Chemistry Study Guide
... Coefficiant: the large number to the left of some of the chemical formulas indicates the number of that molecule in the equation. Ex: ...
... Coefficiant: the large number to the left of some of the chemical formulas indicates the number of that molecule in the equation. Ex: ...
Phys 282 EXP 8
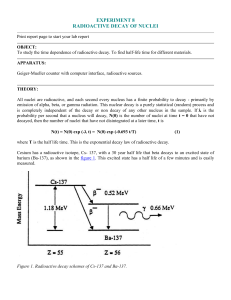
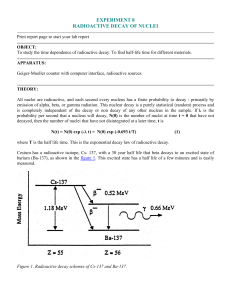
... Cesium has a radioactive isotope, Cs- 137, with a 30 year half life that beta decays to an excited state of barium (Ba-137), as shown in the figure 1. This excited state has a half life of a few minutes and is easily measured. ...
... Cesium has a radioactive isotope, Cs- 137, with a 30 year half life that beta decays to an excited state of barium (Ba-137), as shown in the figure 1. This excited state has a half life of a few minutes and is easily measured. ...
experiment 8 radioactive decay of nuclei
... Cesium has a radioactive isotope, Cs- 137, with a 30 year half life that beta decays to an excited state of barium (Ba-137), as shown in the figure 1. This excited state has a half life of a few minutes and is easily measured. ...
... Cesium has a radioactive isotope, Cs- 137, with a 30 year half life that beta decays to an excited state of barium (Ba-137), as shown in the figure 1. This excited state has a half life of a few minutes and is easily measured. ...
+1/2 and
... electron cloud. Hydrogenic atoms (or ions) have only one electron on their outside (valence) shell. This is a good approximation for these type atoms. All other atoms are far from this model, their description is very complicate. ...
... electron cloud. Hydrogenic atoms (or ions) have only one electron on their outside (valence) shell. This is a good approximation for these type atoms. All other atoms are far from this model, their description is very complicate. ...
Missing Link
... The speed of light is more basic than space and time Because the gravitational/electromagnetic interaction precedes the relative positioning of events. ...
... The speed of light is more basic than space and time Because the gravitational/electromagnetic interaction precedes the relative positioning of events. ...
lect1-4
... Physical Picture of Planck’s Hypothesis The physical background behind Planck’s proposal was that the atomic oscillators behave like simple (quantum) harmonic oscillators, which have a potential energy given by ...
... Physical Picture of Planck’s Hypothesis The physical background behind Planck’s proposal was that the atomic oscillators behave like simple (quantum) harmonic oscillators, which have a potential energy given by ...
Chapter 4: Struct of Atom
... then one has to integrate to account for it S At different times, the direction of F will be different ...
... then one has to integrate to account for it S At different times, the direction of F will be different ...
Chapter 30 Quantum Physics
... weak that only one particle is present at a time – we still see the diffraction pattern produced by constructive and destructive interference. Also, as the diffraction pattern builds, we ...
... weak that only one particle is present at a time – we still see the diffraction pattern produced by constructive and destructive interference. Also, as the diffraction pattern builds, we ...
Downlad - Inspiron Technologies
... pass between the instant the light is turned on and the time an electron is ejected from the metal This time interval is required for the electron to absorb the incident radiation before it acquires enough energy to escape from the metal Experimental Result Electrons are emitted almost instantan ...
... pass between the instant the light is turned on and the time an electron is ejected from the metal This time interval is required for the electron to absorb the incident radiation before it acquires enough energy to escape from the metal Experimental Result Electrons are emitted almost instantan ...
Electrons
... for the particular elements that are present in the gas. •Thus the emission (bright line) spectrum is a fingerprint of the gas (or gases) that is (are) emitting the light. •This is because the shells are in fixed positions, the difference in energies between them (and the wavelength of the line in i ...
... for the particular elements that are present in the gas. •Thus the emission (bright line) spectrum is a fingerprint of the gas (or gases) that is (are) emitting the light. •This is because the shells are in fixed positions, the difference in energies between them (and the wavelength of the line in i ...
PPT | 187.5 KB - Joint Quantum Institute
... level of the enclosed ultracold atoms will soon impose limits on the accuracy of the best atomic clocks. Although only important at a precision level of a part in 1015, accurate knowledge of this shift is more pertinent now that clocks are closing in on the part-per-1018 level of precision New calcu ...
... level of the enclosed ultracold atoms will soon impose limits on the accuracy of the best atomic clocks. Although only important at a precision level of a part in 1015, accurate knowledge of this shift is more pertinent now that clocks are closing in on the part-per-1018 level of precision New calcu ...
How are quantum numbers used to describe electrons
... How many orbitals in the 4th energy level? How many electrons can be in the 4th energy level? For the known elements, ________ orbitals and ______ electrons is the maximum number in energy levels 5-7. What rules are used to explain how electrons fill orbitals? Pauli exclusion principle—no two electr ...
... How many orbitals in the 4th energy level? How many electrons can be in the 4th energy level? For the known elements, ________ orbitals and ______ electrons is the maximum number in energy levels 5-7. What rules are used to explain how electrons fill orbitals? Pauli exclusion principle—no two electr ...
Set #4 - comsics
... electron typically spends about 10-8 s in an excited state before it drops to a lower state by emitting a photon. How many revolutions does an electron in an n = 2 Bohr orbit make in 10-8 s? ...
... electron typically spends about 10-8 s in an excited state before it drops to a lower state by emitting a photon. How many revolutions does an electron in an n = 2 Bohr orbit make in 10-8 s? ...
Document
... (A) increases, increases (B) decreases, decreases (C) increases, decreases (D) decreases, increases (E) More information is needed to answer this question 43. Which of the following is an acid-base neutralization reaction? (A) 2Al(s) + 3H2SO4(aq) Al2(SO4)3(aq) + 3H2(g) (B) SO2(g) + H2O(l) H2SO3( ...
... (A) increases, increases (B) decreases, decreases (C) increases, decreases (D) decreases, increases (E) More information is needed to answer this question 43. Which of the following is an acid-base neutralization reaction? (A) 2Al(s) + 3H2SO4(aq) Al2(SO4)3(aq) + 3H2(g) (B) SO2(g) + H2O(l) H2SO3( ...
DARLLENWCH Y DARN ISOD AC ATEBWCH Y CWESTIYNAU SY
... the Rutherford model was unstable because, according to classical mechanics and electromagnetic theory, any charged particle moving on a curved path emits electromagnetic radiation; thus, the electrons would lose energy and spiral into the nucleus. To remedy the stability problem, Bohr modified the ...
... the Rutherford model was unstable because, according to classical mechanics and electromagnetic theory, any charged particle moving on a curved path emits electromagnetic radiation; thus, the electrons would lose energy and spiral into the nucleus. To remedy the stability problem, Bohr modified the ...
Photoelectric effect
... The phototube contains a potassium photo-cathode and a loop of platinum wire for the anode. Potassium is used because of its low work function and platinum because of its high work function and high melting point. (What does this tell you about the contact potential generated between the anode and c ...
... The phototube contains a potassium photo-cathode and a loop of platinum wire for the anode. Potassium is used because of its low work function and platinum because of its high work function and high melting point. (What does this tell you about the contact potential generated between the anode and c ...
Electrons in Atoms
... 1. when all the possible mathematical solutions are graphed, a 3-D shape results (a "cloud" of probability) (orbital) 2. although drawn spherical, atom is not necessarily spherical C. Similarity to Bohr: based on quantized energy levels of eD. Unlike Bohr: Does not define the exact path of an e-, ju ...
... 1. when all the possible mathematical solutions are graphed, a 3-D shape results (a "cloud" of probability) (orbital) 2. although drawn spherical, atom is not necessarily spherical C. Similarity to Bohr: based on quantized energy levels of eD. Unlike Bohr: Does not define the exact path of an e-, ju ...
Electrons in Atoms
... 1. when all the possible mathematical solutions are graphed, a 3-D shape results (a "cloud" of probability) (orbital) 2. although drawn spherical, atom is not necessarily spherical C. Similarity to Bohr: based on quantized energy levels of eD. Unlike Bohr: Does not define the exact path of an e-, ju ...
... 1. when all the possible mathematical solutions are graphed, a 3-D shape results (a "cloud" of probability) (orbital) 2. although drawn spherical, atom is not necessarily spherical C. Similarity to Bohr: based on quantized energy levels of eD. Unlike Bohr: Does not define the exact path of an e-, ju ...
Chapter 9: Electrons in Atoms
... Electromagnetic radiation is a form of energy transition in which electric and magnetic fields are propagated as waves through empty space (a vacuum) or through a medium such as glass. An electric field is the region around as electrically charged particle. A magnetic field is found in the region su ...
... Electromagnetic radiation is a form of energy transition in which electric and magnetic fields are propagated as waves through empty space (a vacuum) or through a medium such as glass. An electric field is the region around as electrically charged particle. A magnetic field is found in the region su ...
X-ray fluorescence

X-ray fluorescence (XRF) is the emission of characteristic ""secondary"" (or fluorescent) X-rays from a material that has been excited by bombarding with high-energy X-rays or gamma rays. The phenomenon is widely used for elemental analysis and chemical analysis, particularly in the investigation of metals, glass, ceramics and building materials, and for research in geochemistry, forensic science and archaeology.