* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Unit 2: Atomic Structure
Survey
Document related concepts
Transcript
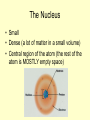
Unit 2: Atomic Structure The Nucleus • Small • Dense (a lot of matter in a small volume) • Central region of the atom (the rest of the atom is MOSTLY empty space) Subatomic Particles Table O Proton Neutron Electron Charge Location positive neutral negative In nucleus In nucleus Size 1 amu (atomic mass unit) Atomic # = # protons (known as nuclear charge) 1 amu Outside nucleus 1/1836 the mass of a proton Info Mass # = atomic # (protons) + # of neutrons When an atom is neutral, # protons = #electrons Subatomic Particles • • Protons and Neutrons are also called nucleons Protons + neutrons = mass # (rounded atomic mass) 1. Example: carbon has an atomic mass of 12.0111, so the mass # is 12 (round to tenths if possible) 2. Protons and neutrons make up most of the mass of the atom • Periodic Table 1. There is a key at the top of the table 2. The first oxidation # for each element is referred to as the preferred charge • Always assume an atom is neutral unless told otherwise