* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Viral load testing for HIV treatment monitoring in Uganda
Survey
Document related concepts
Human cytomegalovirus wikipedia , lookup
Hepatitis B wikipedia , lookup
Herpes simplex virus wikipedia , lookup
Sexually transmitted infection wikipedia , lookup
Epidemiology of HIV/AIDS wikipedia , lookup
Microbicides for sexually transmitted diseases wikipedia , lookup
Transcript
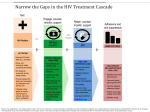
POLICY BRIEF - DECEMBER 2015 Viral load testing for HIV treatment monitoring in Uganda One of the strategic objectives of the Uganda National HIV and AIDS Strategic Plan (NSP) 2015/16-2019/20 is to strengthen monitoring of chronic HIV care and treatment through scale-up of viral load monitoring and surveillance for drug resistance (UAC 2015a). Ministry of Health has revised the Uganda National ART Guidelines to recommend routine viral load monitoring, and is in the process of rolling out viral load testing services, with a goal of expanding access to all patients on anti-retroviral therapy (ART) by 2018. This brief explains the rationale for viral load testing and the need to support Ministry of Health in implementing its scale up plan. Background M easurement of HIV viral load, which for the past two decades has been used for routine monitoring of treatment effectiveness in HIVinfected patients in developed countries, have not been widely used in ART programmes in poorresource settings such as Uganda, due to their prohibitive cost and inadequate laboratory capacity. A survey of availability of key HIV and TB medicines and diagnostics in Uganda in December 2014 and January 2015 found the majority of a sample of HIV ART centres did not have viral load machines or conduct viral load testing. Only eight centres (out of 107) reported having viral load testing machines, and only six reported to send samples to a lab at another site. At two of the sites that offered the tests clients pay for the tests (HEPS-Uganda 2015). In the absence of routine viral load testing, treatment failure has generally been defined by clinical criteria and CD4 cell count. To be diagnosed with AIDS, a person with HIV must have an AIDS-defining condition (symptom) or have a CD4 count less than 200 cells/mm³.1 World Health Organisation (WHO) prescribes the clinical manifestations of AIDS that clinicians can identify through observation, history taking and simple examination to enable them make important clinical decisions, such as initiating ART or switching a client to a stronger regimen, in resource-constrained settings where access to laboratory tests is limited or non-existent. In the Uganda case, clinical staging has been used in combination with CD4 count for many of the ART clients. A CD4 count is a lab test that measures the number of immune cells (CD4 cells) in the client’s blood and is an indicator of the strength of the client’s immune system and how much damage HIV has done to it. However, the use of the WHO clinical staging system and the CD4 count – even when used in combination – may not be effective in detecting treatment failure in a timely manner. Changes in CD4 cell counts are difficult to interpret as a result of individual variations in the immunological response to ART. In addition, studies have demonstrated the poor predictive value of the WHO immunologic criteria for virologic failure, and have shown that delayed detection of treatment failure leads to accumulation of HIV drug resistance (MOH 2014). At the same time, improvements in technology and an associated gradual reduction in prices over the recent years are making the use of viral load testing possible in low-income countries as well. And in response, WHO issued “Consolidated guidelines on the use of antiretroviral drugs for treating and preventing HIV infection” in 2013, recommending viral load monitoring as the preferred monitoring tool for the diagnosis and confirmation of ART treatment failure.2 ART access trends and HIV drug resistance T he number of people living with HIV (PLHIV) who are enrolled on ART in Uganda has increased rapidly in recent years from about 330,000 in 2011 to about 750,896 as at the end of 2014, with 125,744 being enrolled in 2014 alone.3 This figure is set to continue expanding, given that an additional 212,376 people at the end of 2014 were in HIV care at the different accredited treatment centres but yet to be enrolled on ART. Overall, it is estimated that 1.5 million Ugandans were living with HIV at the end of 2014 (UAC 2015). According to WHO (2012), some degree of HIV drug resistance is anticipated to emerge among people on treatment even when appropriate ART is provided and high levels of adherence are achieved. There are two categories of HIV drug resistance: 1) transmitted resistance, which occurs when previously uninfected individuals are infected with a drug-resistant virus; and 2) acquired resistance, which occurs when resistance mutations emerge because of drug-selective pressure in individuals receiving ART. Yet Uganda’s rapidly expanding ART programme faces a major threat from the challenge of timely identification of cases of treatment failure. Drug resistance from treatment failure as well as among people newly infected with HIV are already becoming an “increasing concern” (WHO 2012; MOH 2014). Ministry of Health reports that approximately 11.6% of patients at three ART sites have shown evidence of drug resistance. The PharmAccess African Studies to Evaluate Resistance (PASER) monitoring cohort study report for 2008-2012 found that the prevalence of transmitted drug resistance among people who have never taken antiretroviral (ARV) medication was substantially higher in Uganda in comparison to other African countries, having risen from 8.6% to 12% over the five year period.4 WHO (2012) has warned that rising population-level HIV drug resistance could potentially restrict future therapeutic options and increase treatment costs by requiring new and more expensive antiretroviral regimens. Hence WHO stresses the need to monitor HIV drug resistance and to take steps to minimise its emergence and escalation. Viral load monitoring for early detection of treatment failure H ighly active antiretroviral therapy (HAART), the current recommended treatment for HIV, entails taking a combination (regimen) of three or more ART medications from at least two different classes of medicines. While these medicines cannot cure HIV/AIDS, when taken effectively and in combination – each class uses a different mode of action to block the virus – they can suppress HIV sufficiently for a person living with HIV (PLHIV) to live a longer, healthier life. Successful combination ART should give a fall in viral load of 30-100 fold within six weeks, with the viral load falling below the limit of detection within four to six months.5 When one or more of the medicines begins to fail, usually due to poor adherence, the virus will start to reproduce again, leading to a rise in the viral load, which happens at the expense of the body’s immune system. This failure needs to be detected early so the client can be switched to a second-line treatment. The main rationale for viral load monitoring as the preferred approach compared with immunological and clinical monitoring is to provide an early and more accurate indication of treatment failure and the need to switch to second-line drugs, reducing the accumulation of drug-resistance mutations and improving clinical outcomes. Measuring viral load can also help to discriminate between treatment failure and non-adherence, and can serve as a proxy for the risk of transmission at the population level (Petti et al. 2007). WHO recommends routine viral load monitoring (every 6-12 months) to enable treatment failure to be detected earlier and more accurately. In settings with limited access to viral load testing, a targeted viral load strategy to confirm failure suspected based on immunological or clinical criteria should be used to avoid unnecessary switching to second-line ART. Targeted viral load monitoring is less costly than routine viral load testing, but as with clinical and immunological monitoring, has the potential to delay switching to second-line ART and may subsequently increase the risk of disease progression, selection of ARV drug resistance and HIV transmission.6 In addition, viral load testing combined with an adherence intervention may help patients with poor adherence to therapy maintain use7 of their firstline regimen, preventing unnecessary switches in treatment. The cost of providing second-line ART has been estimated to range between 2.4-10 times when compared to that of first line (Long et al. 2010). Viral load testing for early diagnosis of HIV infection in infants I n most resource-limited settings, children born to HIV-infected mothers are tested with an antibody test to determine their HIV infection status. These tests are only conclusive after 15-18 months because of the potential for false-positive results associated with persisting maternal antibodies. Earlier identification of HIV infection in exposed infants and referral for ART are essential. Where resources permit, most experts agree that viral load testing performed when the child is aged 4-14 weeks is optimal. In Uganda, Ministry of Health has established routine, early infant HIV testing, in which at-risk infants are identified during regular postnatal follow-up visits (e.g., vaccination visits) and are tested as early as 4-6 weeks of age. Ministry of Health plan for rolling out viral load testing M inistry of Health is in the process of rolling viral load testing to ultimately enable access to all patients on ART across all accredited facilities by 2018. The scale-up plan shows that viral load testing was to commence in March 2014. An estimated 100,000 tests were to be conducted in 2014, increasing to 200,000 in 2015, and then to 400,000 in 2016. Projections indicated that US$2 million was required in 2014, which was projected to rise to US$8.1 million in 2016. These targets were set on the basis of funding commitments from the Global Fund. They were to be revised upwards as additional funds became available. Accordingly, Uganda is set to implement the biggest roll-out of viral load testing in sub Saharan Africa following an additional US$12 million commitment from PEPFAR. Conclusion S caling up viral load testing to all ART clients in Uganda is long overdue. Viral load testing, when implemented for early and accurate detection of treatment failure, has benefits at both individual and population levels. At the individual level, it enables ART clients to be switched to more effective regimens before their health deteriorates. At the population level, it may help minimise the spread of drug resistant strains of HIV. It is also important to note that the scale-up of testing services calls for substantial investments in laboratory capacity, human resources, supplies and logistics, enhanced adherence support especially for people identified with high viral load. This calls for strengthening community structures, i.e. community linkage facilitators and counsellors, among others. This calls for commitment from not only the Ministry of Health, but also from development partners, and implementing partners. Advocacy is needed at all levels to secure and sustain this commitment. References HEPS-Uganda (2015). Assessment of the accessibility and management of essential HIV/AIDS medicines and diagnostics in Uganda Long L1, Fox M, Sanne I, and Rosen S. (2010). The high cost of second-line antiretroviral therapy for HIV/AIDS in South Africa. PubMed. 24(6):915-9. doi: 10.1097/QAD.0b013e3283360976. Ministry of Health (2014). Uganda Viral Load Monitoring Implementation Plan. Version 3.3 Petti CA, Calmy1 A, Ford N, Hirschel B, Reynolds SJ, Lynen L, Goemaere E, Garcia de la Vega1 F, Perrin L, and Rodriguez W (2007). HIV Viral Load Monitoring in Resource-Limited Regions: Optional or Necessary? Clinical Infectious Diseases. (2007) 44 (1):128-134.doi: 10.1086/510073. http://cid.oxfordjournals.org/content/44/1/128. full Uganda AIDS Commission (UAC, 2015a). National HIV and AIDS Strategic Plan 2015/16-2019/20 Uganda AIDS Commission (UAC, 2015b). The HIV and AIDS Uganda Country Progress Report 2014. WHO (2012). The HIV drug resistance report 2012 (Endnotes) 1 AIDSinfo – Educational Materials: Acquired Immunodeficiency Syndrome (AIDS). https://aidsinfo.nih.gov/educationmaterials/glossary/3/acquired-immunodeficiency-syndrome 2 http://apps.who.int/iris/bitstream/10665/85321/1/9789241505727_eng.pdf 3 The HIV and AIDS Uganda Country Progress Report 2014. http://www.unaids.org/sites/default/files/country/documents/ UGA_narrative_report_2015.pdf 4 http://www.pharmaccess.org/RunScript.asp?p=ASP\Pg0.asp 5 DHHS Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the Use of Antiretroviral Agents in HIV-1-Infected Adults and Adolescents. May 4, 2006. 6 http://www.who.int/hiv/pub/guidelines/arv2013/art/artmonitoring/en/index3.html For more information, Kenneth Mwehonge, 2015 AVAC HIV Prevention Research Advocacy Fellow, hosted at Coalition for Health Promotion & Social Development (HEPS-Uganda); Plot 351A, Balintuma Road; P.O. Box 2426, Kampala, Uganda; Tel: +256-414-270970; Email: [email protected]; Web: www.heps.or.ug