* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download QSARs and Inorganic Chemistry
Survey
Document related concepts
Transcript
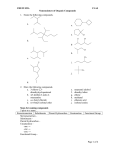
QSARs and Inorganic Chemistry • What is QSAR? • Quantitative Structure-Activity Relationship • Way to quantitatively correlate structure to physical properties or biological activity • Can you correlate systematic changes in structure and/or composition to a measurable trend in properties? • Related to the physical-organic chemistry concept of Hammett parameters • Hammett asked “How do electronic effects influence reaction equilibria, Keq?” • Original studies used the dissociation of p-substituted benzoic acids • As early example of a linear-free energy relationship Hammett Equation • Substituent Constant • Quantitative description of electron donating or withdrawing ability of a substituent -Z sz -H 0.00 -CH3 -0.17 -Cl 0.23 -OH -0.37 -NO2 0.78 -Z • Plot log (K/K0) vs s • Slope is r • If EWG increases K, r = positive • If EDG increases K, r = negative sz • These are defined values! • σz = log Kz – log KH K log = sr Ko • The Hammett equation has been modified to understand correlations in rate (k), thermodynamic values (ΔG, ΔH, ΔS), coupling constants (J, aH), etc. in place of equilibrium constants Modification of σ • Substituent constants (σ) are not “one size fits all” • Formally, σ describes electronic effects seen in parasubstituted benzoic acids • Includes both inductive and resonance effects • σ has been modified to separate out these two effects • These values are redefined as σR and σI , resonance and inductive respectively • Additional modifications to σ have been published • These include: • • • • σ. = radical intermediates σ-= negatively charged intermediates σ+ = positively charged intermediates σm = meta substituted compounds… How is this applied to inorganic chemistry? • Correlating structure and property relationships can give information regarding: • Mechanistic information • k/K changes with changing properties • Intermediates • Predictive power • Regular trends can be elucidated • Help guide future studies/synthetic efforts • Structural changes can be made to: • Ligands • Metal center Example 1: Ligand Substitution in Coordination Complexes • “Linear free‐energy relationships in semiquinone species and their Mn(II) and Cu(II) complexes” • Is there a correlation between substituent and physical properties for semiquinone complexes? • Correlation found for electronic transitions (see below) and redox potentials • Different strengths of correlations found for Cu(II) and Mn(II) complexes • Rationalized on the possible exchange pathways present in Cu(II) vs Mn(II) 10 Substituent, Z σ p-OCH3 -0.22 6 p-t-Bu -0.11 4 H 0 m-CN 0.68 0 m-NO2 0.71 -2 p-CN 0.91 -4 p-NO2 1.23 -6 -0.5 Δυ 8 2 0 0.5 σ 1 1.5 Mn(II) complex Δν versus σm, or σ−. Blue squares are the MLCT transition. Green squares are n π* transition. Δν = νH – νZ. Sloop, J. C., Shultz, D. A., Marcus, M. B. and Shepler, B. J. Phys. Org. Chem., 2012, 25, 101–109. Example 2: Property Evaluation • “Mesoporous Thin Films of “Molecular Squares” as Sensors for Volatile Organic Compounds” • Is there are correlation between electronic structure of guest and binding constant in rhenium-based molecular squares? • Rational design of materials for specific guest absorption • 350 Binding constant 300 Toluene 250 200 150 p-fluorotoluene Benzene 100 50 0 -0.2 Fluorobenzene -0.15 -0.1 -0.05 0 0.05 Binding stronger for groups with electron donating groups • The authors suggest the driving force for binding is, in part, a chargetransfer interaction between the electron-rich aromatic guests and the electron- deficient pyrazine ligands. • Guests with electron withdrawing groups have lower electron transfer rates. 0.1 σ of guest molecule Keefe, M.H.; Slone, R.V.; Hupp, J.T.; Czaplewski, K.F.; Snurr, R.Q.; Stern, C.L. Langmuir, 2000, 16, 3964–3970. Example 3: Properties of Metal Ions • “Estimating Bioconcentration Factors, Lethal Concentrations and Critical Body Residues of Metals in the Mollusks… Using Ion Characteristics” • Relating metal bioconcentration factors and LC50s to properties of metal • Regression plots of acute toxicity vs metal properties were generated: Property Equation Variance Statistical Significance Covalent Index Log LC50 = 2.8 – 0.7 Χ2mr 0.79 0.04 Hydrolysis Constant Log LC50 = 1.1 + 0.4log(KOH) 0.05 0.71 Softness Index Log LC50 = 1.0 + 0.2σP 0.31 0.33 Ionic Index Log LC50 = -0.19 + 0.25Z2/r 0.05 0.71 • Correlation of LC50 and covalent index is strong and significant! Van Kolck, M.; Huijbregts, M.A.J.; Veltman, K.; Hendriks, A.J. Environmental Toxicology and Chemistry, 27, 2008, 272–276. Further Reading • T.H. Lowry, K. S. Richardson. Mechanism and Theory in Organic Chemistry, 2nd ed. Harper Collins, 1987, pp 143 – 159. • Walker, J. Newman, M.C., Enache M. Fundamental QSARs for Metal Ions. Taylor & Francis, Boca Raton, FL, 2012. • Journals that publish QSAR/SAR related research • http://www.qsarworld.com/literature-qsar-journals.php • Review with values of σ for many organic and inorganic substituents • C. Hansch, A. Leo and R. W. Taft (1991). "A survey of Hammett substituent constants and resonance and field parameters". Chem. Rev. 91 (2): 165–195. • http://pubs.acs.org/doi/abs/10.1021/cr00002a004 Learning Outcomes • Define QSAR • Describe the Hammett equation including definitions of each variable • Give examples of how QSAR can be used to predict properties of inorganic systems