* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter 5 Gases
History of manufactured fuel gases wikipedia , lookup
Bernoulli's principle wikipedia , lookup
Aliso Canyon gas leak wikipedia , lookup
Gaseous detection device wikipedia , lookup
Gas chromatography wikipedia , lookup
Diamond anvil cell wikipedia , lookup
Industrial gas wikipedia , lookup
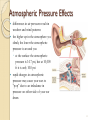
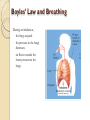
Chapter 5 Gases Air Pressure & Shallow Wells Gases •Are mostly empty space • Occupy containers uniformly and completely • The densities of gases are much smaller than those of liquids and solid and highly variable depending on temperature and pressure • Expand infinitely • Diffuse and mix rapidly 2 Gases Pushing gas molecules are constantly in motion as they move and strike a surface, they push on that surface ◦ push = force if we could measure the total amount of force exerted by gas molecules hitting the entire surface at any one instant, we would know the pressure the gas is exerting ◦ pressure = force per unit area Pressure: Force Unit area 3 Atmospheric Pressure Effects differences in air pressure result in weather and wind patterns the higher up in the atmosphere you climb, the lower the atmospheric pressure is around you ◦ at the surface the atmospheric pressure is 14.7 psi, but at 10,000 ft it is only 10.0 psi rapid changes in atmospheric pressure may cause your ears to “pop” due to an imbalance in pressure on either side of your ear drum 4 The Pressure of a Gas result of the constant movement of the gas molecules and their collisions with the surfaces around them the pressure of a gas depends on several factors ◦ number of gas particles in a given volume ◦ volume of the container ◦ average speed of the gas particles 5 Measuring Air Pressure Units Pa (SI unit) torr mm Hg atm bar Conversions 1 atm = 760 mm Hg (exact) 1 torr = 1 mm Hg 1 bar = 1 x 105 Pa (exact) 1 atm = 101 325 Pa 1 atm = 14.7 psi (exact) Barometer Gases and Gas Pressure The Gas Laws Ideal Gas: A gas whose behavior follows the gas laws exactly. The physical properties of a gas can be defined by four variables: P T V n pressure (atm) temperature (calculation must be in Kelvin) volume (L) number of moles The Ideal Gas Law, PV = nRT, - models the behavior of ideal gases. Other gas laws can be derived from the Ideal Gas Law for either one set of conditions or for two sets of conditions (initial and final conditions). To derive gas laws for two sets of conditions, solve the Ideal Gas Law for R PV L atm R = 0.08206 ---- = R K mol nT Boyle’s law o pressure of a gas is inversely proportional to its volume o constant T and amount of gas o as P increases, V decreases by the same factor 1 V a P (constant n and T) Two sets of conditions P1 x V1 = P2 x V2 Boyles’ Law and Breathing During an inhalation, • the lungs expand. • the pressure in the lungs decreases. • air flows towards the lower pressure in the lungs. Examples A cylinder with a movable piston has a volume of 7.25 L at 4.52 atm. What is the volume at 1.21 atm? A balloon is put in a bell jar and the pressure is reduced from 782 torr to 0.500 atm. If the volume of the balloon is now 2780 mL, what was it originally? Charles’s Law In Charles’s Law, • the Kelvin temperature of a gas is directly related to the volume. • P and n are constant. • when the temperature of a gas increases, its volume increases. Charles' Law & Absolute Zero • For two conditions, Charles’s law is written T1 Volume (L) of 1 g O2 @ 1500 torr Volume (L) of 1 g O2 @ 2500 torr 0.5 = V2 (P and n constant) T2 Charles’s Law can be used to approximate absolute zero. At a temperature of absolute zero (0K), theoretically an ideal gas has no volume. Volume (L) of 0.5 g O2 @ 1500 torr Volume (L) of 0.5 g SO2 @ 1500 torr 0.4 Volume, L • V1 0.6 0.3 0.2 0.1 0 -300 -200 -100 0 Temperature, °C 100 200 Charles’ Law – A Molecular View the pressure of gas inside and outside the balloon are the same at low temperatures, the gas molecules are not moving as fast, so they don’t hit the sides of the balloon as hard – therefore the volume is small • the pressure of gas inside and outside the balloon are the same • at high temperatures, the gas molecules are moving faster, so they hit the sides of the balloon harder – causing the volume to become larger Examples A gas has a volume of 2.57 L at 0.00°C. What was the temperature at 2.80 L? The temperature inside a balloon is raised from 25.0°C to 250.0°C. If the volume of cold air was 10.0 L, what is the volume of hot air? Avogadro’s Law Va n (constant T and P) V n Vinitial ninitial =k = Vfinal nfinal Examples A 0.225 mol sample of He has a volume of 4.65 L. How many moles must be added to give 6.48 L? A chemical reaction occurring in a cylinder equipped with a moveable piston produces 0.621 mol of a gaseous product. If the cylinder contianed 0.120 mol of gas before the reaction and had an initial volume of 2.18L, what was its volume after reaction? The Ideal Gas Law Ideal Gas Law: PV = nRT R is the gas constant and is the same for all gases . R is independent of the particular gas studied L atm R = 0.082058 K mol since the volume of a gas varies with pressure and temperature, chemists have agreed on a set of conditions to report our measurements so that comparison is easy – we call these standard conditions Standard Temperature and Pressure (STP) for Gases T = 0 °C (273.15 K) P = 1 atm Examples A 0.250 mol sample of argon gas has a volume of 9.00L at a pressure of 875 mmHg. What is the temperature (in oC) of the gas? What volume is occupied by 25.7 g of carbon dioxide gas at 25.0oC and 371 torr? Molar Volume solving the ideal gas equation for the volume of 1 mol of gas at STP gives 22.4 L ◦ 6.022 x 1023 molecules of gas we call the volume of 1 mole of gas at STP the molar volume ◦ it is important to recognize that one mole of different gases have different masses, even though they have the same volume 19 Concept problem What is the volume occupied by 2.75 moles of N2 gas at STP? Assuming ideal behavior, which of the following gas samples will have the greatest volume at STP? a. 1 g H2 b. 1 g O2 c. 1 g Ar Gas Density and Molar Mass The density of a gas is proportional to its molar mass. As the molar mass of a gas increases, so does the density of the gas. Matter often separates according to its density, with less dense matter floating on matter of higher density PV nRT m PV RT M Rearrange m P M V RT m P d M V RT 21 Examples Calculate the density of gaseous hydrogen at a pressure of 1.32 atm and a temperature of -45.0oC. A sample of gas has a mass of 0.827g. Its volume is 0.270L at a temperature of 88.0oC and a pressure of 975 mmHg. Find its molar mass Partial Pressure when gases are mixed together, their molecules behave independent of each other the pressure of a single gas in a mixture of gases is called its partial pressure we can calculate the partial pressure of a gas if the sum of the partial pressures of all the gases in the mixture equals the total pressure ◦ Dalton’s Law of Partial Pressures PT = P1 + P2 + P3 +.... 23 The partial pressure of each gas in a mixture can be calculated using the ideal gas law for two gases, A and B, mixed together nA x R x T nB x R x T PA PB V V the temperatu re and volume of everything in the mixture are the same n total n A n B n total x R x T Ptotal PA PB V 24 Example PHe=341 mmHg, PNe=112 mmHg, Ptot = 662 mmHg, V = 1.00 L, T=298 K Find the partial pressure of neon in a mixture with total pressure 3.9 atm, volume 8.7 L, temperature 598 K, and 0.17 moles Xe. Mole Fraction the fraction of the total pressure that a single gas contributes is equal to the fraction of the total number of moles that a single gas contributes the ratio of the moles of a single component to the total number of moles in the mixture is called the mole fraction, c the partial pressure of a gas is equal to the mole fraction of that gas times the total pressure PA nA Ptotal n total nA cA n total PA c A Ptotal 26 Deep Sea Divers & Partial Pressure its also possible to have too much O2, a condition called oxygen toxicity PO2 > 1.4 atm oxygen toxicity can lead to muscle spasms, tunnel vision, and convulsions its also possible to have too much N2, a condition called nitrogen narcosis also known as Rapture of the Deep when diving deep, the pressure of the air divers breathe increases – so the partial pressure of the oxygen increases at a depth of 55 m the partial pressure of O2 is 1.4 atm divers that go below 50 m use a mixture of He and O2 called heliox that contains a lower percentage of O2 than air 27 Mountain Climbing & Partial Pressure our bodies are adapted to breathe O2 at a partial pressure of 0.21 atm partial pressures of O2 lower than 0.1 atm will lead to hypoxia ◦ unconsciousness or death climbers of Mt Everest carry O2 in cylinders to prevent hypoxia ◦ on top of Mt Everest, Pair = 0.311 atm, so PO2 = 0.065 atm 28 Example Find the mole fractions and partial pressures in a 12.5 L tank with 24.2 g He and 4.32 g O2 at 298 K A diver breathes a heliox mixture with an oxygen mole fraction of 0.050. What must the total pressure be for the partial pressure of oxygen to be 0.21 atm? Collecting Gases gases are often collected by having them displace water from a container the problem is that since water evaporates, there is also water vapor in the collected gas the partial pressure of the water vapor, called the vapor pressure, depends only on the temperature 30 Vapor Pressure of Water Tro, Chemistry: A Molecular 31 Examples 1.02 L of O2 collected over water at 293 K with a total pressure of 755.2 mmHg. Find mass O2. 0.12 moles of H2 is collected over water in a 10.0 L container at 323 K. Find the total pressure.