Spring Book Problems - Blue Valley Schools
... (a) If two objects of different temperatures are placed in contact, will heat naturally flow from the object with higher internal energy to the object with lower internal energy? (b) Is it possible for heat to flow even if the internal energies of the two objects are the same? Explain. In warm regio ...
... (a) If two objects of different temperatures are placed in contact, will heat naturally flow from the object with higher internal energy to the object with lower internal energy? (b) Is it possible for heat to flow even if the internal energies of the two objects are the same? Explain. In warm regio ...
All About Meteorology - Library Video Company
... sleet and hail that falls back to the Eart h . This process, called the water cycl e , is happening all the time! Meteorologists regularly ch e ck the temperature of the air around us by using therm o m e t e rs.They measure the amount of water vapor in the air, or the humidity, by using a tool call ...
... sleet and hail that falls back to the Eart h . This process, called the water cycl e , is happening all the time! Meteorologists regularly ch e ck the temperature of the air around us by using therm o m e t e rs.They measure the amount of water vapor in the air, or the humidity, by using a tool call ...
Chapter 5
... Yet sometimes we can see an increase in temperature with an increase in altitude, and this is called a temperature inversion. ...
... Yet sometimes we can see an increase in temperature with an increase in altitude, and this is called a temperature inversion. ...
FREE Sample Here
... Draw a diagram showing how air temperature normally changes with height. Begin at the ground and end in the upper thermosphere. Be sure to label the four main layers. Give one important characteristic of each layer. Where on your diagram would the top of Mt. Everest, the ozone layer, and the ionosph ...
... Draw a diagram showing how air temperature normally changes with height. Begin at the ground and end in the upper thermosphere. Be sure to label the four main layers. Give one important characteristic of each layer. Where on your diagram would the top of Mt. Everest, the ozone layer, and the ionosph ...
Characterization Techniques and Epitaxy
... In-situ monitoring of growth Extremely easy to prevent introduction of impurities ...
... In-situ monitoring of growth Extremely easy to prevent introduction of impurities ...
Review of heat and water movement in field soils
... there has never been a satisfactory comparison of water vapor ¯uxes measured in the ®eld with theory. At least two factors have led to this, ®rst, most of the experimental work has been laboratory oriented with steady state boundary conditions imposed and second, there have been relatively few ®eld ...
... there has never been a satisfactory comparison of water vapor ¯uxes measured in the ®eld with theory. At least two factors have led to this, ®rst, most of the experimental work has been laboratory oriented with steady state boundary conditions imposed and second, there have been relatively few ®eld ...
Lecture25-12
... liquid will vaporize. Once a sufficient number have done so, some will begin to condense back into the liquid. Equilibrium is reached when the numbers in each phase remain constant. ...
... liquid will vaporize. Once a sufficient number have done so, some will begin to condense back into the liquid. Equilibrium is reached when the numbers in each phase remain constant. ...
FREE Sample Here
... Draw a diagram showing how air temperature normally changes with height. Begin at the ground and end in the upper thermosphere. Be sure to label the four main layers. Give one important characteristic of each layer. Where on your diagram would the top of Mt. Everest, the ozone layer, and the ionosph ...
... Draw a diagram showing how air temperature normally changes with height. Begin at the ground and end in the upper thermosphere. Be sure to label the four main layers. Give one important characteristic of each layer. Where on your diagram would the top of Mt. Everest, the ozone layer, and the ionosph ...
Physics, Chapter 17: The Phases of Matter
... containing a liquid and its saturated vapor is heated to the critical temperature, the meniscus dividing the liquid from the vapor phase disappears. At temperatures above the critical temperature the substance cannot exist as a liquid; that is, no matter how great the pressure, it cannot be put into ...
... containing a liquid and its saturated vapor is heated to the critical temperature, the meniscus dividing the liquid from the vapor phase disappears. At temperatures above the critical temperature the substance cannot exist as a liquid; that is, no matter how great the pressure, it cannot be put into ...
Meteorology Today 10E
... Draw a diagram showing how air temperature normally changes with height. Begin at the ground and end in the upper thermosphere. Be sure to label the four main layers. Give one important characteristic of each layer. Where on your diagram would the top of Mt. Everest, the ozone layer, and the ionosph ...
... Draw a diagram showing how air temperature normally changes with height. Begin at the ground and end in the upper thermosphere. Be sure to label the four main layers. Give one important characteristic of each layer. Where on your diagram would the top of Mt. Everest, the ozone layer, and the ionosph ...
Weather Digital Resources
... Reading Passages ● The Weather and Climate Video Segments ● Heat Energy and the Atmosphere ● Sailing Around the World Using Global Winds Part 1 ● Sailing Around the World Using Global Winds Part 2 ● Heat Energy and the Atmosphere ● Surface Wind and Convection Currents ...
... Reading Passages ● The Weather and Climate Video Segments ● Heat Energy and the Atmosphere ● Sailing Around the World Using Global Winds Part 1 ● Sailing Around the World Using Global Winds Part 2 ● Heat Energy and the Atmosphere ● Surface Wind and Convection Currents ...
Equations of State Ideal Gas
... • When is the ideal gas assumption viable? – for a low density gas where: ∗ the gas particles take up negligible volume ∗ the intermolecular potential energy between particles is small ∗ particles act independent of one another – Under what conditions can it be used? ∗ low density ∗ high temperature ...
... • When is the ideal gas assumption viable? – for a low density gas where: ∗ the gas particles take up negligible volume ∗ the intermolecular potential energy between particles is small ∗ particles act independent of one another – Under what conditions can it be used? ∗ low density ∗ high temperature ...
thermodynamics properties of pure substances
... Some tables may not always give the internal energy. When it is not listed, the internal energy is calculated from the definition of the enthalpy as ...
... Some tables may not always give the internal energy. When it is not listed, the internal energy is calculated from the definition of the enthalpy as ...
Rankine cycle analysis 5
... The Rankine cycle is the most common of all power generation cycles and is diagrammatically depicted via Figures 1 and 2. The Rankine cycle was devised to make use of the characteristics of water as the working fluid. The cycle begins in a boiler (State 4 in figure 1), where the water is heated unti ...
... The Rankine cycle is the most common of all power generation cycles and is diagrammatically depicted via Figures 1 and 2. The Rankine cycle was devised to make use of the characteristics of water as the working fluid. The cycle begins in a boiler (State 4 in figure 1), where the water is heated unti ...
Weather
... ”W* = [ (g/To) Qs D ]1/3 where D is the boundary layer depth (or thermal depth), Qs the surface heating, and (g/To) a known buoyancy constant “ This parameter assumes that buoyancy results solely from surface heating - but if convective clouds are present then additional buoyancy will be released al ...
... ”W* = [ (g/To) Qs D ]1/3 where D is the boundary layer depth (or thermal depth), Qs the surface heating, and (g/To) a known buoyancy constant “ This parameter assumes that buoyancy results solely from surface heating - but if convective clouds are present then additional buoyancy will be released al ...
Solutions!
... About 0.0043 g of O2 can be dissolved in 100 mL of water at 20oC. Express in ppm. [memorize: 1 mL water has mass of 1 g] • ppm = 0.0043g x ...
... About 0.0043 g of O2 can be dissolved in 100 mL of water at 20oC. Express in ppm. [memorize: 1 mL water has mass of 1 g] • ppm = 0.0043g x ...
G dry
... Determining the temperature in a rising air parcel consider a rising parcel of air -->> • As the parcel rises, it will adiabatically expand and cool (recall our discussion in chapter 5 about rising parcels of air) • adiabatic - a process where the parcel temperature changes due to an expansion or c ...
... Determining the temperature in a rising air parcel consider a rising parcel of air -->> • As the parcel rises, it will adiabatically expand and cool (recall our discussion in chapter 5 about rising parcels of air) • adiabatic - a process where the parcel temperature changes due to an expansion or c ...
Weather
... What is wind and how is it measured? • Wind is air that moves horizontally, or parallel to the ground. • Over a short distance, air moves from higher pressure to lower pressure. • An anemometer is used to measure wind speed. • A wind vane or wind sock is used to measure wind ...
... What is wind and how is it measured? • Wind is air that moves horizontally, or parallel to the ground. • Over a short distance, air moves from higher pressure to lower pressure. • An anemometer is used to measure wind speed. • A wind vane or wind sock is used to measure wind ...
Wind
... released, it reacted with ammonia to create nitrogen; in addition, bacteria would also convert ammonia into nitrogen. As more plants appeared, the levels of oxygen increased significantly (while carbon dioxide levels dropped). At first it combined with various elements (such as iron), but eventually ...
... released, it reacted with ammonia to create nitrogen; in addition, bacteria would also convert ammonia into nitrogen. As more plants appeared, the levels of oxygen increased significantly (while carbon dioxide levels dropped). At first it combined with various elements (such as iron), but eventually ...
No Slide Title
... Determining the temperature in a rising air parcel consider a rising parcel of air -->> • As the parcel rises, it will adiabatically expand and cool (recall our discussion in chapter 5 about rising parcels of air) • adiabatic - a process where the parcel temperature changes due to an expansion or c ...
... Determining the temperature in a rising air parcel consider a rising parcel of air -->> • As the parcel rises, it will adiabatically expand and cool (recall our discussion in chapter 5 about rising parcels of air) • adiabatic - a process where the parcel temperature changes due to an expansion or c ...
chapter
... The diagram below shows the trends in the temperature of North America during the last 200,000 years, as estimated by scientists. What is the total number of major glacial periods that have occurred in North America in the last ...
... The diagram below shows the trends in the temperature of North America during the last 200,000 years, as estimated by scientists. What is the total number of major glacial periods that have occurred in North America in the last ...
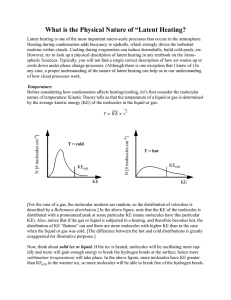
What is the Physical Nature of “Latent Heating?
... where the sum is over all of the molecules in the liquid (i) and vapor (j). If we were to simply transfer a vapor molecule of some KE from liquid to the vapor state (without any electrostatic forces and bond-breaking), the total KE would remain the same! Thus, the temperature of the liquid-vapor sys ...
... where the sum is over all of the molecules in the liquid (i) and vapor (j). If we were to simply transfer a vapor molecule of some KE from liquid to the vapor state (without any electrostatic forces and bond-breaking), the total KE would remain the same! Thus, the temperature of the liquid-vapor sys ...
Humidity

Humidity is the amount of water vapor in the air. Water vapor is the gaseous state of water and is invisible. Humidity indicates the likelihood of precipitation, dew, or fog. Higher humidity reduces the effectiveness of sweating in cooling the body by reducing the rate of evaporation of moisture from the skin. This effect is calculated in a heat index table or humidex.There are three main measurements of humidity: absolute, relative and specific. Absolute humidity is the water content of air at a given temperature expressed in gram per cubic metre. Relative humidity, expressed as a percent, measures the current absolute humidity relative to the maximum (highest point) for that temperature. Specific humidity is a ratio of the water vapor content of the mixture to the total air content on a mass basis.