* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download 17 Oct 08 - Seattle Central College
Survey
Document related concepts
Transcript
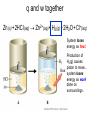
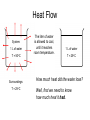
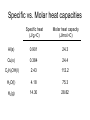
Plan for Fri, 17 Oct 08 • Lecture – Main points of 6.1 • System vs. surroundings • Kinetic vs. potential energy • Work vs. heat – Enthalpy and Calorimetry (6.2) • Quiz 3 Thermodynamic Talking Points • Energy of the universe is constant, but it is not uniformly distributed. • We can monitor the changes in energy due to physical or chemical processes by artificially partitioning the universe into a “system” and its “surroundings.” • The energy content of a system is partitioned into: – Kinetic: the energy of an object in motion – Potential: the energy of an object derived from its location in a field of force Thermodynamic Talking Points • Energy can be exchanged between the system and surroundings in two main ways: – Heat (q): energy flow between objects due to a temperature difference. • Heat is a transfer of energy manifested by changes in the microscopic thermal motions of particles • Changes in vibrations, rotations and translations of molecules • Changes in bonding patterns – Work (w): energy acting over a distance that results in the movement of an object. • Work is an energy transfer manifested by changes in the macroscopic physical variables of a system. • To accelerate a baseball from 0 mph to 60 mph, we must perform work on it. q without w • If we run an experiment in which the volume is fixed, then w = -PDV = 0 DE = q + 0 • Combustion produces gaseous products. • If we don’t allow the gaseous products to change their volume, the energy lost by the system is in terms of heat only. C(s) + O2(g) CO2(g) + heat w without q • If we run an experiment in which the volume is allowed to change, but no heat transfer is allowed, then q=0 DE = w • Isothermal expansion of a gas only loses energy as Some energy is expended by work. the system to push back the • Is any PV-work done if the atmosphere as it expands. gas expands into a vacuum? q and w together Zn (s) + 2HCl (aq) Zn2+(aq) + H2 (g) + 2H2O + Cl-(aq) System loses energy as heat. Production of H2(g) causes piston to move... system loses energy as work done on surroundings. q, PV work, and non-PV work The combustion of fuel in the space shuttle results in: 1. The emission of heat (q) 2. The expansion of the system against constant atmospheric pressure (PVwork) 3. The rapid movement of the shuttle over a distance (non-PV-work) If we could keep the shuttle from moving as its fuel combusted, what would happen to this non-PV-work energy? It would be transferred as heat to the surroundings. Internal Combustion Engine 1. Intake. Mixture of air and gas enters combustion chamber. 2. Compression. The mixtures are placed under pressure. 3. Combustion/Expansion. Spark ignites the mixture, resulting in rapid production of gaseous products and heat. The hot mixture expands, pressing on and moving parts of the engine and performing useful work. 4. Exhaust. The cooled combustion products are exhausted. Four-stroke cycle 1. Intake 2. compression 3. power 4. exhaust Work • There really isn’t anything useful about PV-work…it is simply an accounting of the energy required to change the volume of your system against constant external pressure. • In this way, the combustion of gas in your car, even though it results in the production of gases, is not PVwork… • The volume change in the system relative to the amount of gas produced is so small that the pressure does not remain constant. • Useful work comes from a clever partitioning of the heat energy (the enthalpy), which you will learn about in 162 (HAND-WAVING ALERT). Heat vs. Temperature q Temperature is an index of the average molecular KE. Heat is an exchange of energy due to a temperature difference. Go to Slide Show View (press F5) to play the video or animation. (To exit, press Esc.) This media requires PowerPoint® 2000 (or newer) and the Macromedia Flash Player (7 or higher). [To delete this message, click inside the box, click the border of the box, and then press delete.] Copyright © Houghton Mifflin Company. All rights reserved. Heat vs. Temperature • Add 100 J of heat to a collection of 100 water molecules. • Add 100 J of heat to a collection of 1,000,000 water molecules. • Which system will exhibit the greatest increase in T? – The collection of 100 water molecules. – Same amount of heat delivered, different average velocities, different final temperatures. • An extensive physical property depends directly on the amount of substance present. – Examples: enthalpy, mass, volume, length • An intensive physical property is not related to the amount of substance. – Examples: temperature, density, concentration, color Measuring DE We would like to know about the internal energy change accompanying chemical processes: DE q w Consider a process carried out at constant P: DE q w DE qP - PDV The heat transferred at const P contains a “pure heat” component and a “non-PV-work” component (HAND-WAVING ALERT)…you will learn about this in 162. The work term at const P represents the energy required to change the volume of the system…this is “PV-work.” Measuring the heat flow at const P gives you the internal energy minus PV-work. qP DE PDV Enthalpy vs. Internal Energy Recall our definition of enthalpy: H E PV Recall the heat transferred at constant P: qP DE PDV qP DE PDV DH q p DH We can track the heat flow in a process occurring at constant P and that will give us direct info about DE. Enthalpy examples • Heat transfer often accompanies physical and chemical processes. • “Heat of Fusion” - enthalpy change associated with melting H2O(s) H2O(l) DHfus = +0.334 kJ/mol ENDO • “Heat of Vaporization” - enthalpy change associated with boiling H2O(l) H2O(g) DHvap = +2.26 kJ/mol ENDO • “Heat of Reaction” - enthalpy change associated with a chemical reaction CH4(g) + 2O2(g) CO2(g) + 2H2O(g) DHrxn = -50.1 kJ/mol EXO • “Heat of Solution” – enthalpy change associated with the dissolution of ionic solids in water NH4NO3(s) NH4+(aq) + NO3-(aq) DHsoln = +82.93 kJ/mol ENDO CaCl2(s) Ca2+(aq) + 2Cl-(aq) DHsoln = -26.2 kJ/mol EXO Example • 5.00 g of ammonium nitrate is dissolved in 500. mL of water at 25.00oC. What is the final temperature of the water? NH4NO3(s) NH4+(aq) + NO3-(aq) DHsoln = +82.93 kJ/mol Heat Flow System: 1 L of water T = 50oC Surroundings: T = 25oC The liter of water is allowed to cool, until it reaches room temperature. 1 L of water T = 25oC How much heat did the water lose? Well, first we need to know how much heat it had. Heat Capacity Heat Capacity (C): • the energy required to raise the temp. of a sample of a substance by 1oC; • the ability of a substance to absorb heat. • C is an extensive property Recall: thermal energy is associated with the random motions of atoms and molecules Heat capacity gives you a measure of how much more random those motions can get. q C DT DT T f - Ti Units of C: J/oC Specific Heat Specific heat (s): the energy required to raise the temperature of 1 gram of a substance by 1oC. You can also have molar heat capacities. Specific heat is an intensive version of heat capacity (like density is an intensive version of mass) If we know the specific heat of a substance, the mass, and the temperature change, we can determine the heat flow. q s m DT C Units of s: J/oC.g Specific Heat Specifics • Typically, specific heats depend on: – Degrees of freedom: the number of different motions available to a molecule or atom – Molar mass: bigger molecules have more bonds that can vibrate – Strength of intermolecular attraction: energy can be stored in these attractions…they can vibrate too • Compounds have higher heat capacities than elements…how come? – Greater complexity more ways to store energy higher heat capacity Specific vs. Molar heat capacities Specific heat (J/g.oC) Molar heat capacity (J/mol.oC) Al(s) 0.901 24.3 Cu(s) 0.384 24.4 C2H5OH(l) 2.43 112.2 H2O(l) 4.18 75.3 H2(g) 14.30 28.82 System: 1 L of water T = 50oC The liter of water is allowed to cool, until it reaches room temperature, 25oC. How much heat did the water lose? q s m DT J m 1 L = 1000 mL = 1000 g s 4.184 g C DT T f - Ti -50C+25C = - 25C q 4.184 J 1000 g -25C -104,600 J g C -104.6 kJ