Periodic Trends
... move down a group. Electronegativity of the representative elements (Group A elements) increases as you move across a period. ...
... move down a group. Electronegativity of the representative elements (Group A elements) increases as you move across a period. ...
Serway_PSE_quick_ch45
... To reduce the value of K, more neutrons need to be absorbed, so a larger volume of the control rods must be inside the reactor core. ...
... To reduce the value of K, more neutrons need to be absorbed, so a larger volume of the control rods must be inside the reactor core. ...
File - Science Done Wright
... move down a group. Electronegativity of the representative elements (Group A elements) increases as you move across a period. ...
... move down a group. Electronegativity of the representative elements (Group A elements) increases as you move across a period. ...
Periodic Trends in Atomic Radius, Ionization Energy and
... move down a group. Electronegativity of the representative elements (Group A elements) increases as you move across a period. ...
... move down a group. Electronegativity of the representative elements (Group A elements) increases as you move across a period. ...
Periodic Trends
... Losing/gaining electrons affects size of an atom Gain an electron atom becomes negative atom becomes larger ...
... Losing/gaining electrons affects size of an atom Gain an electron atom becomes negative atom becomes larger ...
Document
... How does atomic radius change down a group? It is larger down the group. Why? Valence electrons are at higher energy levels and are ...
... How does atomic radius change down a group? It is larger down the group. Why? Valence electrons are at higher energy levels and are ...
Document
... move down a group. Electronegativity of the representative elements (Group A elements) increases as you move across a period. ...
... move down a group. Electronegativity of the representative elements (Group A elements) increases as you move across a period. ...
Chapter 7 Periodic Properties of the Elements
... Ionic size also important in predicting lattice energy and in determining the way in which ions pack in a solid. Like atomic size, it is also periodic. ...
... Ionic size also important in predicting lattice energy and in determining the way in which ions pack in a solid. Like atomic size, it is also periodic. ...
Stellar Evolution: 33.2
... • When kinetic energy is sufficiently high, coulomb repulsion that keeps the hydrogen nuclei apart can be overcome and nuclear fusion can take place. • Hydrostatic equilibrium (outward force of fusion balances gravitational inward force) occurs—a star is born. ...
... • When kinetic energy is sufficiently high, coulomb repulsion that keeps the hydrogen nuclei apart can be overcome and nuclear fusion can take place. • Hydrostatic equilibrium (outward force of fusion balances gravitational inward force) occurs—a star is born. ...
Fission vs Fusion Worksheet
... There are two main types of nuclear weapons: atomic bombs, which are powered by fission reactions similar to those in nuclear reactors [power plants], and hydrogen bombs, which derive their explosive power from fusion reactions. An atomic bomb slams together two pieces of fissionable material, usual ...
... There are two main types of nuclear weapons: atomic bombs, which are powered by fission reactions similar to those in nuclear reactors [power plants], and hydrogen bombs, which derive their explosive power from fusion reactions. An atomic bomb slams together two pieces of fissionable material, usual ...
SMP Quiz Session 1
... 4. Some black holes stars exploded. 5. These elements were made by radioacIvity in the Earth's core. ...
... 4. Some black holes stars exploded. 5. These elements were made by radioacIvity in the Earth's core. ...
Unit 2 Chemistry Review
... 11. Several factors can speed up a chemical reaction. Try identifying the following. (catalyst, temperature, surface area, and concentration) (pg.174-177) a) Cutting a solid reactant into little pieces ______________ b) Adding 6M HCl instead of 1M ____________ c) Putting the beaker on a hot plate _ ...
... 11. Several factors can speed up a chemical reaction. Try identifying the following. (catalyst, temperature, surface area, and concentration) (pg.174-177) a) Cutting a solid reactant into little pieces ______________ b) Adding 6M HCl instead of 1M ____________ c) Putting the beaker on a hot plate _ ...
25.3 section summary
... that decreases the number of slow-moving neutrons. In nuclear fusion, nuclei combine to make nuclei of greater mass. The sun’s energy is produced when hydrogen nuclei fuse to make helium nuclei. Fusion releases even more energy than fission. Fusion only occurs at very high temperature- in the excess ...
... that decreases the number of slow-moving neutrons. In nuclear fusion, nuclei combine to make nuclei of greater mass. The sun’s energy is produced when hydrogen nuclei fuse to make helium nuclei. Fusion releases even more energy than fission. Fusion only occurs at very high temperature- in the excess ...
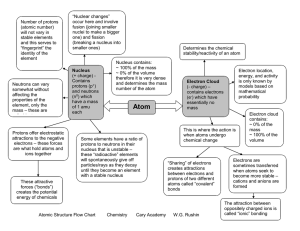
Atomic Structure Flow Chart Chemistry Cary Academy W.G. Rushin
... ~ 0% of the volume therefore it is very dense and determines the mass number of the atom ...
... ~ 0% of the volume therefore it is very dense and determines the mass number of the atom ...
Inertial electrostatic confinement
Inertial electrostatic confinement is a branch of fusion research which uses an electric field to heat a plasma to fusion conditions. Electric fields can do work on charged particles (either ions or electrons), heating them to fusion conditions. This is typically done in a sphere, with material moving radially inward, but can also be done in a cylindrical or beam geometry. The electric field can be generated using a wire grid or a non-neutral plasma cloud.