Worksheet
... Other Decomposition Reactions There are three other decomposition reactions you need to know. The first is the decomposition of a hydrate. When a hydrate decomposes, water is removed. See below for the decomposition of. Ex: Barium iodide dihydrate is strongly heated BaI2·2H2O(s) BaI2(s) + 2H2O( ...
... Other Decomposition Reactions There are three other decomposition reactions you need to know. The first is the decomposition of a hydrate. When a hydrate decomposes, water is removed. See below for the decomposition of. Ex: Barium iodide dihydrate is strongly heated BaI2·2H2O(s) BaI2(s) + 2H2O( ...
Unit 3: Bonding and Nomenclature Content Outline: Chemical
... C. There are 8 geometric shapes of molecules: 1. Linear (Line in one plane) a. Number of atoms bonded to the central atom = 2. b. The molecule has a bond angle of 180O. c. It has a basic molecular formula of: AB2 (“A” is one element; “B” is the other element) For example: Be F2 2. Trigonal-Planer (3 ...
... C. There are 8 geometric shapes of molecules: 1. Linear (Line in one plane) a. Number of atoms bonded to the central atom = 2. b. The molecule has a bond angle of 180O. c. It has a basic molecular formula of: AB2 (“A” is one element; “B” is the other element) For example: Be F2 2. Trigonal-Planer (3 ...
Nanoscale Forces and Their Uses in Self-Assembly
... structures and materials depends crucially on the ability to understand in quantitative detail and subsequently ‘‘engineer’’ the interparticle interactions. This Review provides a critical examination of the various interparticle forces (van der Waals, electrostatic, magnetic, molecular, and entropi ...
... structures and materials depends crucially on the ability to understand in quantitative detail and subsequently ‘‘engineer’’ the interparticle interactions. This Review provides a critical examination of the various interparticle forces (van der Waals, electrostatic, magnetic, molecular, and entropi ...
full text - pdf 452 kB
... possible explanation for this anomalous behavior is shown schematically in Fig. 8. When the metal ion associates with the crown ether, all of the oxygen atoms in the macrocycle are directed inward leaving the hydrophobic methylene groups exposed on the exterior of the ring. Thus, the hydrophobic ext ...
... possible explanation for this anomalous behavior is shown schematically in Fig. 8. When the metal ion associates with the crown ether, all of the oxygen atoms in the macrocycle are directed inward leaving the hydrophobic methylene groups exposed on the exterior of the ring. Thus, the hydrophobic ext ...
Combined Bethe-Saltpeter equations and time
... The many-body effects beyond the independent-particle approximation, in particular the core-hole photoelectron interaction and local-field effects due to the screening of the x-ray field, lead to systematic differences between the oneelectron calculations of x-ray absorption spectra 共XAS兲 and the ex ...
... The many-body effects beyond the independent-particle approximation, in particular the core-hole photoelectron interaction and local-field effects due to the screening of the x-ray field, lead to systematic differences between the oneelectron calculations of x-ray absorption spectra 共XAS兲 and the ex ...
Introduction to Organic Chemistry Curriculum
... Overview of Course (Briefly describe what students should understand and be able to do as a result of engaging in this course): This one semester course focuses on basic structure, naming, functions, and reactions of various classes of organic compounds. This course will also discuss current event t ...
... Overview of Course (Briefly describe what students should understand and be able to do as a result of engaging in this course): This one semester course focuses on basic structure, naming, functions, and reactions of various classes of organic compounds. This course will also discuss current event t ...
Bonding. A. Ionic bonds form when anions and cations arise
... Covalent bonds form when it is not possible for electrons to be transferred and so must be shared between atoms. 1. Generally this is the case when two nonmetals bond. The tendency of nonmetals is to gain electrons according to the octet rule, a very easy thing to accomplish when they bond with meta ...
... Covalent bonds form when it is not possible for electrons to be transferred and so must be shared between atoms. 1. Generally this is the case when two nonmetals bond. The tendency of nonmetals is to gain electrons according to the octet rule, a very easy thing to accomplish when they bond with meta ...
g - Porterville College Home
... a. First word is the first element in the formula. Second word is the second element with the usual modified “-ide” ending. b. Prefixes are added to each word based on the subscript of each element. Refer to Greek prefix table (e.g. tri = 3 atoms) c. Prefix exception: Mono is never used on the first ...
... a. First word is the first element in the formula. Second word is the second element with the usual modified “-ide” ending. b. Prefixes are added to each word based on the subscript of each element. Refer to Greek prefix table (e.g. tri = 3 atoms) c. Prefix exception: Mono is never used on the first ...
Coulomb and Spin-Orbit Interaction Effects in a
... In the single particle Hamiltonian, SOI is represented by a term proportional with spin and momentum. It is caused by an electric field and the magnetic component of its Lorentz transform. Its simplest case is the so-called Pauli SOI for core electrons: in this case the electric field close to the n ...
... In the single particle Hamiltonian, SOI is represented by a term proportional with spin and momentum. It is caused by an electric field and the magnetic component of its Lorentz transform. Its simplest case is the so-called Pauli SOI for core electrons: in this case the electric field close to the n ...
Lecture 2
... According to Pearson's hard soft [Lewis] acid base (HSAB) principle: Hard [Lewis] acids prefer to bind to hard [Lewis] bases and Soft [Lewis] acids prefer to bind to soft [Lewis] bases At first sight, HSAB analysis seems rather similar to the Type A and Type B system. However, Pearson classified a v ...
... According to Pearson's hard soft [Lewis] acid base (HSAB) principle: Hard [Lewis] acids prefer to bind to hard [Lewis] bases and Soft [Lewis] acids prefer to bind to soft [Lewis] bases At first sight, HSAB analysis seems rather similar to the Type A and Type B system. However, Pearson classified a v ...
van der waals forces - Department of Theoretical Physics
... the contribution among permanent dipoles is Keesom interaction, Debye interaction betveen ...
... the contribution among permanent dipoles is Keesom interaction, Debye interaction betveen ...
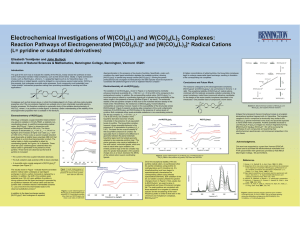
Electrochemical Investigations of W(CO) (L) and W(CO) (L) Complexes:
... moeity, such as Re(CO)3(LL), where LL = a polypyridyl ligand such as 2,2’-bipyridine (bpy), 1,10phenanthroline or related ligands, would be bridged to a non-emissive second metal center, W(CO)5 in this example, that undergoes reversible redox processes. Such compounds could potentially have “redox-t ...
... moeity, such as Re(CO)3(LL), where LL = a polypyridyl ligand such as 2,2’-bipyridine (bpy), 1,10phenanthroline or related ligands, would be bridged to a non-emissive second metal center, W(CO)5 in this example, that undergoes reversible redox processes. Such compounds could potentially have “redox-t ...
Chapter 7
... metal cation and a nonmetal anion. • Ionic compounds are electrically neutral: the total positive charge is equal to the total negative charge. • Ionic compounds are held together by the attraction of the cations to the anions, this attraction is known as an ionic bond. ...
... metal cation and a nonmetal anion. • Ionic compounds are electrically neutral: the total positive charge is equal to the total negative charge. • Ionic compounds are held together by the attraction of the cations to the anions, this attraction is known as an ionic bond. ...
Dr. Atiya Abbasi Lecture 04_ IEC_ 16 Jan.ppt
... Thus if a protein has no net charge at a certain pH (also known as isoelectric point pI) it will not interact with the charged medium. However, at a pH above its isoelectric point, a protein will bind to a positively charged medium or anion exchanger and, at a pH below its pI, the protein will behin ...
... Thus if a protein has no net charge at a certain pH (also known as isoelectric point pI) it will not interact with the charged medium. However, at a pH above its isoelectric point, a protein will bind to a positively charged medium or anion exchanger and, at a pH below its pI, the protein will behin ...
Molecules and Ions
... Ionic formulas ALWAYS have a ZERO net charge – i.e. the (+) and (-) ionic charges in ANY formula cancel. If the above rule is followed, the ionic compound must exist and is probably sitting on a shelf in the chemistry stock room! Task: Construct and name as many ionic compounds as possible from the ...
... Ionic formulas ALWAYS have a ZERO net charge – i.e. the (+) and (-) ionic charges in ANY formula cancel. If the above rule is followed, the ionic compound must exist and is probably sitting on a shelf in the chemistry stock room! Task: Construct and name as many ionic compounds as possible from the ...
Molecules and Ions
... Ionic formulas ALWAYS have a ZERO net charge – i.e. the (+) and (-) ionic charges in ANY formula cancel. If the above rule is followed, the ionic compound must exist and is probably sitting on a shelf in the chemistry stock room! Task: Construct and name as many ionic compounds as possible from the ...
... Ionic formulas ALWAYS have a ZERO net charge – i.e. the (+) and (-) ionic charges in ANY formula cancel. If the above rule is followed, the ionic compound must exist and is probably sitting on a shelf in the chemistry stock room! Task: Construct and name as many ionic compounds as possible from the ...
AtomMoleculeNaming_G1
... • The Observations That Led to the Nuclear Atom Model • The Atomic Theory Today • Elements: A First Look at the Periodic Table • Compounds: Introduction to Bonding • Formulas, Names, and Masses of Compounds • Mixtures: Classification and Separation ...
... • The Observations That Led to the Nuclear Atom Model • The Atomic Theory Today • Elements: A First Look at the Periodic Table • Compounds: Introduction to Bonding • Formulas, Names, and Masses of Compounds • Mixtures: Classification and Separation ...
chemistry-2nd-edition-julia-burdge-solution
... The molecular formula as written, C9H20, contains the simplest whole number ratio of the atoms present. In this case, the molecular formula and the empirical formula are the same. Dividing all subscripts by 2, the simplest whole number ratio of the atoms in P4O10 is P2O5. Dividing all subscripts by ...
... The molecular formula as written, C9H20, contains the simplest whole number ratio of the atoms present. In this case, the molecular formula and the empirical formula are the same. Dividing all subscripts by 2, the simplest whole number ratio of the atoms in P4O10 is P2O5. Dividing all subscripts by ...
Vorlesung Supramolekulare Chemie
... but they add up. If we now look at the enthalpies of covalent bonds, it is obvious that entropy does not play a significant role in the formation of bonds. Example: Reaction of two molecules forming one new C-C bond: ΔG = - 415 KJ/mol + 23 KJ/mol. The entropic effect accounts for less than 5% of the ...
... but they add up. If we now look at the enthalpies of covalent bonds, it is obvious that entropy does not play a significant role in the formation of bonds. Example: Reaction of two molecules forming one new C-C bond: ΔG = - 415 KJ/mol + 23 KJ/mol. The entropic effect accounts for less than 5% of the ...
Ch. 9
... – F-(fluoride), Cl-(chloride), O2-(oxide), S2-(sulfide), N3(nitride), P3-(phosphide), As3- (arsenide) ...
... – F-(fluoride), Cl-(chloride), O2-(oxide), S2-(sulfide), N3(nitride), P3-(phosphide), As3- (arsenide) ...
Lecture 2
... or cations with d electrons not available for π-bonding Soft acids are cations with a moderate positive charge (2+ or lower), Or cations with d electrons readily availbale for π-bonding ...
... or cations with d electrons not available for π-bonding Soft acids are cations with a moderate positive charge (2+ or lower), Or cations with d electrons readily availbale for π-bonding ...
Importance of Molecular Simulation for Studying Structural Properties
... Naturally occurring cationic polysaccharide known for its biocompatibility and biodegradability is chitosan (QN). The QN is a copolymer of the amino polysaccharide which is obtained by alkaline deacetylation of chitin [11], the main structural material in the exoskeletons of crustaceans and other an ...
... Naturally occurring cationic polysaccharide known for its biocompatibility and biodegradability is chitosan (QN). The QN is a copolymer of the amino polysaccharide which is obtained by alkaline deacetylation of chitin [11], the main structural material in the exoskeletons of crustaceans and other an ...
Chem 115 POGIL Worksheet - Week 10 Periodic Trends Why? The
... Properties of metalloids fall between metals and nonmetals. To behave as metals, metalloids such as Si, Ge, As would have to form very highly charged cations (Si4+, Ge4+, As5+). Such small and highly charged cations would have a strongly attractive effect on the electrons around any anions with whic ...
... Properties of metalloids fall between metals and nonmetals. To behave as metals, metalloids such as Si, Ge, As would have to form very highly charged cations (Si4+, Ge4+, As5+). Such small and highly charged cations would have a strongly attractive effect on the electrons around any anions with whic ...
Cation–pi interaction

Cation–π interaction is a noncovalent molecular interaction between the face of an electron-rich π system (e.g. benzene, ethylene, acetylene) and an adjacent cation (e.g. Li+, Na+). This interaction is an example of noncovalent bonding between a monopole (cation) and a quadrupole (π system). Bonding energies are significant, with solution-phase values falling within the same order of magnitude as hydrogen bonds and salt bridges. Similar to these other non-covalent bonds, cation–π interactions play an important role in nature, particularly in protein structure, molecular recognition and enzyme catalysis. The effect has also been observed and put to use in synthetic systems.