From Ultracold Atoms to Condensed Matter Physics
... harmonic confinement. On top of this harmonic trap, in analogy to the periodic potential felt by electrons in a solid, one can introduce a periodic potential using a combination of lasers. The lasers that create the lattice are detuned from an optical absorption line and generate an electric dipole ...
... harmonic confinement. On top of this harmonic trap, in analogy to the periodic potential felt by electrons in a solid, one can introduce a periodic potential using a combination of lasers. The lasers that create the lattice are detuned from an optical absorption line and generate an electric dipole ...
Minimum electrophilicity principle in Lewis acid–base complexes of
... with these acids, are considered here. It is expected that more stable complexes are formed by stronger acids. Therefore, according to the MHP and MEP, for each set of complexes which are formed for a given base and different acids, the compound with the higher hardness or lesser electrophilicity be ...
... with these acids, are considered here. It is expected that more stable complexes are formed by stronger acids. Therefore, according to the MHP and MEP, for each set of complexes which are formed for a given base and different acids, the compound with the higher hardness or lesser electrophilicity be ...
Benzylamine reacts with nitrous acid to form unstable
... But aniline does not undergo H−bonding with water to a very large extent due to the presence of a large hydrophobic −C6H5 group. Hence, aniline is insoluble in water. (iii) Methylamine in water reacts with ferric chloride to precipitate hydrated ferric ...
... But aniline does not undergo H−bonding with water to a very large extent due to the presence of a large hydrophobic −C6H5 group. Hence, aniline is insoluble in water. (iii) Methylamine in water reacts with ferric chloride to precipitate hydrated ferric ...
Ionic Liquids Beyond Simple Solvents: Glimpses at the State of the
... the term “designer solvents” (see below). Early examples include all kinds of transition-metal catalysis—often with the twist that ILs are perfect media for biphasic processes.[7] Furthermore, since ILs are relatively polar and ionic interactions are strong, internal pressure effects like the ones k ...
... the term “designer solvents” (see below). Early examples include all kinds of transition-metal catalysis—often with the twist that ILs are perfect media for biphasic processes.[7] Furthermore, since ILs are relatively polar and ionic interactions are strong, internal pressure effects like the ones k ...
Stabilization of Quinapril by Incorporating Hydrogen Bonding
... pattern in the former case. In view of the above, attempts have been made to design a system wherein addition of a molecule could improve hydrogen bonding which in turn could impart stability to quinapril hydrochloride, preferably the free base. One such approach is to form a ‘co-crystal’ with a sui ...
... pattern in the former case. In view of the above, attempts have been made to design a system wherein addition of a molecule could improve hydrogen bonding which in turn could impart stability to quinapril hydrochloride, preferably the free base. One such approach is to form a ‘co-crystal’ with a sui ...
Chemical Reactivity as Described by Quantum Chemical Methods
... In recent years our group was active in the development and/or use of DFT based concepts as such or within the context of the afore mentioned and other principles. Also performance testing was one of our objectives : setting standards for computational DFT in order that it can be used for a given ty ...
... In recent years our group was active in the development and/or use of DFT based concepts as such or within the context of the afore mentioned and other principles. Also performance testing was one of our objectives : setting standards for computational DFT in order that it can be used for a given ty ...
Exact Coulomb cutoff technique for supercell calculations
... Even if, in principle, the surface terms should always be taken into account, in practice they are only relevant when we calculate energy differences between states with different total charge. These terms can be neglected in the case of a neutral cell whose lowest nonzero multipole is quadrupole.15 ...
... Even if, in principle, the surface terms should always be taken into account, in practice they are only relevant when we calculate energy differences between states with different total charge. These terms can be neglected in the case of a neutral cell whose lowest nonzero multipole is quadrupole.15 ...
The SimSoup Guide - Chris Gordon
... This can be understood as follows. Electrons orbit an atomic nucleus in one or more shells. Electron shells ...
... This can be understood as follows. Electrons orbit an atomic nucleus in one or more shells. Electron shells ...
What`s in a Name? - Department of Chemistry | Washington
... PbO) and 2. compounds containing polyatomic ions, except for the oxoacids (e.g., CaSO4, NH4NO3, KCN, but excluding H2SO4, HNO3, etc.). For the sake of naming compounds, both of these categories will be classified as ionic compounds in this tutorial. To name an ionic compound, one should name the cat ...
... PbO) and 2. compounds containing polyatomic ions, except for the oxoacids (e.g., CaSO4, NH4NO3, KCN, but excluding H2SO4, HNO3, etc.). For the sake of naming compounds, both of these categories will be classified as ionic compounds in this tutorial. To name an ionic compound, one should name the cat ...
Design and Construction of an Open Multistranded β
... precursor peptide B5cys was soluble in water and yielded a CD spectrum similar to that in methanol, B5dimer was largely insoluble. We therefore turned to an alternate sequence, B4cys, which contained many charged residues to enhance solubility. The sequence of the 35-residue B4cys peptide, illustrat ...
... precursor peptide B5cys was soluble in water and yielded a CD spectrum similar to that in methanol, B5dimer was largely insoluble. We therefore turned to an alternate sequence, B4cys, which contained many charged residues to enhance solubility. The sequence of the 35-residue B4cys peptide, illustrat ...
PDF File
... (see also ref 37): The same K1/2 values were observed in concentration dependences in which the maximal rate constant for reaction varied by more than 10-fold, which was accomplished by a 2′-H substitution at position -1 and by varying the pH (38). The affinity of S or P for the ribozyme is very hig ...
... (see also ref 37): The same K1/2 values were observed in concentration dependences in which the maximal rate constant for reaction varied by more than 10-fold, which was accomplished by a 2′-H substitution at position -1 and by varying the pH (38). The affinity of S or P for the ribozyme is very hig ...
Formation Mechanism of Non-Metallic Inclusions in
... It means that the model was initially applied to various quaternary systems, Fe–Cr–Ni–X, where X is one of the considered elements in the thermodynamic analysis (i.e. Al, N, S, Mn, Si, Ca, O). This approach was used to simplify the model and because Fe, Cr and Ni have the highest concentrations, so ...
... It means that the model was initially applied to various quaternary systems, Fe–Cr–Ni–X, where X is one of the considered elements in the thermodynamic analysis (i.e. Al, N, S, Mn, Si, Ca, O). This approach was used to simplify the model and because Fe, Cr and Ni have the highest concentrations, so ...
Molecular Compound
... 1. Determine the type and number of atoms in the molecule The formula shows one carbon atom, one iodine atom, and three hydrogen atoms. 2. Write the electron-dot notation for each type of atom in the molecule. Carbon is from Group 14 and has four valence electrons. Iodine is from Group 17 and has se ...
... 1. Determine the type and number of atoms in the molecule The formula shows one carbon atom, one iodine atom, and three hydrogen atoms. 2. Write the electron-dot notation for each type of atom in the molecule. Carbon is from Group 14 and has four valence electrons. Iodine is from Group 17 and has se ...
Main-group elements as transition metals
... distortion (that is, bending) increases as the group is descended, and it is possible to write these distorted structures using a valencebond approach (Fig. 1d), analogous to Lappert’s representations of the ethylene analogues (Fig. 1b and c). In essence, the heavier alkyne analogues also contain an ...
... distortion (that is, bending) increases as the group is descended, and it is possible to write these distorted structures using a valencebond approach (Fig. 1d), analogous to Lappert’s representations of the ethylene analogues (Fig. 1b and c). In essence, the heavier alkyne analogues also contain an ...
BIOC203W1_Lecture Slides_Enzymes
... ◦ Tyr248: Tyr248 in conjunction with Zn2+ interacts with the substrate to sensitize the susceptible peptide bond in the substrate, which is subsequently cleaved by H2O. ◦ Tyr248 and Arg145: Tyr248 and Arg145 have important hydrogen bonding interactions with the substrate and these interactions ...
... ◦ Tyr248: Tyr248 in conjunction with Zn2+ interacts with the substrate to sensitize the susceptible peptide bond in the substrate, which is subsequently cleaved by H2O. ◦ Tyr248 and Arg145: Tyr248 and Arg145 have important hydrogen bonding interactions with the substrate and these interactions ...
Chapter
... • compounds are made of atoms held together by chemical bonds • bonds are forces of attraction between atoms • the bonding attraction comes from attractions between protons and electrons ...
... • compounds are made of atoms held together by chemical bonds • bonds are forces of attraction between atoms • the bonding attraction comes from attractions between protons and electrons ...
containing complexes of aromatic amino acids
... proton transfer. An intriguing case is one in which the ternary complex contains two molecules of the amino acids (or peptides) as ligands; here, in proton transfer, one amino acid is the proton donor while the other, identical amino acid is the proton acceptor. Fig. 1 shows the dissociation of the ...
... proton transfer. An intriguing case is one in which the ternary complex contains two molecules of the amino acids (or peptides) as ligands; here, in proton transfer, one amino acid is the proton donor while the other, identical amino acid is the proton acceptor. Fig. 1 shows the dissociation of the ...
Shielding vs. Deshielding
... the electron withdrawing or releasing properties of the substituents based on relevant resonance structures and is symbolized by the letter M. The mesomeric effect is negative (-M) when the substituent is an electron-withdrawing group and the effect is positive (+M) when based on resonance the subst ...
... the electron withdrawing or releasing properties of the substituents based on relevant resonance structures and is symbolized by the letter M. The mesomeric effect is negative (-M) when the substituent is an electron-withdrawing group and the effect is positive (+M) when based on resonance the subst ...
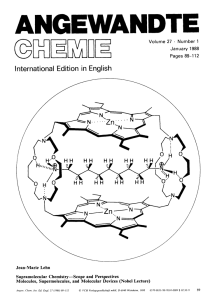
Supramolecular Chemistry—Scope and Perspectives Molecules
... coordination. Supramolecular catalysis by receptors bearing reactive groups effects bond cleavage reactions as well as synthetic bond formation via cocatalysis. Lipophilic receptor molecules act as selective carriers for various substrates and make it possible to set up coupled transport processes l ...
... coordination. Supramolecular catalysis by receptors bearing reactive groups effects bond cleavage reactions as well as synthetic bond formation via cocatalysis. Lipophilic receptor molecules act as selective carriers for various substrates and make it possible to set up coupled transport processes l ...
Effects of electric fields on ultracold Rydberg atom interactions
... many near-degenerate interactions can actually dominate or significantly change a true resonant interaction. Additionally, states which are degenerate at large R may not be degenerate at shorter ranges where several multi-polar interactions may play a role and the atomic states have been mixed by th ...
... many near-degenerate interactions can actually dominate or significantly change a true resonant interaction. Additionally, states which are degenerate at large R may not be degenerate at shorter ranges where several multi-polar interactions may play a role and the atomic states have been mixed by th ...
Chapter 3 Molecules, Compounds, and Chemical Equations
... Metal atoms: cations; nonmetal atoms: anions. • no individual molecule units, instead they have a three-dimensional array of cations and anions made of formula units • many contain polyatomic ions several atoms attached together in one ion Note: Compound must have no total charge; therefore, we mu ...
... Metal atoms: cations; nonmetal atoms: anions. • no individual molecule units, instead they have a three-dimensional array of cations and anions made of formula units • many contain polyatomic ions several atoms attached together in one ion Note: Compound must have no total charge; therefore, we mu ...
Chapter 3 Molecules Molecules, Compounds, and Chemical
... Th properties ti off the th compound are totally diff different t from f the th constituent elements. Tro, Principles of Chemistry: A Molecular Approach ...
... Th properties ti off the th compound are totally diff different t from f the th constituent elements. Tro, Principles of Chemistry: A Molecular Approach ...
Cation–pi interaction

Cation–π interaction is a noncovalent molecular interaction between the face of an electron-rich π system (e.g. benzene, ethylene, acetylene) and an adjacent cation (e.g. Li+, Na+). This interaction is an example of noncovalent bonding between a monopole (cation) and a quadrupole (π system). Bonding energies are significant, with solution-phase values falling within the same order of magnitude as hydrogen bonds and salt bridges. Similar to these other non-covalent bonds, cation–π interactions play an important role in nature, particularly in protein structure, molecular recognition and enzyme catalysis. The effect has also been observed and put to use in synthetic systems.