bonds) connecting the nuclei of three adjacent atoms. 161 Electron group geometry A description of the arrangement of all the electron groups around a central atom in a molecule or polyatomic ion, including the lone pairs. Molecular geometry The description of the arrangement of all the atoms around a central atom in a molecule or polyatomic ion. This description does not consider lone pairs. Isomers Compounds that have the same molecular formula but different molecular structures. Formal charge A tool for evaluating Lewis structures, calculated from the formula: A-group number minus number of bonds minus number of electrons in lone pairs. Resonance structures Two or more Lewis structures for a single molecule or polyatomic ion that differ in the positions of lone pairs and multiple bonds but not in the positions of the atoms in the structure. It is as if the molecule or ion were able to shift from one of these structures to another by shifting pairs of electrons from one position to another. Resonance The hypothetical switching from one resonance structure to another. Resonance hybrid A structure that represents the average of the resonance structures for a molecule or polyatomic ion. Delocalized electrons Electrons that are shared among three or more atoms. Delocalized pi system A system of overlapping p orbitals used to describe the bonding in resonance hybrids. Cis isomer A structure that has like groups on different carbons (which are linked by a double bond) and on the same side of the double bond. Trans isomer A structure that has like groups on different carbons (which are linked by a double bond) and on different sides of the double bond. Triglyceride A compound with three hydrocarbon groups attached to a three carbon backbone by ester functional groups. Saturated fat A triglyceride with single bonds between all of the carbon atoms. Unsaturated fat A triglyceride that has one or more carbon carbon double bonds. Hydrogenation A process by which hydrogen is added to an unsaturated triglyceride to convert double bonds to single bonds. This can be done by combining the unsaturated triglyceride with hydrogen gas and a platinum catalyst. Noncovalent interaction All forces of attraction between particles other than covalent, ionic, or metallic bonds. Intermolecular forces Attractions between molecules. Dipole-dipole attraction The intermolecular attraction between the partial negative end of one polar molecule and the partial positive end of another polar molecule. Hydrogen bond The intermolecular attraction between a nitrogen, oxygen, or fluorine atom of one molecule and a hydrogen atom bonded to a nitrogen, oxygen, or fluorine atom in another molecule. London dispersion forces, London forces, or dispersion forces The attractions produced between molecules by instantaneous and induced dipoles. Hydrophilic (“water loving”): A polar molecule or ion (or a portion of a molecule or polyatomic ion) that is attracted to water. Hydrophobic (“water fearing”): A nonpolar molecule (or a portion of a molecule or polyatomic ion) that is not expected to mix with water. Lipid bilayer Cell membrane. 162 Chapter 8 & 9 Covalent Bonding and Molecular Structures Molecular Orbital Theory The goal of molecular orbital theory is to describe molecules in a similar way to how we describe atoms, that is, in terms of orbitals, orbital diagrams, and electron configurations. For example, to give you a glimpse at where we are headed, the following are orbital diagrams for O2 and O. ∗ σ2p π ∗2p π 2p π∗2p π 2p σ2p ∗ σ2s σ2s ∗ σ1s σ1s O2 2s 1s 2p O Each line in the molecular orbital diagram represents a molecular orbital, which is the volume within which a high percentage of the negative charge generated by the electron is found. The molecular orbital volume encompasses the whole molecule. We assume that the electrons would fill the molecular orbitals of molecules like electrons fill atomic orbitals in atoms. • The molecular orbitals are filled in a way that yields the lowest potential energy for the molecule. • The maximum number of electrons in each molecular orbital is two. (We follow the Pauli exclusion principle.) • Orbitals of equal energy are half filled with parallel spin before they begin to pair up. (We follow Hund’s Rule.) Before we continue with a description of a model used to generate molecular orbital diagrams, let’s get a review of light and electron waves and how two waves can interact. The wave description of light describes the effect that the light has on the space around it. This effect is to generate an oscillating electric and magnetic fields. These fields can vary in intensity, which is reflected in varying brightness of light. 163 Source Wavelength, λ, the distance between two peaks Magnetic field, perpendicular to electric field Electric field, perpendicular to magnetic field Figure 8,9.1 Electromagnetic Waves Radiant energy The wave description of the electron describes the variation in the intensity of negative charge generated by the electron. Nucleus, about 0.00001 the diameter of the atom The negative charge is most intense at the nucleus and decreases in intensity with distance outward. Figure 8,9.2 1s Orbital Light waves can interact in-phase, which leads to an increase in the intensity of the light (brighter) and out-of-phase, which leads to a decrease in the intensity of the light (less bright). Figure 8,9.3 Wave Interference In-phase Brighter Dark Out-of-phase Electron waves can also interact in-phase and out-of-phase. In-phase interaction leads to an increase in the intensity of the negative charge. Out-of-phase interaction leads to a decrease in the intensity of the negative charge. One common approximation that allows us to generate molecular orbital diagrams for some small diatomic molecules is called the Linear Combination of Atomic Orbitals (LCAO) approach. The following assumptions lie at the core of this model. • Molecular orbitals are formed from the overlap of atomic orbitals. • Only atomic orbitals of about the same energy interact to a significant degree. • When two atomic orbitals overlap, they interact in two extreme ways to form two molecular orbitals, a bonding molecular orbital and an antibonding molecular orbital. 164 Chapter 8 & 9 Covalent Bonding and Molecular Structures For example, our model assumes that two 1s atomic orbitals can overlap in two extreme ways to form two molecular orbitals. One of the ways the atomic orbitals interact is in-phase, which leads to wave enhancement similar to the enhancement of two in-phase light waves. Where the atomic orbitals overlap, the in‑phase interaction leads to an increase in the intensity of the negative charge in the region where they overlap. This creates an increase in negative charge between the nuclei and an increase in the plus‑minus attraction between the electron charge and the nuclei for the atoms in the bond. The greater attraction leads to greater stability and lower potential energy. Because electrons in the molecular orbital are lower potential energy than in separate atomic orbitals, energy would be required to shift the electrons back into the 1s orbitals of separate atoms. This keeps the atoms together in the molecule, so we call this orbital a bonding molecular orbital. The molecular orbital formed is symmetrical about the axis of the bond. Symmetrical molecular orbitals are called sigma, σ, molecular orbitals. The symbol σ1s is used to describe the bonding molecular orbital formed from two 1s atomic orbitals. The second way that two atomic orbitals interact is out‑of‑phase. Where the atomic orbitals overlap, the out-of‑phase interaction leads to a decrease in the intensity of the negative charge. This creates a decrease in negative charge between the nuclei and a decrease in the plus-minus attraction between the electron charge and the nuclei for the atoms in the bond. The lesser attraction leads to lower stability and higher potential energy. The electrons are more stable in the 1s atomic orbitals of separate atoms, so electrons in this type of molecular orbital destabilize the bond between atoms. We call this type of molecular orbital an antibonding molecular orbital. The molecular orbital formed is symmetrical about the axis of the bond, so it is a sigma molecular orbital with a symbol of σ*1s. The asterisk indicates an antibonding molecular orbital. The following diagram shows the bonding and antibonding molecular orbitals formed from the interaction of two 1s atomic orbitals. 165 Decreased negative charge between the nuclei leads to decreased attractions between the negative charge from the electrons and the positively charged nuclei. This makes the sigma 1s anitbonding molecular orbital higher potential energy than the separate 1s atomic orbitals. σ∗1s out-ofphase nuclei in-phase 1s 1s σ1s Increased negative charge between the nuclei leads to increased attractions between the negative charge from the electrons and the positively charged nuclei. This makes the sigma 1s bonding molecular orbital lower potential energy than the separate 1s atomic orbitals. ∗ σ1s 1s 1s σ1s Figure 8,9.4 Formation of Sigma 1s Molecular Orbitals When two larger atoms combine to form a diatomic molecules (such as O2, F2, or Ne2), more atomic orbitals interact. The LCAO approximation assumes that only the atomic orbitals of about the same energy interact. For O2, F2, and Ne2, the orbital energies are different enough in energy so only orbitals of the same energy interact to a significant degree. Like for hydrogen, the 1s from one atom overlaps the 1s from the other atom to form a σ1s bonding molecular orbital and a σ*1s antibonding molecular orbital. The shapes would be similar to those formed from the 1s orbitals for hydrogen. The 2s atomic orbital from one atom overlaps the 2s atomic orbital from the other atom to form a σ2s bonding molecular orbital and a σ*2s antibonding molecular orbital. The shapes of these molecular orbitals would be similar to those for the σ1s and σ*1s molecular orbitals. Both σ2s and σ*2s molecular orbitals are higher energy and larger than the σ1s and σ*1s molecular orbitals. The p atomic orbitals of the two atoms can interact in two different ways, parallel or end-on. The molecular orbitals are different for each type of interaction. The end-on interaction between two 2px atomic orbitals yields sigma molecular orbitals, which are symmetrical about the axis of the bond. 166 Chapter 8 & 9 Covalent Bonding and Molecular Structures Figure 8,9.5 Formation of Sigma 2p Molecular Orbitals σ*2p out-ofphase 2px 2px nuclei in-phase σ2p The two 2py atomic orbitals overlap in parallel and form two pi molecular orbitals. Pi molecular orbitals are asymmetrical about the axis of the bond. Figure 8,9.6 Formation of Pi 2p Molecular Orbitals π 2p * out-ofphase nuclei in-phase 2py 2py π 2p There is less overlap between parallel p orbitals than between two p orbitals overlapping end-on. The 2pz-2pz overlap generates another pair of π2p and π*2p molecular orbitals. The 2pz-2pz overlap is similar to the 2py-2py overlap. To visualize this overlap, picture all of the orbitals in the image above rotated 90°, so that the axes that run through each atomic and molecular orbitals are perpendicular to the paper. The molecular orbitals formed have the same potential energy as the molecular orbitals formed from the 2py-2py overlap. There is less overlap for the parallel atomic orbitals. When the interaction is in-phase, less overlap leads to less electron charge enhancement between the nuclei. This leads to less electron charge between the nuclei for the pi bonding molecular orbital than for the sigma bonding molecular orbital. Less electron character between the nuclei means less plus-minus attraction, less stabilization, and higher potential energy for the pi bonding molecular orbital compared to the sigma bonding molecular orbital. When the interaction is out-of-phase, less overlap leads to less shift of electron charge from between the nuclei. This leads to more electron charge between the nuclei for the pi antibonding molecular orbital than for the sigma antibonding molecular orbital. More electron charge between the nuclei means more plus-minus attraction and lower potential energy for the pi antibonding molecular orbital compared to the sigma antibonding molecular orbital. 167 The expected molecular orbital diagram from the overlap of 1s, 2s, and 2p atomic orbitals is as follows. We will use this diagram to describe O2, F2, Ne2, CO, and NO. π ∗2p ∗ σ2p π 2p π∗2p π 2p σ2p ∗ σ2s σ2s ∗ σ1s σ1s We assume that the electrons would fill the molecular orbitals of molecules like electrons fill atomic orbitals in atoms. We use the following procedure when drawing molecular orbital diagrams. • Determine the number of electrons in the molecule. We get the number of electrons per atom from their atomic number on the periodic table. (Remember to determine the total number of electrons, not just the valence electrons.) • Fill the molecular orbitals from bottom to top until all the electrons are added. Describe the electrons with arrows. Put two arrows in each molecular orbital, with the first arrow pointing up and the second pointing down. • Orbitals of equal energy are half filled with parallel spin before they begin to pair up. We describe the stability of the molecule with bond order. bond order = 1/2 (#e− in bonding MO’s − #e− in antibonding MO’s) We use bond orders to predict the stability of molecules. • If the bond order for a molecule is equal to zero, the molecule is unstable. • A bond order of greater than zero suggests a stable molecule. • The higher the bond order is, the more stable the bond. We can use the molecular orbital diagram to predict whether the molecule is paramagnetic or diamagnetic. If all the electrons are paired, the molecule is diamagnetic. If one or more electrons are unpaired, the molecule is paramagnetic. 168 Chapter 8 & 9 Covalent Bonding and Molecular Structures EXAMPLES: 1. The molecular orbital diagram for a diatomic hydrogen molecule, H2, is ∗ σ1s σ1s • The bond order is 1. Bond Order = 1 2 (2 - 0) = 1 • The bond order above zero suggests that H2 is stable. • Because there are no unpaired electrons, H2 is diamagnetic. 2. The molecular orbital diagram for a diatomic helium molecule, He2, shows the following. ∗ σ1s σ1s • The bond order is 0 for He2. Bond Order = 1 2 (2 - 2) = 0 • The zero bond order for He2 suggests that He2 is unstable. • If He2 did form, it would be diamagnetic. 3. The molecular orbital diagram for a diatomic oxygen molecule, O2, is π ∗2p ∗ σ2p π 2p π∗2p π 2p σ2p ∗ σ2s σ2s ∗ σ1s σ1s • O2 has a bond order of 2. Bond Order = 1 2 (10 - 6) = 2 • The bond order of two suggests that the oxygen molecule is stable. • The two unpaired electrons show that O2 is paramagnetic. 169 4. The molecular orbital diagram for a diatomic fluorine molecule, F2, is π ∗2p ∗ σ2p π 2p π∗2p π 2p σ2p ∗ σ2s σ2s ∗ σ1s σ1s • F2 has a bond order of 1. Bond Order = 1 2 (10 - 8) = 1 • The bond order of one suggests that the fluorine molecule is stable. • Because all of the electrons are paired F2 is diamagnetic. 5. The molecular orbital diagram for a diatomic neon molecule, Ne2, is π ∗2p ∗ σ2p π 2p π∗2p π 2p σ2p ∗ σ2s σ2s ∗ σ1s σ1s • Ne2 has a bond order of 0. Bond Order = 1 2 (10 - 10) = 0 • The bond order zero for Ne2 suggests that Ne2 is unstable. • If Ne2 did form, it would be diamagnetic. 170 Chapter 8 & 9 Covalent Bonding and Molecular Structures We can describe diatomic molecules composed of atoms of different elements in a similar way. The bond between the carbon and oxygen in carbon monoxide is very strong despite what looks like a strange and perhaps unstable Lewis Structure. C O The plus formal charge on the more electronegative oxygen and the minus formal charge on the less electronegative carbon would suggest instability. The molecular orbital diagram predicts CO to be very stable with a bond order of three. π ∗2p ∗ σ2p π 2p π∗2p π 2p σ2p ∗ σ2s σ2s ∗ σ1s σ1s We predict the nitrogen monoxide molecule to be unstable according to the Lewis approach to bonding. N O The unpaired electron and the lack of an octet of electrons around nitrogen would suggest an unstable molecule. NO is actually quite stable. The molecular orbital diagram predicts this by showing the molecule to have a bond order of 2.5. π ∗2p ∗ σ2p π 2p π∗2p π 2p σ2p ∗ σ2s σ2s ∗ σ1s σ1s 171 Figure 8,9.7 Bonding in F2 Electrons in a sigma molecular orbital lead to a sigma bond. nuclei 2px 2px σ2p Figure 8,9.8 Bonding in H2 Electrons in a sigma molecular orbital lead to a sigma bond. nuclei 1s 1s σ1s Formation of sp3 hybrid orbitals One 2s and three 2p atomic orbitals blend 2s 2px 2py 2pz to form four equivalent sp3 hybrid orbitals, which are arranged in a tetrahedral geometry with angles of 109.5°. sp3 109.5° 109.5° 109.5° sp3 sp3 109.5° sp3 sp3 sp3 sp3 109.5° sp3 Figure 8,9.9 Formation of sp3 Hybrid Atomic Orbitals 172 Chapter 8 & 9 Covalent Bonding and Molecular Structures Overlap of atomic orbitals in methane, CH4 H 109.5° C H H H H 1s sp3 109.5° 109.5° C Figure 8,9.10 Bonding in CH4 C sp3 109.5° 1s H H C 1s sp3 sp3 1s H 109.5° C Four C-H sigma bonds due to sp3-1s overlap Formation of sp2 hybrid orbitals One 2s and two 2p atomic orbitals blend 2s 2px 2py 2pz to form three equivalent sp2 hybrid orbitals, which are arranged in a trigonal planar geometry with angles of 120°. This leaves one 2p atomic orbital unhybridized. The axis through the 2p orbital is 90° from the axes through the sp2 hybrid orbitals. sp2 sp2 2p 90° 120° 120° sp2 120° 2 2p sp2 Figure 8,9.11 Formation of sp2 Hybrid Atomic Orbitals sp2 sp2 173 Figure 8,9.12 Bonding in C2H4 Overlap of atomic orbitals in ethene (ethylene), C2H4 1s H sp2 2p 2p sp2 sp2 sp2 1s H sp2 sp2 C C (with stylized orbitals) (with stylized orbitals) 2p 2p sp2 sp2 One C-C sigma bond due to sp2-sp2 overlap and one C-C pi bond due to 2p-2p overlap 1s H 1s H sp2 1s 1s sp2 sp2 sp2 4 C-H sigma bonds due to sp2-1s overlap 1s 1s 174 Chapter 8 & 9 Covalent Bonding and Molecular Structures Figure 8,9.13 Bonding in C2H2 Overlap of atomic orbitals in ethyne (acetylene), C2H2 2px 2py 2pz 2py 1s sp 1s sp H sp C sp (with stylized orbitals) H C (with stylized orbitals) 2pz 2pz sp C C C C sp One C-C sigma bond due to sp-sp overlap C sp C One C-C pi bond due to 2py-2py overlap 1s sp H Two C-H sigma bonds due to sp-1s overlap C H Figure 8,9.14 Bonding in NH3 C One C-C pi bond due to 2pz-2pz overlap 1s 2py 2py Overlap of atomic orbitals in ammonia, NH3 lone pair N H H 107° H The lone pair is more repulsive than the bond groups, so the hydrogen atoms are pushed closer together than we would predict from sp3 hybridization. The angle is about 107° instead of 109.5°. 107° N sp3 1s H N H 1s sp3 107° 1s H Three N-H sigma bonds due to sp3-1s overlap sp3 N 107° 175 Figure 8,9.15 Bonding in NH4+ Overlap of atomic orbitals in ammonium, NH4+ H 109.5° N H H H H 1s sp3 109.5° 109.5° N N sp3 109.5° 1s H H 1s N sp3 sp3 1s H 109.5° N Four N-H sigma bonds due to sp3-1s overlap Figure 8,9.16 Bonding in H2O Overlap of atomic orbitals in water, H2O H O lone pairs 105° H The lone pairs are more repulsive than the bond groups, so the hydrogen atoms are pushed closer together than we would predict from sp3 hybridization. The angle is about 105° instead of 109.5°. 1s H sp3 Two O-H sigma bonds due to sp3-1s overlap O 105° O 105° sp3 1s H 176 Chapter 8 & 9 Covalent Bonding and Molecular Structures Figure 8,9.17 Bonding in BF3 Overlap of atomic orbitals in boron trifluoride, BF3 F sp3 sp2 sp2 120° sp3 B sp2 sp3 F F Three B-F sigma bonds due to sp2-sp3 overlap Formation of sp3d hybrid orbitals One s, three p, and one d atomic orbitals blend 3s 3p 3dz2 3p 3dxz 3p 3dyz 3dx2- y2 3dxy to form five equivalent sp3d hybrid orbitals, which are arranged in a trigonal bipyramid geometry with angles of 90°, 120°, and 180°. This leaves four d atomic orbitals unhybridized. axial equatorial 90° 90° 120° 90° equatorial equatorial 90° 180° 90° 90° 180° 120° 90° axial sp3d sp3d 90° 120° Figure 8,9.18 Formation of sp3d Hybrid Atomic Orbitals sp3d 120° sp3d 90° 120° 120° 90° 90° 120° 90° sp3d 180° 120° 90° 177 Figure 8,9.19 Bonding in PF5 Overlap of atomic orbitals in phosphorus pentafluoride, PF5 sp3d-sp3 overlap sp3d-sp3 overlap FF sp3d-sp3 overlap F 120° P 180° 90° F 120° F sp3d-sp3 overlap F sp3d-sp3 overlap Five P-F sigma bonds due to sp3d-sp3 overlap Figure 8,9.20 Bonding in SF4 Overlap of atomic orbitals in sulfur tetrafluoride, SF4 sp3d-sp3 overlap FF F 120° 90° 180° S F sp3d-sp3 overlap sp3d-sp3 overlap lone pair sp3d-sp3 overlap F Four S-F sigma bonds due to sp3d-sp3 overlap 178 Chapter 8 & 9 Covalent Bonding and Molecular Structures Figure 8,9.21 Bonding in IF3 Overlap of atomic orbitals in iodine trifluoride, IF3 sp3d-sp3 overlap FF sp3d-sp3 overlap I lone pairs 180° 90° F sp3d-sp3 overlap F 3 Three I-F sigma bonds due to sp3d-sp3 overlap Figure 8,9.22 Bonding in XeF2 Overlap of atomic orbitals in xenon difluoride, XeF2 FF sp3d-sp3 overlap 180° lone pairs Xe lone pair sp3d-sp3 overlap F Two Xe-F sigma bonds due to sp3d-sp3 overlap 179 Figure 8,9.23 Formation of sp3d2 Hybrid Atomic Orbitals Formation of sp3d2 hybrid orbitals One s, three p, and two d atomic orbitals blend 3s 3px 3dz2 3py 3dxz 3pz 3dyz 3dx2- y2 3dxy to form six equivalent sp3d2 hybrid orbitals, which are arranged in an octahedral geometry with angles of 90° and 180°. This leaves three d atomic orbitals unhybridized. sp3d2 sp3d2 180° 90° 180° 180° 90° 90° sp3d2 sp3d2 90° 90° 90° sp3d2 sp3d2 180° 180° 180° sp3d2 sp3d2 sp3d2 90° sp3d2 180° sp3d2 90° sp3d2 Overlap of atomic orbitals in sulfur hexafluoride, SF6 sp3d2-sp3 overlap sp3d2-sp3 overlap FF F sp3d2-sp3 overlap F sp3d2-sp3 overlap S F sp3d2-sp3 overlap F F sp3d2-sp3 overlap Six S-F sigma bonds due to sp3d2-sp3 overlap Figure 8,9.24 Bonding in SF6 180 Chapter 8 & 9 Covalent Bonding and Molecular Structures Figure 8,9.25 Bonding in IF5 Overlap of atomic orbitals in iodine pentafluoride, IF5 sp3d2-sp overlap sp3d2-sp3 overlap sp3d2-sp3 overlap FF F F sp3d2-sp3 overlap I F lone pair F sp3d2-sp3 overlap Five I-F sigma bonds due to sp3d2-sp3 overlap Figure 8,9.26 Bonding in XeF4 Overlap of atomic orbitals in xenon tetrafluoride, XeF4 sp3d2-sp3 overlap sp3d2-sp3 overlap lone pair F F sp3d2-sp3 overlap Xe F lone pair F sp3d2-sp3 overlap Four Xe-F sigma bonds due to sp3d2-sp3 overlap 181 The following assumptions lie at the core of the valence‑bond model. • Only the highest energy electrons participate in bonding. • Covalent bonds usually form to pair unpaired electrons. • Covalent bonds arise due to the overlap of atomic orbitals on adjacent atoms. When the columns on the periodic table are numbered by the A group convention, the number of valence electrons in each atom of a representative element is equal to the element’s group number on the periodic table. One valence electron 1 H Number of valence electrons equals the A-group number 3A 5 B 4A 6 C 5A 7 6A 8 O 15 16 33 As 7A 9 N P 8A F S 34 17 Cl 35 Se Br 52 53 Te I 2 He 10 Ne 18 Ar 36 Kr Figure 8,9.27 Valence Electrons 54 Xe Table 8,9.1 Most Common Covalent Bonding Patterns Element Frequency of pattern Number of bonds Number of lone pairs Example H always 1 0 H B usually 3 0 B C usually 4 0 rarely 3 1 C usually 3 1 N commonly 4 0 usually 2 2 commonly 1 3 O rarely 3 1 O usually 1 3 X N, P, & As O, S, & Se F, Cl, Br, & I C or C or N O or O C 182 Chapter 8 & 9 Covalent Bonding and Molecular Structures Formal Charge = Group Number − Number of lines − Number of dots Formal Charge = (The number of valence electrons necessary to be uncharged) − (the number of electrons from bonds assuming equal sharing) − (the number of electrons from lone pairs) Sample Study Sheet 8,9.1: Explanation of Bonding Patterns Valence Bond Model Tip-off: You will be given a bonding pattern for a nonmetal atoms and asked to explain how it is possible in terms of the valence bond model. General Procedure Step 1 Write, “Only the highest energy electrons participate in bonding.” Step 2 Draw the orbital diagram for the valence electrons. Include the empty d orbitals for the third period nonmetals and below. Step 3 If there is a formal charge, add or subtract electrons. a. Add one electron for -1. b. Subtract one electron for +1. Step 4 If necessary, promote one or more electrons from a pair to an empty orbital to get the number of unpaired electrons equal to the number of bonds to be explained. Step 5 Rewrite the orbital diagram, showing the predicted hybrid orbitals. Step 6 Write, “Covalent bonds form in order to pair unpaired electrons.” Step 7 Indicate that the unpaired electrons form the bonds. Step 8 Indicate that the paired electrons are lone pairs. Exercise 8,9.1 - Explanation of Bonding Patterns Explain the following bonding patterns a. C – 4 bonds, no lone pairs, and no formal charge b. N – 4 bonds, no lone pairs, and a +1 formal charge c. O – 1 bond, 3 lone pairs, and a −1 formal charge d. S – 4 bonds, 1 lone pair, and no formal charge e. I – 5 bonds, 1 lone pair, and no formal charge 183 Tip-off: You will be given a Lewis structure, and you will be asked the following. • What is the hybridization for each atom in the structure? • Write a description of the formation of each bond in terms of the overlap of atomic orbitals. • Describe the electron group geometry around each atom that has two or more atoms attached to it. • Draw the geometric sketch of the molecule, including bond angles. • Describe the molecular geometry around each atom that has two or more atoms attached to it. General Steps for predicting hybridization for each atom in a structure. Step 1 Count the number of electron groups around each atom. An electron group is any one of the following. • single bond • double or triple bond (counts as one group) • lone pair Step 2 Apply the following guidelines. See Table 8,9.2. 1 group - No hybridization (Hydrogen atoms are the only atoms that have only one group around them in a Lewis structure. The 1s orbital is the only occupied atomic orbital for hydrogen atoms. Because there is no other orbital with which to blend, there is no hybridization. Hydrogen atoms use the 1s atomic orbital to form bonds.) 2 groups - sp hybridization 3 groups - sp2 hybridization 4 groups - sp3 hybridization 5 groups - sp3d hybridization 6 groups - sp3d2 hybridization General Steps for describing the bonds in terms of the overlap of atomic orbitals. Step 1 Describe single bonds that do not involve hydrogen as due to the overlapping of two hybrid orbitals, one from each atom. Step 2 Describe single bonds that involve hydrogen as due to the overlapping a hybrid orbital with a 1s orbital for the hydrogen atom. Step 3 Describe double bonds as one bond forming from the overlap of two hybrid orbitals, one from each atom. Describe the second bond as due to the overlap of two p orbitals, one from each atom. Step 4 Describe triple bonds as one bond forming from the overlap of two hybrid orbitals, one from each atom. Describe each of the other two bonds as due to the overlap of two p orbitals, one from each atom. Sample Study Sheet 8,9.2: From Lewis Structures 184 Chapter 8 & 9 Covalent Bonding and Molecular Structures To determine the electron group geometry around each atom that is attached to two or more atoms, use the number of electron groups around each atom and the guidelines found on Table 8,9.2. Use one or more of the following geometric sketches shown on Table 8,9.2 for the geometric sketch of you molecule. To describe the molecular geometry around each atom that has two or more atoms attached to it, count the number of bond groups and apply the guidelines found on Table 8,9.2. (Single, double, and triple bonds all count as one bond group.) Note that if all of the electron groups attached to the atom are bond groups (no lone pairs), the molecular geometry is the same as the electron group geometry. 185 Table 8,9.2 Hybridization and Geometry e− groups 2 Hybrid Orbitals sp 3 sp2 General Geometric Sketch 180° 120° 4 sp3 e− group geometry linear bond angles 180° bond groups 2 lone pairs 0 molecular geometry linear trigonal planar 120° 3 0 trigonal planar tetrahedral 109.5° 2 1 bent 4 3 0 1 2 2 tetrahedral trigonal pyramid bent 5 0 4 3 2 1 2 3 6 5 0 1 4 2 109.5° 5 sp3d trigonal bipyramid 90° 90°, 120°, 180° 180° 120° 120° 6 sp3d2 octahedral 180° 90° 90°, 180° trigonal bipyramid see-saw T-shaped linear 180° 90° octahedral square pyramid square planar 186 Chapter 8 & 9 Covalent Bonding and Molecular Structures Table 8,9.3 Common Covalent Bonding Patterns for the Nonmetal Elements Element Number of bonds Number of lone pairs Formal charge H always 1 0 0 C usually 4 0 0 rarely 3 1 -1 usually 3 1 0 commonly 4 0 +1 usually 2 2 0 commonly 1 3 -1 rarely 3 1 +1 F always 1 3 0 P and As usually 3 1 0 commonly 5 0 0 commonly 4 0 +1 usually 2 2 0 commonly 4 1 0 commonly 6 0 0 commonly 1 3 -1 possibly 3 1 +1 usually 1 3 0 possibly 3 2 0 possibly 5 1 0 possibly 7 0 0 rarely 2 3 0 rarely 4 2 0 N O S and Se halogens except F Xe Table 8,9.4 usually none The Most Common Bonding Pattern for Each Nonmetal Element Group Number of Covalent Bonds Number of Lone Pairs 4A (C) 4 0 5A (N, P, As) 3 1 6A (O, S, Se) 2 2 7A (halogens) 1 3 8A (noble gases) none 4 187 O O O O N O O N O O N O + + + Tip-off: You are asked to draw a resonance hybrid. Sample Study General Steps Sheet 8,9.3: • Draw the skeletal structure with solid lines for the bonds that are found in all of the resonance forms. • Where there is sometimes a multiple bond and sometimes not, draw a dotted line. Resonance Hybrids • Draw in all of the lone pairs that are found on every one of the resonance forms. (Leave off the lone pairs that are on one or more resonance form but not on all of them.) • Put on full formal charges on those atoms that have the formal charge in all of the resonance forms. • Put on partial formal charges on the atoms that have formal charges in one or more of the resonance forms but not in all of them. The resonance hybrid for the nitrate polyatomic ion is below. O O N A bond found in all the resonance structures A bond found in at least one but not all the resonance structures − O A lone pair found in all the resonance structures Figure 8,9.28 Resonance Hybrid 188 Chapter 8 & 9 Sample Study Sheet 8,9.4: Resonance and Resonance forms Covalent Bonding and Molecular Structures Tip-off - You are asked to draw a Lewis structure for a molecule or a polyatomic ion, and the structure drawn has the following form. X Y Z Z can have more than one lone pair. X and Y can have lone pairs. The X‑Y bond can be a triple bond. The Y‑Z bond can be a double bond. Z cannot be F with one bond and three lone pairs or O with two bonds and two lone pairs. General Steps: Follow these steps when writing the resonance forms. • Shift one of the lone pairs on one adjacent atom down to form a multiple bond. • Shift one of the multiple bonds up to form a lone pair. You might find it useful to draw the arrows to indicate the hypothetical shift of electrons. • Repeat this process for each adjacent atom with a lone pair. X Y Z Sample Study Sheet 8,9.5: Drawing Lewis Structures X Y Z Tip-off: You are asked to draw a Lewis structure from a chemical formula for a molecule or a polyatomic ion. General Steps (As you become more experienced with the process of drawing Lewis structures, you will see shortcuts that will allow you to bypass some or all of the following steps.) Step 1 Determine the number of valence electrons for the molecule or polyatomic ion. • For neutral molecules, the total number of valence electrons is the sum of the valence electrons of each atom. Remember that the number of valence electrons for any one of the representative elements is equal to its group number, using the A group convention for numbering the groups. For example, chlorine, Cl, is in group 7A, so it has seven valence electrons. Hydrogen has one valence electron. • For polyatomic cations, the total number of valence electrons is the sum of the valence electrons for each atom minus the charge. • For polyatomic anions, the total number of valence electrons is the sum of the valence electrons for each atom plus the charge. 189 Step 2 Draw a reasonable skeletal structure joining the atoms with single bonds. One or more of the following guidelines might help you with this step. • Try to set the atoms in space to gain the most common number of bonds for each atom. Table 8,9.3 lists the most common bonding patterns for the nonmetal elements. • Apply the following guidelines in deciding what goes in the center of your structure. ∗ Hydrogen and fluorine atoms are never in the center. ∗ Oxygen atoms are rarely in the center. ∗ The element with the fewest atoms in the formula is usually in the center. ∗ The atom that is capable of making the most bonds is usually in the center. See Table 8,9.3. ∗ The atom with the lowest electronegativity is often in the center. • Oxygen atoms rarely bond to other oxygen atoms. • The molecular formula often reflects the structural formula. • Carbon atoms commonly bond to other carbon atoms. Step 3 Subtract 2 electrons from the total for each of the single bonds (lines) described in Step 2 above. Step 4 Try to distribute the remaining electrons as lone pairs to get eight total electrons around each atom except hydrogen, beryllium, and boron. When an atom has a total of eight electrons around it, we say it has an octet of electrons. Atoms of the noble gases other than helium have an octet of electrons around them. The orbital diagram and electron‑dot structure for the valence electrons of neon show this. 2p Ne 2s The stability of the octet is reflected in the fact that atoms in reasonable Lewis structures often have this octet. ∗ Carbon, nitrogen, oxygen and fluorine always have 8 electrons around them in a reasonable Lewis structure. ∗ Beryllium and boron can have less than eight electrons but never more than eight. ∗ The nonmetal elements below the second period (P, S, Cl, Se, Br, and I) usually have eight electrons around them but often have more. ∗ The metalloids arsenic, As, and tellurium, Te, form covalent bonds in similar ways to phosphorus, P, and sulfur, S, so they usually have eight electrons around them but often have more. ∗ Hydrogen will always have two electrons total from its one bond. 190 Chapter 8 & 9 Covalent Bonding and Molecular Structures Step 5 Do one of the following. • If in Step 4 you were able to get eight electrons around each atom other than those of hydrogen, beryllium, and boron and if you used all of the remaining valence electrons, go to Step 6. • If you have electrons remaining after each of the atoms other than those of hydrogen, beryllium, and boron have their octet, you can put more than eight electrons around the elements in the third period and below. • If you do not have enough electrons to get octets of electrons around each atom (other than hydrogen, beryllium, and boron), convert one lone pair into a multiple bond for each 2 electrons that you are short. ∗ If you would need two more electrons to get octets, convert one lone pair in your structure to a second bond between two atoms. ∗ If you would need four more electrons to get octets, convert two lone pairs into bonds. This could mean creating a second bond in two different places or creating a triple bond in one place. ∗ If you would need six more electrons to get octets, convert three lone pairs into bonds. ∗ Etc. Step 6 Check your structure. • First check to see if all of the atoms have their most common bonding pattern. If each atom has its most common bonding pattern, your structure is a reasonable structure. Skip Step 7, and proceed to Step 8. • Determine the formal charge for each atom that has other than its most common bonding pattern. (Atoms with their most common bonding pattern always have a zero formal charge.) Formal Charge = Group Number − Number of lines − Number of dots ∗ If all of the formal charges are zero, skip Step 7, and go to Step 8. ∗ If you have formal charges, continue with Step 7. Step 7 Try to rearrange your structure to eliminate or at least reduce any formal charges. (This step is unnecessary if there are no formal charges.) • One way to eliminate formal charges is to return to Step 2 and try another skeleton. • Formal charges can also be eliminated by converting lone pairs into bonds. This process leads to placing more than eight electrons around some atoms. Here is where we hit controversy. Chemists are not in agreement about whether it is better to emphasize the importance of octets in reasonable Lewis structures or better to minimize formal charges. 191 Step 8: Once you have a reasonable Lewis structure, consider the possibility of resonance. • If resonance is possible, write all of the reasonable resonance structures for the original Lewis structure. • Draw the resonance hybrid from the resonance forms. It is possible to generate more than one reasonable Lewis structure for a given formula. When this happens, the following rules will help you decide which is most likely. • The more usual or common the bond is, the more stable the structure. See Table 8,9.3. • A structure with formal charges is less stable than one without formal charges. • “+” formal charges are more likely on the less electronegative elements and “-” formal charges are more likely on the more electronegative elements. It is possible to have two Lewis structures for the same molecular formula, which are considered equally reasonable according to the above criteria. The substances represented by the Lewis structures are isomers, molecules that have the same molecular formula but different structural formulas. The dimethyl ether and ethanol are isomers of C2H6O. H H C H H O C H H dimethyl ether H H H C C H H ethanol O H 192 Chapter 8 & 9 Covalent Bonding and Molecular Structures THE FOLLOWING IS A SUMMARY OF THE STEPS FOR DRAWING LEWIS STRUCTURES FROM CHEMICAL FORMULAS. Step 1 Determine the number of valence electrons for the molecule or polyatomic ion. Step 2 Draw a reasonable skeletal structure joining the atoms with single bonds. Step 3 Subtract 2 electrons from the total for each of the single bonds described in Step 2 above. Step 4 Try to distribute the remaining electrons as lone pairs to get eight total electrons around each atom except hydrogen, beryllium, and boron. Step 5 Do one of the following. • If in Step 4 you were able to get eight electrons around each atom other than those of hydrogen, beryllium, and boron and if you used all of the remaining valence electrons, go to Step 6. • If you have electrons remaining after the atoms other than those of hydrogen, beryllium, and boron have their octet, you can put more than eight electrons around the elements in the third period and below. • If you do not have enough electrons to get octets of electrons around each atom (other than those of hydrogen, beryllium, and boron), convert one lone pair into a multiple bond for each 2 electrons that you are short. Step 6 Check your structure. Step 7 Try to rearrange your structure to eliminate or at least reduce any formal charges. Step 8 Once you have a reasonable Lewis structure, consider the possibility of resonance. If resonance is possible, draw the reasonable resonance structures and the resonance hybrid for the structure. Exercise 8,9.2 - Drawing Lewis Structures Draw a reasonable Lewis structure for each of the following. If the structure has resonance, draw all of the reasonable resonance structures and the resonance hybrid. a. CH3Br h. C3H7OH b. ClF3 i. HCO2− c. CH2O j. CH2CHF d. CN− k. HCO2H e. CF3CHCl2 l. NH2COCH3 f. C2H6O m. SO42− g. C2H4F2 n. PO43− 193 Write chemical formulas from names. Draw Lewis structures. Predict molecular geometry. Use electronegativities to predict bond polarity. Predict molecular polarity Identify types of substances Predict types of attractions between particles. Predict relative strengths of these attractions. Predict relative melting points and boiling points. Figure 8,9.29 Connections Between Skills This figure shows the connections between the skills covered in earlier chapters and the skills presented in this chapter. To master the new skills, you need to have mastered the old ones. Start at the top of the sequence and work your way down, convincing yourself as you go along that you can do each task. For a molecule to be polar, it must meet two criteria: • It must contain polar bonds. • It must have an asymmetrical distribution of its polar bonds. Electrons shift toward the chlorine atom, forming partial plus and minus charges. δ+ Hydrogen attracts electrons less. δ− H Cl Chlorine attracts electrons more. Figure 8,9.30 The HCl molecule. The polar covalent H‑Cl bond (or bond dipole) leads to an overall separation of positive and negative charge in the molecule. Therefore, the HCl molecule is a polar molecule, or a molecular dipole. H δ+ δ− O 105° H δ+ Asymmetrical and polar F δ− δ+ B δ− 120° δ− F F Symmetrical and nonpolar 194 Chapter 8 & 9 Sample Study Sheet 8,9.6: Predicting Molecular Polarity Covalent Bonding and Molecular Structures Tip-off – You are asked to predict whether a molecule is polar or nonpolar; or you are asked a question that cannot be answered unless you know whether a molecule is polar or nonpolar. (For example, you are asked to predict the type of attraction holding the particles together in a given liquid or solid.) General Steps Step 1 Draw a reasonable Lewis structure for the substance. Step 2 Identify each bond as either polar or nonpolar. (If the difference in electronegativity for the atoms in a bond is greater than 0.4, we consider the bond polar. If the difference in electronegativity is less than 0.4, the bond is essentially nonpolar.) • If there are no polar bonds, the molecule is nonpolar. • If the molecule has polar bonds, move on to Step 3. Step 3 If there is only one central atom, examine the electron groups around it. • If there are no lone pairs on the central atom, and if all the bonds to the central atom are the same, the molecule is nonpolar. • If the central atom has at least one polar bond and if the groups bonded to the central atom are not all identical, the molecule is probably polar. Move on to Step 4. Step 4 Draw a geometric sketch of the molecule. Step 5 Determine the symmetry of the molecule using the following steps. • Describe the polar bonds with arrows pointing toward the more electronegative element. Use the length of the arrow to show the relative polarities of the different bonds. (A greater difference in electronegativity suggests a more polar bond, which is described with a longer arrow.) • Decide whether the arrangement of arrows is symmetrical or asymmetrical ∗ If the arrangement is symmetrical and the arrows are of equal length, the molecule is nonpolar. ∗ If the arrows are of different lengths, and if they do not balance each other, the molecule is polar. ∗ If the arrangement is asymmetrical, the molecule is polar. Exercise 8,9.3 - Predicting Molecular Polarity Decide whether the molecules represented by the following formulas are polar or nonpolar. (You may need to draw Lewis structures and geometric sketches in order to do this.) a. BCl3 e. CBr4 b. C3H8 f. CH3CO2H c. CS2 g. Br2O d. NH3 h. IF3 195 Tip-off – You are asked to predict the relative strengths of attractions between particles of two substances, or you are asked a question that cannot be answered unless you know those relative strengths. (For example, you are asked to compare certain properties of substances, such as their relativemelting and boiling point temperatures.) General Steps Step 1 Determine the type of attraction between the particles using the following steps. Step 1(a) Classify each substance as either a metallic element, an element composed of atoms, an element composed of molecules, an ionic compound, or a molecular compound. • Metallic elements have metallic bonds. • The particles in elements that are composed of atoms (the noble gases) and molecules (the other nonmetallic elements) are held together by London forces. • The ions in ionic compounds are held together by ionic bonds. • Continue to Step 1(b) for molecular compounds. Step 1(b) For molecular compounds, draw the Lewis structure for the molecule. • If the Lewis structure contains an O‑H, N‑H, or H‑F bond, the attractions that form when the substance condenses are hydrogen bonds enhanced by London forces. • For other molecular compounds, do Step 1(c). Step 1(c): If there are no O‑H, N‑H, or H‑F bonds, determine the polarity of the bonds. • If there are no polar bonds, the molecules are nonpolar and form London forces when they condense. • If there is at least one polar bond, do Step 1(d). Step 1(d): Predict whether the polar bonds are symmetrically or asymmetrically arranged. • If the distribution of polar bonds is symmetrical and their dipoles equal, the molecules are nonpolar and form London forces when they condense. • If the distribution of polar bonds is asymmetrical, or symmetrical with unequal dipoles, the molecules are polar and form dipole‑dipole attractions enhanced by London forces when they condense. Sample Study Sheet 8,9.7: Predicting Types of Attractions and Relative Strengths of Attractions Between Particles 196 Chapter 8 & 9 Covalent Bonding and Molecular Structures Step 2 Although we cannot predict the relative strengths of attractions between all particles, we can apply one of the following guidelines to predict the relative strengths of attractions between some particles. • For substances that contain particles of about the same size, the substances with chemical bonds (ionic, covalent, or metallic) have stronger attractions between particles than substances with intermolecular attractions (hydrogen bonds, dipole‑dipole attractions, or London forces). Chemical bonds are generally stronger than intermolecular attractions. • For molecular substances that contain molecules of about the same size, substances with hydrogen bonds have stronger attractions between the particles than substances with either dipole‑dipole attractions or London forces, and substances with dipole‑dipole attractions have stronger attractions between the particles than London forces. Hydrogen bonds are generally stronger than dipole‑dipole attractions, which are generally stronger than London forces. • For molecular substances that have the same type of intermolecular attraction, larger molecules form stronger mutual attractions. Larger molecules tend to have stronger attractions. Figure 8,9.31 Scheme for Predicting Types of Attractions What kind of substance? Metallic element Metallic bonds Diamond Other nonmetallic elements Covalent bonds London forces Ionic compound Molecular compound Ionic bonds Do the molecules contain O-H, N-H, or H-F bonds? No Yes Are any bonds polar? No London forces Hydrogen bonds Yes Symmetrical or asymmetrical arrangement of polar bonds? Asymmetrical Dipole-dipole attractions Symmetrical London forces 197 Table 8,9.5 Solid and Liquid Form Type of substance Particles to visualize Metal Cations in a sea of electrons Atoms Carbon atoms Molecules Elements Noble Gases Carbon (diamond) Other Nonmetal Elements Ionic Compounds Molecular Compounds Nonpolar Molecular Polar molecules without H‑F, O‑H, or N‑H bond Molecules with H‑F, O‑H or N‑H bond Network crystals Examples Au Type of attraction between particles in solid or liquid Metallic bond Xe C(dia) H2, N2, O2, F2, Cl2, Br2, I2, S8, Se8, P4 NaCl London forces Covalent bonds London forces Hydrocarbons London forces Molecules HCl Dipole-Dipole forces Molecules HF, H2O, alcohols, NH3 Hydrogen bonds Atoms SiO2 Covalent bonds Cations and Anions Molecules Ionic bond Exercise 8,9.4 - Predicting Type of Attraction What is the primary type of attraction that holds the particles of each of these substances in the liquid or solid form. a. Ir e. NH4OH b. AlF3 f. BF3 c. C2H6 g. CH2O d. CH3NH2 h. C3H7OH 198 Chapter 8 & 9 Covalent Bonding and Molecular Structures Exercise 8,9.5 - Predicting Types and Strengths of attractions Between Particles For each of the following substances, write the name for the type of particle that forms its basic structure and the name of the primary type of attraction that can form between these particles. From each pair of substances, choose the one that you expect would have stronger interparticle attractions. a. ethanol, C2H5OH (in alcoholic beverages) dimethyl ether, CH3OCH3 (refrigerant and solvent for propellant sprays) b. manganese, Mn (improves corrosion resistance and hardness when alloyed with other metals) carbon tetrachloride, CCl4 (used to add chlorine atoms to organic compounds) c. propane, C3H8 (fuel and aerosol propellant) n-heptane, C7H16 (standard for octane rating – 100% n-hexane is zero octane) d. acetone, CH3COCH3 (solvent for paints and varnishes) lithium nitrate, LiNO3 (used to make ceramics and in rocket propellants) 199 Hydrophobic and Hydrophilic Substances Organic compounds are often polar in one part of their structure and nonpolar in another part. The polar section, which is attracted to water, is called hydrophilic (literally, “water loving”), and the nonpolar part of the molecule, which is not expected to be attracted to water, is called hydrophobic (“water fearing”). If we need to predict the relative water solubility of two similar molecules, we can expect the one with the proportionally larger polar portion to have higher water solubility. We predict that the molecule with the proportionally larger nonpolar portion will be less soluble in water. As an example, we can compare the structures of epinephrine (adrenaline) and amphetamine. Epinephrine is a natural stimulant that is released in the body in times of stress. Amphetamine (sold under the trade name Benzedrine) is an artificial stimulant that causes many of the same effects as epinephrine. The three –OH groups and the N H bond in the epinephrine structure cause a greater percentage of that molecule to be polar, so we predict that epinephrine would be more soluble in water than amphetamine. Nonpolar OH NHCH3 HO NH2 Polar Polar Nonpolar HO amphetamine epinephrine The molecular representations above are known as line drawings. The corners, where two lines meet, represent carbon atoms, and the end of any line that does not have a symbol attached also represents a carbon atom. We assume that each carbon has enough hydrogen atoms attached to yield four bonds total. Compare the line drawings to the more complete Lewis structures below: H H C C H H C C C H C C H H H C H C H H NH2 amphetamine HO HO C C H OH C C C H C C H H H C NHCH3 H epinephrine The difference in polarity between epinephrine and amphetamine has an important physiological consequence. The cell membranes that separate the blood stream from the brain cells have a nonpolar interior and a polar exterior. The more polar a molecule is, the more likely it is to be attracted to the exterior of the cell membrane enough to be blocked from moving into the cell. This tends to prevent polar substances in the blood from moving into the brain tissue. Epinephrine is too polar to move from the blood stream into the brain, but amphetamine is not. The stimulant effects of amphetamine are in part due to its ability to pass through the blood brain barrier. Methamphetamine is a stimulant that closely resembles amphetamine. With only 200 Chapter 8 & 9 Covalent Bonding and Molecular Structures one polar N-H bond, it has very low water solubility, but it can be induced to dissolve in water when converted to the much more polar, ionic form called methamphetamine hydrochloride. On the street, the solid form of this illicit ionic compound is known as crystal meth. Nonpolar CH3NH methamphetamine Nonpolar Slightly polar CH3NH2+ Cl − Very polar methamphetamine hydrochloride