Quantum Cheshire Cat
... Now that we understand what weak measurement is, we need to implement it into the photon experiment in order to prove that quantum Cheshire Cat really exists. We replace detector D1 with a CCD camera that measures displacement of the beam from the central position [2]. We can realize measurement of ...
... Now that we understand what weak measurement is, we need to implement it into the photon experiment in order to prove that quantum Cheshire Cat really exists. We replace detector D1 with a CCD camera that measures displacement of the beam from the central position [2]. We can realize measurement of ...
A hands-on introduction to single photons and quantum mechanics for undergraduates
... This article describes our incorporation of single-photon experiments used in our second-year course titled “Introduction to Relativistic and Quantum Physics.” Throughout the paper, we follow the experimental-based approach we use with our students, introducing concepts as needed to explain observed ...
... This article describes our incorporation of single-photon experiments used in our second-year course titled “Introduction to Relativistic and Quantum Physics.” Throughout the paper, we follow the experimental-based approach we use with our students, introducing concepts as needed to explain observed ...
BAT
... Name: ___________________________________________ Date: ____________________ Period: ___ The final will be comprehensive, covering chapters 1-18, with the focus on chapters 10 – 18. The following questions will be what you have to Be Able To do for the final. Complete the following questions. Where ...
... Name: ___________________________________________ Date: ____________________ Period: ___ The final will be comprehensive, covering chapters 1-18, with the focus on chapters 10 – 18. The following questions will be what you have to Be Able To do for the final. Complete the following questions. Where ...
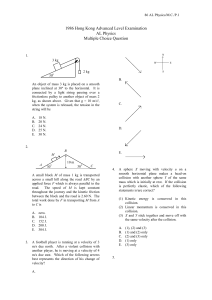
1. An object of mass 3 kg is placed on a smooth plane inclined at 30º
... A. were at rest until the switch was closed but now move along the wire with a drift velocity of a few millimetres per second. B. were at rest until the switch was closed but now move along the wire at a speed approaching the speed of light. C. had random motion onto which was imposed a drift veloci ...
... A. were at rest until the switch was closed but now move along the wire with a drift velocity of a few millimetres per second. B. were at rest until the switch was closed but now move along the wire at a speed approaching the speed of light. C. had random motion onto which was imposed a drift veloci ...
laser - SlideBoom
... laser action. A coherent beam resulted which all of the photons are in phase. ...
... laser action. A coherent beam resulted which all of the photons are in phase. ...
Bose-Einstein Condensation in Cold Atoms: a New State of Matter
... unlike photons, cannot be created or destroyed, so amplification of an atom laser (see figure 4)involves pumping more atoms into the ground quantum state through quantum coherence. Matter waves are also affected by gravity, as their masses are large compared to photons (that have zero mass).[9] Appl ...
... unlike photons, cannot be created or destroyed, so amplification of an atom laser (see figure 4)involves pumping more atoms into the ground quantum state through quantum coherence. Matter waves are also affected by gravity, as their masses are large compared to photons (that have zero mass).[9] Appl ...
CYL_Practice_harmonic_oscillator_rigid_rotor
... 8.22(a) Use the data in Exercise 8.21a to calculate the energy needed to excite a CH4 molecule from a state with 1= I to a state with 1= 2. 8.22(b) Use the data in Exercise 8.21b to calculate the energy needed to excite an SF6 molecule from a state with 1= 2 to a state with 1= 3. 8.23(a) What is the ...
... 8.22(a) Use the data in Exercise 8.21a to calculate the energy needed to excite a CH4 molecule from a state with 1= I to a state with 1= 2. 8.22(b) Use the data in Exercise 8.21b to calculate the energy needed to excite an SF6 molecule from a state with 1= 2 to a state with 1= 3. 8.23(a) What is the ...
A Physical Model for Atoms and Nuclei—Part 3
... the emitted spectra from hot solids and gases, they discovered that solids emit a continuous spectrum of electromagnetic radiation while monoatomic gases emit radiation concentrated at a number of discrete wavelengths. Each of these wavelength components is called a line, because the spectroscopes u ...
... the emitted spectra from hot solids and gases, they discovered that solids emit a continuous spectrum of electromagnetic radiation while monoatomic gases emit radiation concentrated at a number of discrete wavelengths. Each of these wavelength components is called a line, because the spectroscopes u ...
1 Chem. 152 Term Symbols for Atoms with Equivalent Electrons Prof
... Equivalent electrons have the same n and l values, so the possiblity exists that they might end up with all four quantum numbers the same, which is forbidden by the Pauli Principle. In this case you have to look at all allowable combinations of ML and MS values, and from those values infer the L and ...
... Equivalent electrons have the same n and l values, so the possiblity exists that they might end up with all four quantum numbers the same, which is forbidden by the Pauli Principle. In this case you have to look at all allowable combinations of ML and MS values, and from those values infer the L and ...
Atomic Orbitals Lab - North Carolina High School Computational
... electrons are found in fixed and quantifiable levels called orbits, or, in older terminology, shells. There are specific numbers of electrons that can be found in each orbit – 2 for the first orbit, 8 for the second, and so forth. The Bohr model is very useful for predicting the behaviors of a numbe ...
... electrons are found in fixed and quantifiable levels called orbits, or, in older terminology, shells. There are specific numbers of electrons that can be found in each orbit – 2 for the first orbit, 8 for the second, and so forth. The Bohr model is very useful for predicting the behaviors of a numbe ...
o Schrödinger equation for o Two-electron atoms. o Multi
... 3. This forces space part of wavefunction to be antisymmetric. 4. Antisymmetric space wavefunction implies a larger average distance between electrons than a symmetric function. Results as square of antisymmetric function must go to zero at the origin => probability for small separations of the tw ...
... 3. This forces space part of wavefunction to be antisymmetric. 4. Antisymmetric space wavefunction implies a larger average distance between electrons than a symmetric function. Results as square of antisymmetric function must go to zero at the origin => probability for small separations of the tw ...
Topic 3: Periodicity
... chromium. The M2+ ion is the most stable for Mn to Zn (the increased nuclear charge makes it more difficult to remove a third electron). In the higher oxidation states the elements usually not exist as a free metal ions, but covalently bonded or as a oxyanions (MnO4-). ...
... chromium. The M2+ ion is the most stable for Mn to Zn (the increased nuclear charge makes it more difficult to remove a third electron). In the higher oxidation states the elements usually not exist as a free metal ions, but covalently bonded or as a oxyanions (MnO4-). ...
untitled - PhysRevLett.111.243901
... thickness. For example, with the thinnest sample (2L=l0c 0:44) the reflection enhancement at the target arrival time and the corresponding transmission enhancement were measured to be 100% and 41%, respectively, the best experimental demonstration reported so far (see Fig. S4 in [18]). The enhance ...
... thickness. For example, with the thinnest sample (2L=l0c 0:44) the reflection enhancement at the target arrival time and the corresponding transmission enhancement were measured to be 100% and 41%, respectively, the best experimental demonstration reported so far (see Fig. S4 in [18]). The enhance ...
BWilliamsPaper - FSU High Energy Physics
... think of light as being a stream of particles that together, act like a wave, with some photons canceling each other out, and some photons building on each other. Unfortunately, this doesn’t work. Let’s say we slow down the source to emit only one photon at a time, say one every ten seconds. With o ...
... think of light as being a stream of particles that together, act like a wave, with some photons canceling each other out, and some photons building on each other. Unfortunately, this doesn’t work. Let’s say we slow down the source to emit only one photon at a time, say one every ten seconds. With o ...
Study of excited states of fluorinated copper phthalocyanine by inner
... the carbon and fluorine K-edge NEXAFS spectra on α, it is found that the average molecular tilt angle of FCuPc is 30◦ for the 50 Å-thick film on the MoS2 . This tilt angle of FCuPc is larger than that of the CuPc film on MoS2 (β = 10◦ ) [10]. A typical ion time-of-flight mass spectrum of FCuPc near ...
... the carbon and fluorine K-edge NEXAFS spectra on α, it is found that the average molecular tilt angle of FCuPc is 30◦ for the 50 Å-thick film on the MoS2 . This tilt angle of FCuPc is larger than that of the CuPc film on MoS2 (β = 10◦ ) [10]. A typical ion time-of-flight mass spectrum of FCuPc near ...
Detection of Liquefied and Gaseous form of CO2
... carbon dioxide in gaseous environment [1] and have sensitivities of 20–50 PPM [2]. The main components in the present Non-Dispersive Infrared sensor device are an infrared source (lamp), a sample chamber,Reference chamber, a Light detector chamber. Before starting the process the sample chamber is f ...
... carbon dioxide in gaseous environment [1] and have sensitivities of 20–50 PPM [2]. The main components in the present Non-Dispersive Infrared sensor device are an infrared source (lamp), a sample chamber,Reference chamber, a Light detector chamber. Before starting the process the sample chamber is f ...
LESSON No. 2 – Structure of atom
... (15) Calculate de-Broglie wavelength of an electron (mars = 9.1X10-31kg)moving at 1% speed of light (h = 6.63X10-34kg m2s-1). ...
... (15) Calculate de-Broglie wavelength of an electron (mars = 9.1X10-31kg)moving at 1% speed of light (h = 6.63X10-34kg m2s-1). ...
Ch. 5 PPT Part 3
... – So if there are two electrons in one orbital, they spin in opposite directions ...
... – So if there are two electrons in one orbital, they spin in opposite directions ...
Chemistry I
... 52. Increasing the temperature of a liquid solvent when dissolving a solid solute a. always increases the rate at which a solid solute dissolves b. often increases the amount of solid solute that can dissolve c. both a and b d. neither a and b ...
... 52. Increasing the temperature of a liquid solvent when dissolving a solid solute a. always increases the rate at which a solid solute dissolves b. often increases the amount of solid solute that can dissolve c. both a and b d. neither a and b ...
ppt
... Chladni Figures and One-Electron Atoms Double-minimum potentials generate one-dimensional bonding, A different technique is needed to address multi-dimensional problems. Solving Schroedinger’s three-dimensional differential equation might have been daunting, but it was not, because the necessary for ...
... Chladni Figures and One-Electron Atoms Double-minimum potentials generate one-dimensional bonding, A different technique is needed to address multi-dimensional problems. Solving Schroedinger’s three-dimensional differential equation might have been daunting, but it was not, because the necessary for ...
X-ray fluorescence

X-ray fluorescence (XRF) is the emission of characteristic ""secondary"" (or fluorescent) X-rays from a material that has been excited by bombarding with high-energy X-rays or gamma rays. The phenomenon is widely used for elemental analysis and chemical analysis, particularly in the investigation of metals, glass, ceramics and building materials, and for research in geochemistry, forensic science and archaeology.