AP Chemistry Summer Packet More Chapter Two and Chapter
... e. It depends on the element. Many transition metals have multiple oxidation states; the oxidation state is dependent on what it is bonding with. 73. When calcium and oxygen and, an ionic compound will be formed. Calcium, a metal, will lose electrons, and oxygen will gain electrons. The two element ...
... e. It depends on the element. Many transition metals have multiple oxidation states; the oxidation state is dependent on what it is bonding with. 73. When calcium and oxygen and, an ionic compound will be formed. Calcium, a metal, will lose electrons, and oxygen will gain electrons. The two element ...
Inorganic Chemistry 412 / 512
... Give one example of an inorganic polymer that contains N in the polymer backbone, and state one interesting/useful physical or chemical property. [5] The ones we that we discussed in class: o oligo- or polyphosphazenes (such as (NPCl2)n) – we talked about synthesis and structures of these, some are ...
... Give one example of an inorganic polymer that contains N in the polymer backbone, and state one interesting/useful physical or chemical property. [5] The ones we that we discussed in class: o oligo- or polyphosphazenes (such as (NPCl2)n) – we talked about synthesis and structures of these, some are ...
Household Items That May Contain Mercury
... Although it is not recommended to turn off the chemical process involved in making drugs, hazardous materials personnel may be met with having to make that decision based on a risk versus benefit analysis. It would be very easy to state that the process should never be turn off by qualified personne ...
... Although it is not recommended to turn off the chemical process involved in making drugs, hazardous materials personnel may be met with having to make that decision based on a risk versus benefit analysis. It would be very easy to state that the process should never be turn off by qualified personne ...
chapter 12_LO - Faculty Websites
... What is the difference between organic compounds and inorganic compounds? Why are there so many more organic compounds than inorganic compounds? You should be able to recognize and describe the different properties of organic compounds. Which elements are typically found in organic compounds? What i ...
... What is the difference between organic compounds and inorganic compounds? Why are there so many more organic compounds than inorganic compounds? You should be able to recognize and describe the different properties of organic compounds. Which elements are typically found in organic compounds? What i ...
Ionic Bonding
... compounds formed by each of the following pairs of elements: (a) strontium and oxygen (b) sodium and sulfur (c) silver and iodine (d) barium and fluorine (e) calcium and bromine (f) lithium and chlorine 10. Write the chemical formulas for the following ionic compounds: (a) mercury(II) sulfide, cinna ...
... compounds formed by each of the following pairs of elements: (a) strontium and oxygen (b) sodium and sulfur (c) silver and iodine (d) barium and fluorine (e) calcium and bromine (f) lithium and chlorine 10. Write the chemical formulas for the following ionic compounds: (a) mercury(II) sulfide, cinna ...
naming-and-formulas-chem-1-ab
... number of electrons that must be added to or removed from an atom in a combined state to convert the atom into the elemental form. ...
... number of electrons that must be added to or removed from an atom in a combined state to convert the atom into the elemental form. ...
Molecules, Compounds, and Chemical Equations (Chapter 3)
... % C and x determined from amount of CO2 produced % H and y determined from amount of H2O produced % O (if present) and z must be determined by difference ...
... % C and x determined from amount of CO2 produced % H and y determined from amount of H2O produced % O (if present) and z must be determined by difference ...
Chapt3
... % C and x determined from amount of CO2 produced % H and y determined from amount of H2O produced % O (if present) and z must be determined by difference ...
... % C and x determined from amount of CO2 produced % H and y determined from amount of H2O produced % O (if present) and z must be determined by difference ...
The structure of Matter
... O Compounds that contain only carbon and hydrogen are called hydrocarbons. O Two of the simplest hydrocarbons are methane and ethane. O Many hydrocarbons are used as fuels. ...
... O Compounds that contain only carbon and hydrogen are called hydrocarbons. O Two of the simplest hydrocarbons are methane and ethane. O Many hydrocarbons are used as fuels. ...
Elements Combine to Form Compounds
... formula (compound)…. Metals and Non- Metals determine the type of compound (Ionic or Molecular) follow the rules outline for Ionic or Molecular ...
... formula (compound)…. Metals and Non- Metals determine the type of compound (Ionic or Molecular) follow the rules outline for Ionic or Molecular ...
Naming Compounds
... Two types of compounds Molecular (covalent) compounds: formed between non- metals only this type of bonding involves sharing of electrons Examples: (we need to know) 1) Carbon Dioxide (gas) - CO2 2) Water - H2O 3) Sucrose (table sugar) – C12H22O11 ...
... Two types of compounds Molecular (covalent) compounds: formed between non- metals only this type of bonding involves sharing of electrons Examples: (we need to know) 1) Carbon Dioxide (gas) - CO2 2) Water - H2O 3) Sucrose (table sugar) – C12H22O11 ...
Summary from Organic Chemistry Packet:
... • Recognize the terms cis-, trans- isomers – Unsaturated molecules – Orientation around the double bond ...
... • Recognize the terms cis-, trans- isomers – Unsaturated molecules – Orientation around the double bond ...
Chapter Two:
... the ratios of the masses of the second element that combine with 1 gram of the first element can always be reduced to small whole numbers. ...
... the ratios of the masses of the second element that combine with 1 gram of the first element can always be reduced to small whole numbers. ...
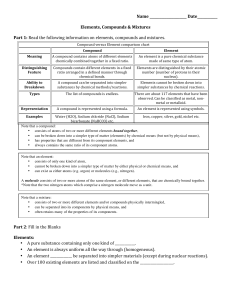
Compound vs Element chart
... • The atoms are _________________ combined in some way. Often times (but not always) they come together to form groups of atoms called molecules. • A compound is always homogeneous (uniform). • Compounds ___________________ be separated by physical means. Separating a compound requires a che ...
... • The atoms are _________________ combined in some way. Often times (but not always) they come together to form groups of atoms called molecules. • A compound is always homogeneous (uniform). • Compounds ___________________ be separated by physical means. Separating a compound requires a che ...
bonding notes for votech
... #e- lost by one particle = #e- gained by another Overall charge on a compound is always 0 ...
... #e- lost by one particle = #e- gained by another Overall charge on a compound is always 0 ...
Full research publication
... Apparently, DMSO molecules polarize the carbonyl group of the acyl-level, creating conditions for the delocalization of double bonds in the chelate stabilized by intramolecular hydrogen bonds in the steady-chelating heterocyclic NH. At the same time, the ester moiety of the molecule does not form a ...
... Apparently, DMSO molecules polarize the carbonyl group of the acyl-level, creating conditions for the delocalization of double bonds in the chelate stabilized by intramolecular hydrogen bonds in the steady-chelating heterocyclic NH. At the same time, the ester moiety of the molecule does not form a ...
compound - Coal City Unit #1
... • neutral group of atoms held together by covalent bonds • two or more atoms combined in a definite ratio • atoms may be of the same or different elements ...
... • neutral group of atoms held together by covalent bonds • two or more atoms combined in a definite ratio • atoms may be of the same or different elements ...
Affinity Chromatography using fusion proteins
... http://binfo.ym.edu.tw/bioflash/emboss/iep/iep.htm ...
... http://binfo.ym.edu.tw/bioflash/emboss/iep/iep.htm ...
a) air c) milk f) beer
... What is the ratio of oxygen in the two compounds for a fixed amount of nitrogen? ...
... What is the ratio of oxygen in the two compounds for a fixed amount of nitrogen? ...
Organometallic Chemistry at the Magnesium− Tris (8
... the quinolinate ligands of Alq3 undergo simple reduction: These calculations show that, even though the LUMO is maximized on the pyridyl ring, some increase in negative charge also accrues to the phenolic ring of the quinolinate ligands. In addition, the 1.8 eV shift for N(1s) to lower binding energ ...
... the quinolinate ligands of Alq3 undergo simple reduction: These calculations show that, even though the LUMO is maximized on the pyridyl ring, some increase in negative charge also accrues to the phenolic ring of the quinolinate ligands. In addition, the 1.8 eV shift for N(1s) to lower binding energ ...
drugs - St Marys Newry
... Also we're sure they already know this, but it never hurts to tell them that you're always around if they want to talk about it. ...
... Also we're sure they already know this, but it never hurts to tell them that you're always around if they want to talk about it. ...
List of phenyltropanes

Phenyltropanes are a family of chemical compounds originally derived from structural modification of cocaine. These compounds present many different avenues of research into therapeutic applications, particularly in addiction treatment. Uses vary depending on their construction and structure-activity relationship ranging from the treating of cocaine dependency to understanding the dopamine reward system in the human brain to treating Alzheimer's & Parkinson's diseases. (Since 2008 there have been continual additions to the list and enumerations of the plethora of types of chemicals that fall into the category of this substance profile.) Many of the compounds were first elucidated in published material by the Research Triangle Institute and are thus named with ""RTI"" serial-numbers. Similarly, a number of others are named for Sterling-Winthrop pharmaceuticals (""WIN"" serial-numbers) and Wake Forest University (""WF"" serial-numbers).