* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chromatography (Principles and Classifications)
Genetic code wikipedia , lookup
Multi-state modeling of biomolecules wikipedia , lookup
Expanded genetic code wikipedia , lookup
Cell-penetrating peptide wikipedia , lookup
Immunoprecipitation wikipedia , lookup
Community fingerprinting wikipedia , lookup
Proteolysis wikipedia , lookup
List of types of proteins wikipedia , lookup
Circular dichroism wikipedia , lookup
Monoclonal antibody wikipedia , lookup
Implicit solvation wikipedia , lookup
Protein adsorption wikipedia , lookup
Gel electrophoresis of nucleic acids wikipedia , lookup
Agarose gel electrophoresis wikipedia , lookup
Western blot wikipedia , lookup
Biochemistry wikipedia , lookup
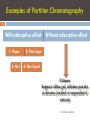
Chapter 4-1 Chromatography Is a technique used to separate and identify the components of a mixture. Dr Gihan Gawish 1 2 Before a protein or other biological macromolecule can be rigorously studied from a structural and functional basis, it must be purified. The problems that can arise during protein purification become clear when one considers that a single protein has to be purified from a mixture of as many 10,000 proteins, each of which are made up of the same constituent amino acids. Proteins differ in size (how many amino acids), charge (how many positively and negatively charged amino acids), and in sequence and presence of specific binding sites on the proteins. Any technique that could be used to purify protein must be based on these inherent differences. Dr Gihan Gawish Chromatography Application in Biomolecules General Principles of Chromatography 3 Separation of molecules by distribution between a stationary phase and a mobile phase. A stationary phase can be solid, gel, or liquid. Also called matrix, resin, or beads. The mobile phase is the solvent and it is usually a liquid, but may also be a gas. Also called eluting buffer The compounds to be separated are considered solutes Dr Gihan Gawish Basis of Chromatography Differential retardation of materials caused by interactions with the stationary phase: Reservoir + Sample mobile phase stationary phase or Resin Frit Flow-through Dr Gihan Gawish 4 Classification of Chromatography by mobile phase: 1. 2. by stationary phase or shape 1. 2. 3. 5 Gas chromatography, Liquid chromatography or its variations Paper chromatography, Thin-layer chromatography Column chromatography Dr Gihan Gawish Classification by the Separation mode The mechanism that causes the stationary phase to retard the movement of molecules 1. Sieve mechanism separation according to size or MW. (molecular sieve = gel filtration = size exclusion = gel permeation) 2. 6 Charge interaction separation based on net charge Dr Gihan Gawish Classification by the Separation mode 7 3. Solubility characteristics separation based on polarity Hydrophobic chromatography, reverse-phase chromatography, Adsorption or normal-phase chromatography 4. Biological or Specific interaction capture any molecule that exhibit such property affinity chromatography, dye-chromatography Antibody-antigen: (Immuno precipitations and other forms) Dr Gihan Gawish Gel Filtration Chromatography also called "Molecular Sieve“ or "Size Exclusion“: (column matrix) "beads" of hydrated, porous polymer. Stationary phase Mobile phase: solvent. 8 buffer or Dr Gihan Gawish Gel Filtration Chromatography 9 Concept of separation: molecules "filter" through the porous beads: 1. 2. 3. Large molecules can’t get through any pores in the beads and move more rapidly through the column, emerging (eluting) sooner. Smaller molecules may enter some pores in the beads, and thus, have more space to "explore" elute later. Ions and small molecules can enter Drthrough all pores Gihan Gawish of the beads elute last Gel Filtration Chromatography The porous beads represent a restricted space or volume Molecules may diffuse to this space if permitted by pore size Absorption Vo 10 Ve Volume Dr Gihan Gawish Vt Gel Filtration Chromatography 11 For every column, three volumes should be distinguished: 1. The void volume, Vo: which is the volume external to the beads This space is accessible to all molecules entering the gel Can be determined by the elution of HMW molecules 2. The total volume, Vt: can be calculated from the dimension of the column 3. The elution volume, Ve, of molecules All molecules must eluted between the void volume and total volume of the column Dr Gihan Gawish Gel Filtration ChromatographyPartition Coefficient In classical and more rigorous science, elution position of any molecules should be reported as partition coefficient (Kav) rather than volume. Kav= 12 (Ve-Vo) (Vt-Vo) Dr Gihan Gawish Gel Filtration Chromatography 13 Kav represents the fraction of bead-restricted volume into which a solute could partition Kav ranges between 0 and 1 for all molecules Kav allows comparison between columns of different dimensions and/or materials Dr Gihan Gawish Examples of Partition Chromatography 14 With adsorptive effect 1- Paper. 3- Gel Without adsorptive effect 2- Thin layer 4- Gas liquid Column Support: sillica gel, cellulose powder or dextran (washed or suspended of solvent) Dr Gihan Gawish Adsorptive Chromatography (adsorptive effect) 15 A stationary phase solid (support; matrix) + liquid (adsorbent) The mobile phase is the solvent The relative movement of each molecule is a result of a balance between the driving force and retardation force. Dr Gihan Gawish Common Materials Used in Adsorption Chromatography in Biochemistry 16 Material Alumina Silica gel Activated carbon Calcium phosphate gel Hydroxyapatite Substance separated Small organic molecules, proteins Sterols, amino acids Peptides, amino acids, carbohydrates Proteins, polynucleotide Nucleic acids Dr Gihan Gawish