Calorimetry Tutorial
... C = heat capacity (J/K) ΔT = change in temperature (K) NOTE: when a sample gains heat (+q) ΔT is positive when a sample loses heat (-q) ΔT is negative ...
... C = heat capacity (J/K) ΔT = change in temperature (K) NOTE: when a sample gains heat (+q) ΔT is positive when a sample loses heat (-q) ΔT is negative ...
Milestones Master Study 2017
... Composition - is the type of matter that makes up the object and the way that the matter is arranged A. Physical changes do not change the composition of an object. B. Chemical changes do change the composition of an object 3 States of Matter - the physical forms of matter, which include solid, liq ...
... Composition - is the type of matter that makes up the object and the way that the matter is arranged A. Physical changes do not change the composition of an object. B. Chemical changes do change the composition of an object 3 States of Matter - the physical forms of matter, which include solid, liq ...
Atomic Structure
... at the same temperature. To measure temperature we use a thermometer. Thermometers contain some material with a property that is dependent on temperature. For example, the thermometer may contain a liquid such as mercury which expands with increasing temperature and moves up a scale. Many digital th ...
... at the same temperature. To measure temperature we use a thermometer. Thermometers contain some material with a property that is dependent on temperature. For example, the thermometer may contain a liquid such as mercury which expands with increasing temperature and moves up a scale. Many digital th ...
CP-S-HW-ch-8-detailed
... object is in equilibrium if the forces are equal in magnitude and opposite in direction. (b) The object is in equilibrium if the net torque on the object is zero. (c) The object is in equilibrium if the forces act at the same point on the object. (d) The object is in equilibrium if the net force and ...
... object is in equilibrium if the forces are equal in magnitude and opposite in direction. (b) The object is in equilibrium if the net torque on the object is zero. (c) The object is in equilibrium if the forces act at the same point on the object. (d) The object is in equilibrium if the net force and ...
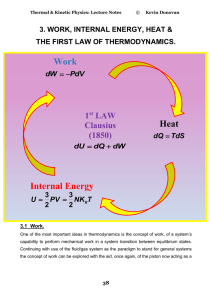
Internal Energy Work Heat
... It is important to note that in defining the work in this way it is essential to ensure that the pressure remains well defined at all times and this requires that the process must be performed quasi statically, that is, it must proceed such that at any instant the system is in an equilibrium state. ...
... It is important to note that in defining the work in this way it is essential to ensure that the pressure remains well defined at all times and this requires that the process must be performed quasi statically, that is, it must proceed such that at any instant the system is in an equilibrium state. ...
POP4e: Ch. 1 Problems
... object is in equilibrium if the forces are equal in magnitude and opposite in direction. (b) The object is in equilibrium if the net torque on the object is zero. (c) The object is in equilibrium if the forces act at the same point on the object. (d) The object is in equilibrium if the net force and ...
... object is in equilibrium if the forces are equal in magnitude and opposite in direction. (b) The object is in equilibrium if the net torque on the object is zero. (c) The object is in equilibrium if the forces act at the same point on the object. (d) The object is in equilibrium if the net force and ...
THERMODYNAMICS LECTURE NOTES
... The pressure exerted by a gas is an example of this. It results from the change in momentum of the molecules, as they collide with the wall. Here we are not concerned with the actions of individual molecules but with the time-averaged force on a given area that can be measured by a pressure gage. Fr ...
... The pressure exerted by a gas is an example of this. It results from the change in momentum of the molecules, as they collide with the wall. Here we are not concerned with the actions of individual molecules but with the time-averaged force on a given area that can be measured by a pressure gage. Fr ...
Teknologi Solid State - Universitas Brawijaya
... • A hydrogen atom, having one electron, can be covalently bonded to only one atom. However, the hydrogen atom can involve itself in an additional electrostatic bond with a second atom of highly electronegative character such as fluorine or oxygen. This second bond permits a hydrogen bond between two ...
... • A hydrogen atom, having one electron, can be covalently bonded to only one atom. However, the hydrogen atom can involve itself in an additional electrostatic bond with a second atom of highly electronegative character such as fluorine or oxygen. This second bond permits a hydrogen bond between two ...
Student Text, pp. 184-188
... concept of energy can be more easily understood by comparing it with force: force is the agent that causes change; energy is a measure of that change. For example, when a net force causes the speed of an object to change, that change is manifested as a change in the kinetic energy of the object. Rem ...
... concept of energy can be more easily understood by comparing it with force: force is the agent that causes change; energy is a measure of that change. For example, when a net force causes the speed of an object to change, that change is manifested as a change in the kinetic energy of the object. Rem ...
Atmospheric Thermodynamics
... where, DQ is the heat added to the system, and DW is the work done by the system. Both heat and work can be either positive or negative, so what is the difference between the two? What is most important to recognize is that “heat” and “work” are just names for energy. Our goal is foremost to keep an ...
... where, DQ is the heat added to the system, and DW is the work done by the system. Both heat and work can be either positive or negative, so what is the difference between the two? What is most important to recognize is that “heat” and “work” are just names for energy. Our goal is foremost to keep an ...
Module - 1: Thermodynamics
... Recall that energy can also transferred between a system and its environment as work W via a force acting on a system. Heat and work, unlike temperature, pressure, and volume, are not intrinsic properties of a system. They have meaning only as they describe the transfer of energy into or out of a sy ...
... Recall that energy can also transferred between a system and its environment as work W via a force acting on a system. Heat and work, unlike temperature, pressure, and volume, are not intrinsic properties of a system. They have meaning only as they describe the transfer of energy into or out of a sy ...
PHYSICS JUNIOR IPE IMPORTANT QUESTIONS BANK PHYSICS
... What are the different types of errors that can occur in a measurement ? Distinguish between fundamental units and derived units. What is dimensional analysis ? Express unifed atomic mass unit in kg ...
... What are the different types of errors that can occur in a measurement ? Distinguish between fundamental units and derived units. What is dimensional analysis ? Express unifed atomic mass unit in kg ...
Thermodynamic Units and Properties Summary
... units of measurement. The operator must be able to convert between these units of measurement to ensure that the plant is operating within established limits. Instrument readings may provide information in units that are different from those provided by a procedure. In this case, the operator will b ...
... units of measurement. The operator must be able to convert between these units of measurement to ensure that the plant is operating within established limits. Instrument readings may provide information in units that are different from those provided by a procedure. In this case, the operator will b ...
Thermodynamic Units and Properties
... units of measurement. The operator must be able to convert between these units of measurement to ensure that the plant is operating within established limits. Instrument readings may provide information in units that are different from those provided by a procedure. In this case, the operator will b ...
... units of measurement. The operator must be able to convert between these units of measurement to ensure that the plant is operating within established limits. Instrument readings may provide information in units that are different from those provided by a procedure. In this case, the operator will b ...
Fulltext PDF
... system of particles) add up to Zero by Newton's III Law? This is a seductive trap - a pseudo III law - that we must beware of. According to the III law, the internal forces do indeed cancel pair-wise so that the total internal force in any system is always zero. But that is not true about work. Take ...
... system of particles) add up to Zero by Newton's III Law? This is a seductive trap - a pseudo III law - that we must beware of. According to the III law, the internal forces do indeed cancel pair-wise so that the total internal force in any system is always zero. But that is not true about work. Take ...
Chapter 17. Statistical thermodynamics 2: applications
... led to the expression for qT are valid. The approximations are valid if many states are occupied, which requires V/Λ3 to be large. That will be so if Λ is small compared with the linear dimensions of the container. • For H2 at 25°C, Λ = 71 pm, which is far smaller than any conventional container is ...
... led to the expression for qT are valid. The approximations are valid if many states are occupied, which requires V/Λ3 to be large. That will be so if Λ is small compared with the linear dimensions of the container. • For H2 at 25°C, Λ = 71 pm, which is far smaller than any conventional container is ...
CENTRAL TEXAS COLLEGE SYLLABUS FOR PHYS 2425
... Satisfactory completion of this course earns the student four semester hours credit in University Physics required by most colleges of students who plan to major in physics, chemistry, certain fields of engineering and other majors which require a calculus-based physics. The student should have take ...
... Satisfactory completion of this course earns the student four semester hours credit in University Physics required by most colleges of students who plan to major in physics, chemistry, certain fields of engineering and other majors which require a calculus-based physics. The student should have take ...